2. What should I know before I use Atrovent UDV?
Do not use if you have ever had an allergic reaction to ipratropium bromide monohydrate,
atropine or any medicine like atropine or any of the ingredients listed at the end
of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines,
or are pregnant or plan to become pregnant or are breastfeeding. For more information, see
Section 2. What should I know before I use Atrovent UDV? in the full CMI.
3. What if I am taking other medicines?
4. How do I use Atrovent UDV?
Follow all directions given to you by your doctor or pharmacist carefully.
Atrovent UDV is intended for inhalation with suitable nebulising equipment and must
not be swallowed.
You will find instructions on how to use Atrovent UDV at the end of this leaflet.
5. What should I know while using Atrovent UDV?
|
Things you should do
|
Stop using Atrovent UDV and tell your doctor immediately if you get sudden tightness
of the chest, coughing, wheezing or breathlessness immediately after using Atrovent
UDV.
If you have an Asthma Action Plan that you have agreed with your doctor, follow it
closely at all times.
If you find that the usual dose of Atrovent UDV is not giving as much relief as before,
or you need to use it more often, contact your doctor so that your condition can be
checked.
|
|
Things you should not do
|
Do not take any other medicines for your breathing problems without checking with
your doctor.
Do not stop using Atrovent UDV or lower the dosage, without checking with your doctor
or pharmacist.
Do not allow the Atrovent UDV solution to enter the eyes.
|
|
Driving or using machines
|
Be careful driving or operating machinery until you know how Atrovent UDV affects
you.
Atrovent UDV may cause dizziness and blurred vision in some people. If you have any
of these symptoms, do not drive, operate machinery or do anything else that could
be dangerous. Children should be careful when performing physical activities.
|
|
Looking after your medicine
|
Keep your Atrovent UDV in a cool, dry place where the temperature stays below 25°C
and protect from light.
|
6. Are there any side effects?
Common but usually mild potential side effects include headaches, dizziness, dry or
sore mouth, throat irritation, cough, nausea, vomiting and digestion related issues
(e.g. constipation, diarrhoea, indigestion, reflux and wind). Serious potential side
effects which require immediate medical attention include allergic reaction, difficulty
in breathing, spasm of the muscles around the voice box, causing choking, heartbeat
changes, eye irritation or vision changes and difficulty in passing urine. For more
information, including what to do if you have any side effects, see Section
6. Are there any side effects? in the full CMI.
Active ingredient(s):
ipratropium bromide monohydrate
Full Consumer Medicine Information (CMI)
This leaflet provides important information about using Atrovent UDV. You should also speak to your doctor or pharmacist if you would like further information
or if you have any concerns or questions about using Atrovent UDV.
Where to find information in this leaflet:
1. Why am I using Atrovent UDV?
Atrovent UDV contains the active ingredient ipratropium bromide monohydrate. It belongs to a group of medicines called anticholinergic bronchodilators.
Atrovent UDV opens up the air passages in people suffering from asthma, chronic bronchitis
and difficulty breathing during or after surgery.
It begins to act quickly after use but may take up to 2 hours to give maximum benefit.
Ask your doctor if you have any questions about why Atrovent UDV has been prescribed
for you. Your doctor may have prescribed Atrovent UDV for another reason.
There is no evidence that Atrovent UDV is addictive.
Atrovent UDV is available only with a doctor's prescription.
Atrovent UDV is used to treat:
asthma
chronic obstructive bronchitis
people who have difficulty breathing during or after surgery using assisted ventilation.
Asthma is a lung condition where the lining of the lungs becomes inflamed (red and
swollen), making it difficult to breathe. This may be due to an allergy to house dust
mites, smoke or other irritants.
Chronic obstructive bronchitis is a lung condition that can cause difficulty in breathing,
wheeziness and constant coughing.
2. What should I know before I use Atrovent UDV?
Warnings
When you must not use Atrovent UDV
Do not use Atrovent UDV if you have an allergy to:
ipratropium bromide monohydrate
similar medicines which contain atropine or medicines like atropine
any of the ingredients listed at the end of this leaflet
any other anticholinergic medicines.
