News-Medical speaks to Robert Schmalzigaug from Evident, to explore the field of bioluminescence imaging and the applications of the LV200 microscope.
Please can you give a brief introduction to bioluminescence microscopy?
Bioluminescence is a natural phenomenon in which light is produced by a chemical reaction. It requires an enzyme called luciferase and a substrate called luciferin. These two molecules can be found in diverse species all over the world, such as beetles and fungi but most often in marine animals like jellyfish, deep-sea fish, shrimp and so forth. However, the species most known for its bioluminescence is probably the firefly. These animals use luminescence to signal various behaviors such as defense or attraction.

The enzymes that create light are proteins encoded by the DNA of these animals. These enzymes have been isolated over the past couple of decades. When these enzymes are introduced into cells or tissues, and the substrate luciferin is added, they produce light. It is that light that can then be visualized with the microscope.
So far, researchers have imaged bioluminescence using traditional microscopes. They have looked at various biological processes from plants to mammalian cells and tissues. The one tricky thing with luminescence is that it is rather dim, so when people use traditional microscopes they have to use very long exposure times to capture an image – about 20-30 minutes. The images captured that way are often very grainy and pixelated, so it is very hard to recognize any detail.
What is bioluminescence microscopy used to visualize?
As I mentioned, luciferases are proteins and they are encoded by DNA so they can be used in a similar way to fluorescence proteins, such as green fluorescent protein (GFP) and red fluorescent protein (RFP). But the light that is created by these bioluminescence proteins is very limited and so until now, the processes that you could visualize had to be really slow – if it takes you 20 minutes to capture an image, you cannot capture a process that happens in seconds.
So, what people have used bioluminescence for until now is mostly to look at gene expression in day and night cycles, famously known as circadian rhythms. A large number of genes are up- or down-regulated over these 24-hour cycles, and these changes happen over periods of hours. Using other methods, it was possible to capture the signal of a large group of cells. However, with bioluminescence microscopy people can look at the rhythms of every individual cell and also the whole population of cells under study.
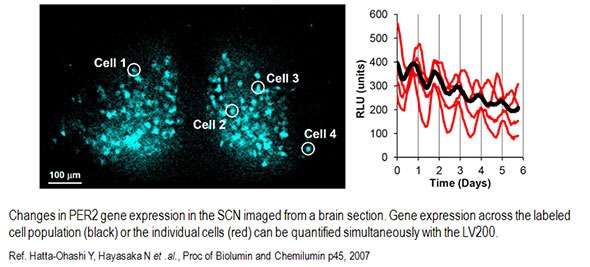
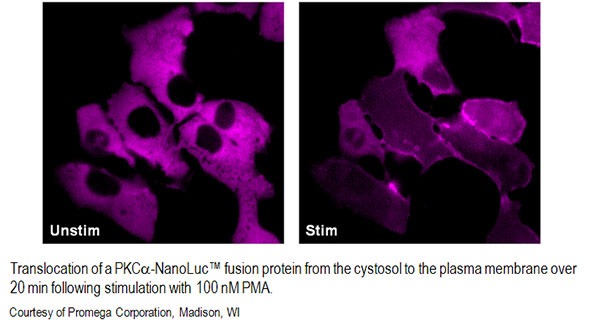
With a specialized microscope like the LV200 from Evident, together with recently developed luciferases that are much brighter, we can now study processes that happen much faster. For example, you can look at calcium signaling that happens within seconds. You can also start looking at how proteins move within cells and see changes on a minute-to-minute or a second-to-second basis. You can also start to look at how proteins interact with each other and wherein the cells they interact.
But there are some other more specialized applications. For example, scientists have started to look at radioactive decay with luminescence. In their metabolic studies, they grow cells on scintillation plates in culture dishes and observe how cells take up radiotracer molecules over time.
When the radiotracers decay, the released radioactive rays pass through the scintillation plate and are transformed into light which you then can image. So it is a different form of luminescence. Instead of having an enzyme and a substrate you have a radioactive compound that decays and produces light with the help of a scintillation plate.
