Introduction
Metabolism of the inositol hexanicotinate
Clinical value and dosing
References
Further reading
Inositol hexanicotinate (also known as inositol hexaniacinate or inositol nicotinate) is the hexanicotinic acid ester of meso-inositol. This chemical compound consists of six molecules of nicotinic acid (niacin) with an inositol molecule in the center of the structure.
It is a popular form of food supplement where it acts as a source of niacin (vitamin B3), one of the essential human nutrients. Although the inositol hexanicotinate complex does not represent an essential nutrient, niacin is pivotal to cellular metabolism, playing the important role in coenzyme function and oxidation-reduction reactions.
 Inositol hexanicotinate is a popular form of food supplement where it acts as a source of niacin (vitamin B3), one of the essential human nutrients. Image Credit: Danijela Maksimovic/Shutterstock
Inositol hexanicotinate is a popular form of food supplement where it acts as a source of niacin (vitamin B3), one of the essential human nutrients. Image Credit: Danijela Maksimovic/Shutterstock
Metabolism of the inositol hexanicotinate
Inositol hexanicotinate is (at least partially) absorbed intact and subsequently hydrolyzed in the body, resulting in a free nicotinic acid and inositol. Although gastrointestinal absorption of inositol hexanicotinate can vary significantly, approximately 70% of an orally ingested dose absorbs into the bloodstream.
Oral administration of inositol hexanicotinate results in a steady increase in the level of free nicotinic acid in blood and plasma. The compound appears to be metabolized slowly, with serum levels of nicotinic acid peaking roughly 6-10 hours after ingestion, compared to the intake of free nicotinic acid when plasma levels can peak after merely half an hour.
The extent of hydrolysis of inositol hexanicotinate appears to be very low; therefore, differences in plasma levels of free nicotinic acid after administering similar oral doses of inositol hexanicotinate and free nicotinic acid may be responsible for different effects observed in clinical studies. In fact, the observed effects for inositol hexanicotinate may not be related to its total nicotinic acid content, but rather to a direct effect of the compound on the organism.
The beneficial effects of free nicotinic acid and extended-release nicotinic acid in lowering the concentration of lipids are well established, but the beneficial effects of inositol hexanicotinate on serum lipids depends on the uptake and subsequent release of the nicotinic acid moieties from the inositol hexanicotinate molecule. Considering the aforementioned low extent of hydrolysis, this mechanism is insufficient to alter plasma lipid profiles significantly.
Clinical value and dosing
Inositol hexanicotinate has a proven beneficial effect on endothelium-dependent vasodilatation (widening of blood vessels), thus, it improves blood flow in conditions where it is compromised. In Europe, it is often prescribed for the symptomatic relief of Raynaud’s phenomenon and severe intermittent claudication. A common dose of inositol hexanicotinate for these conditions is 3 g per day, which can be increased to 4 g per day if necessary.
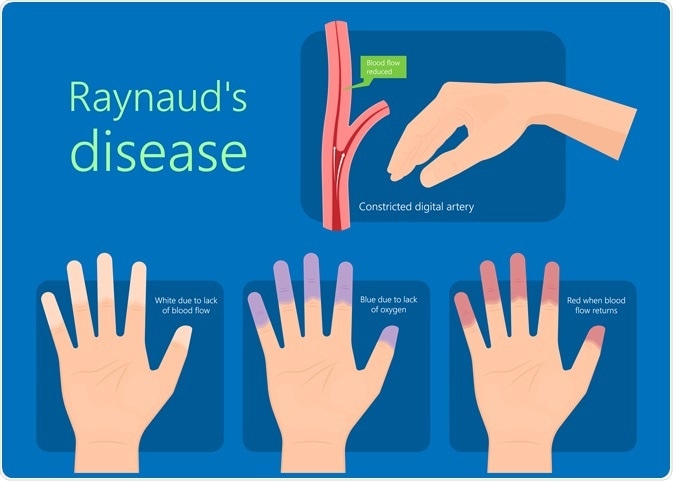 In some places inositol hexanicotinate is prescribed for the symptomatic relief of Raynaud’s phenomenon and severe intermittent claudication. Image Credit: rumruay/Shutterstock
In some places inositol hexanicotinate is prescribed for the symptomatic relief of Raynaud’s phenomenon and severe intermittent claudication. Image Credit: rumruay/Shutterstock
Nicotinic acid given as inositol hexanicotinate in a dose of 10 mg per day would amount to a daily dose of 11 mg inositol hexanicotinate, with the release of 2.4 mg inositol upon hydrolysis. Since the estimated normal dietary intake of inositol amounts up to 1500 mg of myoinositol (naturally occurring inositol) per day, the European Food Safety Authority panel concluded that the intake of 2.4 mg inositol/day, resulting from the abovementioned dose of 11 mg per day, would not be of safety concern.
Inositol hexanicotinate acts as a slow-release supply of nicotinic acid; hence the flushing effect is not a likely event when inositol hexanicotinate is used as a source of niacin due to the slow hydrolysis of the nicotinic acid molecules from the inositol. For that reason, this compound is often described as “no-flush niacin”. No adverse effects have been observed in clinical trials using inositol hexanicotinate up to 4000 mg daily for 3 months.
Even though there is no genotoxicity data available on inositol hexanicotinate, there are no safety concerns. As already mentioned, inositol hexanicotinate is hydrolyzed to inositol and nicotinic acid, which are both endogenous compounds that occur in several dietary products. In addition, the safety of nicotinic acid has been thoroughly evaluated by various authorities – including the Scientific Committee on Food (SCF).
References
- Inositol hexanicotinate (inositol hexaniacinate) as a source of niacin (vitamin B3) added for nutritional purposes in food supplements. Efsa. Last Accessed: 23.01.2023. http://www.efsa.europa.eu/en/efsajournal/pub/949.htm
- Joseph M Keenan. (2012). Wax-matrix extended-release niacin vs inositol hexanicotinate: a comparison of wax-matrix, extended-release niacin to inositol hexanicotinate "no-flush" niacin in persons with mild to moderate dyslipidemia. http://www.ncbi.nlm.nih.gov/pubmed/23351578 doi: 10.1016/j.jacl.2012.10.004.
- Kroner Z. (2011). Vitamins and Minerals. ABC-CLIO, LLC, Santa Barbara, California, pp. 227-234.
- Stargrove MB, Treasure J, McKee DL. (2008). Herb, Nutrient, and Drug Interactions: Clinical Implications and Therapeutic Strategies. Elsevier Health Sciences, pp. 281-305.
Further Reading
Last Updated: Jul 14, 2023