Some of the symptoms of an allergic reaction may include:
shortness of breath, wheezing or troubled breathing
swelling of the face, lips, tongue or other parts of the body
rash, itching or hives on the skin
Do not use Atrovent UDV after the expiry date printed on the pack or if the packaging
is torn or shows signs of tampering.
If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start using Atrovent UDV, contact your doctor
or pharmacist.
Before you start to use it:
Tell your doctor or pharmacist if you have allergies to any other medicines, foods,
preservatives or dyes.
Tell your doctor or pharmacist if you have or have had any of the following medical
conditions:
glaucoma (high pressure in the eye)
difficulty or pain when passing urine
constipation
cystic fibrosis
hyperreactive airways
If you have not told your doctor or pharmacist about any of the above, tell them before
you start using Atrovent UDV.
During treatment, you may be at risk of developing certain side effects. It is important
you understand these risks and how to monitor for them. See additional information
under Section
6. Are there any side effects?
Pregnancy and breastfeeding
Tell your doctor or pharmacist if you are pregnant or intend to become pregnant.
Your doctor or pharmacist will discuss the possible risks and benefits of using Atrovent
UDV during pregnancy.
Tell your doctor or pharmacist if you are breastfeeding or plan to breastfeed.
Your doctor or pharmacist will discuss the possible risks and benefits of using Atrovent
UDV during breastfeeding.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any
medicines, vitamins or supplements that you buy without a prescription from your pharmacy,
supermarket or health food shop.
Some medicines and Atrovent UDV may interfere with each other. These include:
medicines used to treat heart problems such as adrenaline
medicines used to treat asthma or a lung condition called chronic obstructive pulmonary
disease (COPD) such as theophylline, salbutamol or tiotropium
other nebuliser solutions such as disodium cromoglycate.
This is because some nebuliser solutions may not mix well with Atrovent UDV and may
need to be nebulised separately.
You may need different amounts of your medicines, or you may need to take different
medicines.
Your doctor may prescribe Atrovent UDV with other medicines that relax the air passages
for additional symptom relief.
Check with your doctor or pharmacist before mixing other nebuliser solutions with
Atrovent UDV. Some nebulised medicines may not mix well with Atrovent UDV and may
need to be nebulised separately.
Your doctor and pharmacist have more information on medicines to be careful with or
avoid while using Atrovent UDV.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins
or supplements you are taking and if these affect Atrovent UDV.
4. How do I use Atrovent UDV?
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
You will find instructions on how to use Atrovent UDV at the end of this leaflet.
If you are not sure how to use a nebuliser mask, or do not understand the instructions
on the box, ask your doctor or pharmacist for help.
Children should only use Atrovent 250 μg/1mL
UDV on medical advice and with the help of an adult.
Fit the nebuliser mask to your or your child's nose and mouth before nebulising and
inhaling Atrovent UDV to prevent the mist from contacting the eyes.
Should the mist contact the eyes, immediately flush your eyes with cold tap water
for several minutes.
If you find it difficult to breathe in and use your nebulising mask at the same time,
talk to your doctor or pharmacist. They may be able to recommend another method.
How much to use
The usual dose for adults and children is stated below:
Adults:
1 to 2 vials (250 μg to 500 μg) of Atrovent 250 μg/1mL UDV, or
1 vial of Atrovent Adult 500 μg/1mL UDV
diluted to 2-3 mL with normal saline and nebulised until the entire volume of solution
is inhaled, 4 times daily.
Children:
1 vial of Atrovent 250 μg/1mL UDV, diluted to 2-3 mL with normal saline and nebulised
until the entire volume of solution is inhaled, 4 times daily.
Depending on your condition and specific needs, your doctor may advise you to take
a different dose.
Instructions for use
Atrovent UDV are intended for inhalation with suitable nebulising equipment and must
not be swallowed or given by injection.
Diluted solutions should be freshly prepared before use.
At the end of inhalation, leftover solution from the nebuliser bowl should be discarded.