These are the major applications that we currently have, but I am sure there are many more to come, now that we have both the reagents and the instrumentation available to image faster and with more detail.
How does bioluminescence microscopy differ from other types of microscopy?
There are a couple of differences. For brightfield or fluorescence microscopy you need a light source. But as I mentioned earlier, in bioluminescence you have an enzyme and a substrate which undergo a chemical reaction that produces light and so you do not need an external light source.
As for the microscope, you can use a traditional microscope, but those instruments are not designed for the very limited amount of light you get in luminescence. In traditional microscopes, light is reflected by mirrors or passes through additional lenses or prisms in the light path and you lose some of the light at each of these elements.
If you have a light source that produces a lot of light it doesn’t really matter if you lose a little bit here and there. But in bioluminescence, you only have a very limited amount of light, so it is critical to minimize any loss of precious light so that you can acquire images as efficiently and quickly as possible.
To maximize the light reaching the camera, the LV200 has a straight light path from the sample to the camera without any mirrors or prisms. Also, the light path in the LV200 is very short; it is about a third or a half of the length of the light path of traditional microscopes, so the loss of stray light is limited. These features, together with optical components that transmit maximum light, are really the key to the LV200.
What are the benefits of using bioluminescence microscopy?
There are a few key benefits to luminescence. One is that it provides very low background signal so you can see small changes in signal much more easily. The low background comes from the absence of a light source and the fact that regular mammalian cells do not express luciferases of their own. So, adding the substrate luciferin does not create light because there is no enzyme that can convert it.
In fluorescence, you sometimes have a lot of autofluorescence that comes from molecules in cells or tissues that create fluorescent light when illuminated. Autofluorescence can lead to very high background that can make it difficult to see the signal you want, but this issue does not affect luminescence microscopy.
A low background is critical to see as much as possible of your signal. If you have a high background, you can only see the tip of the signal; with luminescence you have a very low background and you can actually see almost all the signal that the sample creates. This can make a big difference particularly if you are looking at periodic changes.
If you, for example, use fluorescence you would only see the tip of the repeating peaks - the resolution between the peaks is very limited. In luminescence, you can see the entire peak, so it is much easier to discern changes and periodicity in your sample.
Also, because you have a low background you can also detect much smaller changes closer to the background level so you have a much broader detection range. For example, if you had gene expression that was only increasing very slightly you would be able to detect this using luminescence because the background is very low, whereas if you were using fluorescence the gene expression would have to go much higher before you could detect it.
Another benefit is that you do not have to worry about photobleaching or phototoxicity in absence of bright light, so cells stay healthier for a longer time when using luminescence.
Why have the applications for luminescence in live cell imaging been limited until now?
There are two parts to this. The luciferases that were originally isolated were very dim so there was very little light produced; consequently, traditional microscopes required long exposure times to acquire an image and that led to very pixelated and low-resolution images.
Also, as we talked about before, most microscopes are not designed for low light applications. They have a longer light path with mirrors and prisms that absorb part of the light.
Together, the dim luciferases and traditional microscope design have limited the applications for luminescence in live cell imaging until now.
Please can you outline the benchtop commercial bioluminescence microscope system recently introduced to North America by Evident?
The LV200 is basically an inverted microscope that is surrounded by a light-tight enclosure. The light-tight enclosure is very important because luminescence light is so dim that we are using a very sensitive camera. Any light from the surroundings or from an LED in the system would provide enough background light to possibly prevent you from detecting a signal.
 With traditional microscopes, you sometimes have to go to great lengths to create a light-tight environment. You have to turn off, cover or remove all the LED diodes on your instrument and you either have to cover the microscope or place it in a completely dark room to keep all the light out.
With traditional microscopes, you sometimes have to go to great lengths to create a light-tight environment. You have to turn off, cover or remove all the LED diodes on your instrument and you either have to cover the microscope or place it in a completely dark room to keep all the light out.
