Active ingredient(s):
azelastine hydrochloride (azel-as-teen hydro-klor-ide) eye drops
Consumer Medicine Information (CMI)
This leaflet provides important information about using EYEZEP. You should also speak to your doctor or pharmacist if you would like further information
or if you have any concerns or questions about using EYEZEP.
Where to find information in this leaflet:
1. Why am I using EYEZEP?
EYEZEP contains the active ingredient azelastine hydrochloride. EYEZEP belongs to a group of medicines called antihistamines. It works by blocking
the action of histamine and other substances produced by the body, which are causing
your allergies.
EYEZEP is used for the treatment and prevention of seasonal and non-seasonal (perennial)
allergic conjunctivitis, caused by allergies, such as to pollen (hay fever) or house
dust mites. The effects are red, itchy and/or watery eyes, sometimes together with sneezing or
a runny, itchy or blocked nose.
EYEZEP is not addictive.
2. What should I know before I use EYEZEP?
Warnings
Do not use EYEZEP if you have an allergy to EYEZEP or any of the ingredients listed
at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine.
Do not give EYEZEP to a child under 4 years, unless directed by the child’s doctor.
The safety and effectiveness of EYEZEP in children under 4 years have not been established.
Do not use EYEZEP after the expiry date printed on the pack.
Do not use EYEZEP after 4 weeks of first opening the bottle.
Do not use EYEZEP if the packaging is torn or shows signs of tampering.
Check with your doctor or pharmacist if you:
have kidney disease
take any medicines for any other condition
have allergies to any other substances, such as foods, preservatives or dyes.
Are wearing contact lenses.
During treatment, you may be at risk of developing certain side effects. It is important
you understand these risks and how to monitor for them. See additional information
under Section
6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor or pharmacist if you are pregnant or intend to become pregnant.
Talk to your doctor or pharmacist if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any
medicines, vitamins or supplements that you buy without a prescription from your pharmacy,
supermarket or health food shop.
Some medicines and EYEZEP may interfere with each other if significant amounts of
the eye drop is absorbed systematically. These include:
Cimetidine, a medicine used to treat stomach ulcers and some other stomach conditions.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins
or supplements you are taking and if these affect EYEZEP.
4. How do I use EYEZEP?
How much to use
Use one drop of EYEZEP in each eye every 12 hours as required. This may be increased
to one drop in each eye four times daily if required.
Follow the instructions provided with the medicine.
Do not exceed the recommended dosage.
How to use EYEZEP
Wash your hands before using EYEZEP. Gently wipe around your eyes with a tissue to
remove any moisture.

1. Remove the protective cap.
2. Unscrew the top of the bottle and check that the dropper is clean.
3. Gently pull your lower eyelid down.
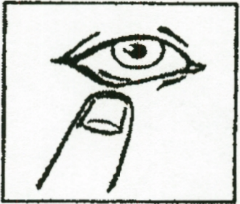
4. Carefully place one drop inside the middle of your lower eyelid. Take care not to
let the dropper touch your eye.

5. Release your lower eyelid and gently press on the inner corner of your eye against
the bridge of your nose. Then keeping your finger pressed against your nose, slowly
blink your eye a few times to spread the drop across the surface. Blot away any excess
medication.

6. Repeat this procedure for your other eye.
If you use too much EYEZEP
If you think that you have used too much EYEZEP, you may need urgent medical attention.
You should immediately:
phone the Poisons Information Centre
(Australia telephone 13 11 26) for advice, or
contact your doctor, or
go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using EYEZEP?
Things you should do
Remind any doctor, dentist or pharmacist you visit that you are using EYEZEP.
If you become pregnant while using EYEZEP, tell your doctor.
A new bottle of EYEZEP is required if used beyond 4 weeks.
The maximum duration of treatment is 6 weeks at a time. Consult your doctor if your
symptoms have not cleared up after 6 weeks.
EYEZEP should be used only when necessary.
Things you should not do
Do not use a double dose to make up for the dose that you missed.
Do not give EYEZEP to anyone else, even if they have the same condition as you.
Do not use EYEZEP to treat any other complaints unless your doctor or pharmacist tells
you to. EYEZEP is not intended for the treatment of eye infections.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how EYEZEP
affects you.
EYEZEP is unlikely to make you feel drowsy. However, if you are drowsy, do not drive
a car or operate machinery.
Drinking alcohol
Tell your doctor or pharmacist if you drink alcohol.
If you drink alcohol, any drowsiness may be worse.
Looking after your medicine
Keep your EYEZEP in a cool dry place where the temperature stays below 30°C.
Do not put EYEZEP in the fridge.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do
not store it:
in the bathroom or near a sink, or
in the car or on window sills.
Keep it where young children cannot reach it.
When to discard your medicine
Do not use EYEZEP for longer than 4 weeks after the bottle is first opened.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy
for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of
them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you
have any further questions.
Less serious/common side effects
Tell your doctor or pharmacist if you notice anything else that may be making you
feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can
report side effects to the Therapeutic Goods Administration online at
www.tga.gov.au/reporting-problems . By reporting side effects, you can help provide more information on the safety of
this medicine.
7. Product details
This medicine is available over-the-counter without a doctor's prescription.
What EYEZEP contains
|
Active ingredient
(main ingredient)
|
Azelastine (as hydrochloride) 0.0457% w/v.
Each drop contains 14 µg azelastine (as hydrochloride).
|
|
Other ingredients
(inactive ingredients)
|
Hypromellose
Disodium edetate
Benzalkonium chloride
Sorbitol solution 70% (crystallising)
Sodium hydroxide
Water for injections
|
|
Potential allergens
|
N/A
|
Do not take this medicine if you are allergic to any of these ingredients.
What EYEZEP looks like
EYEZEP (AUST R 97489) is a clear, colourless, water based solution, which comes in
a polyethylene bottle with dropper attachment. The bottle contains 6 mL of solution
which is equivalent to approximately 180 eye drops.
Who distributes EYEZEP
Viatris Pty Ltd
Level 1, 30 The Bond
30-34 Hickson Road
Millers Point, NSW 2000
www.viatris.com.au
Phone: 1800 274 276
This leaflet was prepared in Oct 2021.
EYEZEP® is a Viatris company trade mark
Eyezep_cmi\Oct21/00