Take the following steps in using Atrovent UDV and if you have any problems, ask your
doctor or pharmacist for assistance.
1. Get your nebuliser ready by following the manufacturer's instructions and the advice
of your doctor.
2. Carefully tear a new vial from the strip. Never use one that has been opened already.
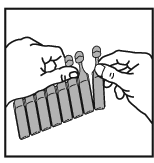
3. Open the vial by twisting off the top, always taking care to hold it in an upright
position.
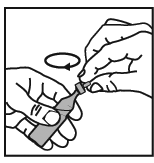
4. Squeeze the contents of the vial into the nebuliser bowl. If dilution is necessary,
this should be carried out using normal saline and as instructed by your doctor.
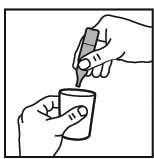
5. Dilute with saline up to a final volume of 2-3 mL.
6. Assemble the nebuliser and take the medicine or give it to your child as directed
by your doctor.
7. After nebulisation, follow the manufacturer’s instructions about cleaning your nebuliser.
When to use Atrovent UDV
If your doctor has advised you to use Atrovent UDV regularly, then use your medicine
at about the same time each day.
Using it at the same time each day will have the best effect. It will also help you
remember when to use it.
If you forget to use Atrovent UDV
If it is almost time for your next dose, skip the dose you missed and have your next
dose when you are meant to.
Otherwise, use it as soon as you remember, and then go back to using your medicine
as you would normally.
Do not use a double dose to make up for the dose that you missed.
This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to use your medicine, ask your pharmacist for some
hints.
How long to use it
Continue using Atrovent UDV for as long as your doctor tells you.
Atrovent UDV helps control your condition, but does not cure it. It is important to
keep using it even if you feel well.
If you use too much Atrovent UDV
If you think that you have used too much Atrovent UDV, you may need urgent medical
attention.
You should immediately:
phone the Poisons Information Centre
(by calling
13 11 26), or
contact your doctor, or
go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
Symptoms of an overdose may include:
fast or irregular heartbeat
dry mouth
blurred vision
5. What should I know while using Atrovent UDV?
Things you should do
Stop using Atrovent UDV and tell your doctor immediately if you get sudden tightness
of the chest, coughing, wheezing or breathlessness immediately after using Atrovent
UDV.
These may be signs of a condition called bronchospasm.
If you have an Asthma Action Plan that you have agreed with your doctor, follow it
closely at all times.
If you find that the usual dose of Atrovent UDV is not giving as much relief as before,
or you need to use it more often, contact your doctor so that your condition can be
checked. This is important to ensure your breathing problem is controlled properly.
Continue using Atrovent UDV for as long as your doctor or pharmacist tells you.
Visit your doctor regularly to check on your asthma condition.
Contact your doctor immediately if your breathing suddenly becomes more difficult
after using Atrovent UDV.
This may be a sign of an allergic reaction.
Contact your doctor immediately if you experience irritation or a feeling of having
something in the eye, or any disturbances with your sight (blurred vision, visual
halos or coloured images) together with red eyes, during or after using Atrovent UDV.
This may mean that you have developed a serious eye condition called narrow-angle
glaucoma. This can happen if the solution gets in your eyes.
Tell any other doctors, dentists, and pharmacists who are treating you that you are
using Atrovent UDV.
Tell your doctor or pharmacist that you are using Atrovent UDV if you are about to
start any new medicine.
Tell the surgeon or anesthetist that you are using Atrovent UDV if you plan to have
surgery.
It may affect other medicines during surgery.
Tell your doctor or pharmacist immediately if you become pregnant while using Atrovent
UDV.
Things you must not do
Do not take any other medicines for your breathing problems without checking with
your doctor.
Do not give Atrovent UDV to anyone else, even if they have the same condition as you.
Do not use Atrovent UDV to treat any other complaints unless your doctor or pharmacist
tells you to.
Do not stop using Atrovent UDV or lower the dosage, without checking with your doctor
or pharmacist.