It is challenging to work in conditions where you cannot see what you are doing. You don’t have all these challenges with the LV200 and I think that is a really great benefit.
As I mentioned before, the LV200 has a light path and several optical components that are specially designed to maximize the transmission of light from the sample to the camera.
A highly sensitive camera that can detect nearly all the luminescent light coming from the sample sits at the end of the light path.
In addition, the LV200 features a light source so that you can acquire bright-field and fluorescence images as well. These reference sample images captured with bright-field or fluorescence are helpful to localize the luminescence signal to specific cells or locations in the cells.
How does the LV200 compare to other bioluminescence microscopes on the market?
The market is very small at this point, because of the limitations we talked about before and only few microscopes specialized for bioluminescence imaging are available. Researchers tell us they like the many options for customization offered with the Evident LV200.
These options include a wide selection of high-sensitivity cameras, high-performance optical components and incubator options to adapt the instrument to the customer’s needs. We also offer an advanced software solution for image capture and analysis.
Where is the LV200 currently available?
We have recently introduced the LV200 in the US and Canada. We are currently evaluating whether we will introduce it in Latin America as well. Outside of the Americas, the LV200 is available in Europe and in Japan.
Would you like to make any further comments?
We showed the instrument at the Society for Neuroscience conference in New Orleans in October and spoke with some of the attending neuroscientists there. I was surprised to hear about some groundbreaking advanced bioluminescence microscopy applications for research. These researchers were very excited about the LV200 and the possibilities it offers to capture high-resolution bioluminescence images.
The field of live cell microscopy is heavily dominated by fluorescence and the use of fluorescent proteins. Researchers have developed many methods and are very accustomed to using these fluorescent proteins in their studies.
What is exciting is that, because both fluorescent proteins and luciferases are encoded by DNA, it will be relatively easy for scientists to adapt their work to take advantage of bioluminescence and create new methods and applications based on the use of luciferases rather than fluorescent proteins.
They will, therefore, be able to take advantage of the benefits of bioluminescence rather quickly. So this is a very exciting time to introduce the LV200 to the market. It is going to be very interesting to see what applications are going to come out of this going forward.
There are two applications already out there for bioluminescence that can greatly profit from the LV200 microscope. Quite a few cancer researchers study how cancer cells injected into a mouse or rat form tumors and how the disease progresses.
They use bioluminescence imaging systems that can take images of the entire mouse and show the size and location of the tumor. However, the resolution of these systems is very limited. Now, with the LV200 microscope, they can follow the disease progression in the entire mouse, and then at any point isolate the cancerous tissue, section it and then look at the tissues on a cellular level.
In a completely unrelated application, a lot of pharma companies are screening for drugs with bioluminescence assays. In these assays, cells are grown in multi-well plates and the instruments detecting the luminescence can only read the signal from an entire well.
These instruments give you a signal for a population but they don’t show you the differences between individual cells. With the LV200 microscope, scientists can get both the signal from the population and the individual cells. In addition, the microscope could be very helpful in developing new assays and confirm that the luminescence signal is reporting what it is supposed to.
For these two applications and in many others, the new microscope system is a powerful instrument that can help them by providing complementary data to what they are receiving through other research means, offering a better and more comprehensive idea of what cells under observation are actually doing. Thus it is not just a new tool for cell biologists; it can also be a complementary tool for people looking at cancer research or drug development.
Where can readers find more information?
LV200 from Evident
About Robert Schmalzigaug
 Robert Schmalzigaug is a product manager at Evident Corporation and manages several product lines for the Scientific Equipment Group.
Robert Schmalzigaug is a product manager at Evident Corporation and manages several product lines for the Scientific Equipment Group.
He was previously a biomedical research scientist at Duke University and is highly experienced in cell biology and neurobiology applications. Robert developed animal models for post-traumatic stress disorder and learning and memory disabilities.
Robert holds a PhD in Cell Biology and has studied at the University of Zürich and the University of Copenhagen. He also holds an MBA from Duke University’s Fuqua School of Business.