Do not allow the Atrovent UDV to enter the eyes.
Driving or using machines and other things to be careful of
Be careful before you drive or operating machinery until you know how Atrovent UDV
affects you.
Atrovent UDV may cause dizziness and blurred vision in some people. If you have any
of these symptoms, do not drive, operate machinery or do anything else that could
be dangerous. Children should be careful when performing physical activities.
Looking after your medicine
Keep your Atrovent UDV in a cool, dry place where the temperature stays below 25°C
and protect from light.
Do not store it:
in the bathroom or near a sink, or
in the car or on window sills.
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it.
A locked cupboard at least one-and-a-half meters above the ground is a good place
to store medicines.
Getting rid of any unwanted medicine
If your doctor tells you to stop using Atrovent UDV or it has passed its expiry date,
ask your pharmacist what to do with any medicine that is left over.
6. Are there any side effects?
Tell your doctor or pharmacist as soon as possible if you do not feel well while you
are using Atrovent UDV.
This medicine helps most people with asthma or chronic obstructive bronchitis, but
it may have unwanted side effects in a few people. All medicines can have side effects.
If you do experience any side effects, most of them are minor and temporary. However,
some side effects may need medical attention.
Do not be alarmed by the following list of possible side effects.
You may not experience any of them.
See the information below and, if you need to, ask your doctor or pharmacist if you
have any further questions about side effects.
If you get any side effects, do not stop using the Atrovent UDV without first talking
to your doctor or pharmacist.
Less serious side effects
Very serious side effects
|
Very serious side effects
|
What to do
|
|
Mouth, throat and airways:
difficulty breathing or worsening of your breathing problems
swelling of the throat
spasm of the muscles around the voice box, causing choking
Heart:
fast or irregular heartbeat, also called palpitations
pounding heartbeat
Signs of allergic reaction:
shortness of breath
wheezing or troubled breathing
swelling of the face, lips, tongue or other parts of the body
rash, itching or hives on the skin
Eyes:
irritation or a feeling of having something in the eye
red eyes
dilated pupils
blurred vision
visual halos or coloured images
|
Call your doctor straight away or go to the Emergency Department at your nearest hospital,
if you notice any of these very serious side effects.
|
Tell your doctor or pharmacist if you notice anything else that may be making you
feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can
report side effects to the Therapeutic Goods Administration online at
www.tga.gov.au/reporting-problems . By reporting side effects, you can help provide more information on the safety of
this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop
taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Atrovent UDV contains
|
Active ingredient
(main ingredient)
|
ipratropium bromide monohydrate
|
|
Other ingredients
(inactive ingredients)
|
sodium chloride
hydrochloric acid
purified water
|
|
Potential allergens
|
None
|
Do not take this medicine if you are allergic to any of these ingredients.
Atrovent UDV does not contain lactose, sucrose, gluten, tartrazine or any other azo
dyes.
What Atrovent UDV looks like
Atrovent UDV is a clear, colourless solution, supplied as Unit Dose Vials containing
1 mL of solution in packs of 30 vials arranged as 3 individually foil-wrapped strips
of 10 unit dose vials, or 10 vials*.
* Not currently distributed in Australia.
Atrovent 250
μg
/ 1mL UDV (AUST R 17909)
Each 1 mL of Atrovent 250 µg/1mL UDV contains 261 micrograms of ipratropium bromide
monohydrate (equivalent to 250 micrograms of ipratropium bromide)
Atrovent Adult UDV 500 μg/ 1mL (AUST R 58203)
Each 1 mL of Atrovent Adult UDV contains 522 micrograms of ipratropium bromide monohydrate
(equivalent to 500 micrograms of ipratropium bromide)
Who distributes Atrovent UDV
Atrovent UDV is supplied in Australia by:
Boehringer Ingelheim Pty Limited
ABN 52 000 452 308
Sydney NSW
This leaflet was prepared in August 2025.
® Atrovent is a registered trademark of Boehringer Ingelheim
© Boehringer Ingelheim Pty Limited 2025