Skip to:
Atorvastatin statin medication prescribed to reduce choleserol. Image Credit: riccar / Shutterstock
What is Atorvastatin Used for?
Atorvastatin belongs to a class of drugs termed “statins”, which are competitive inhibitors of 3-hydroxy-3-methylglutaryl-coenzyme (HMG-CoA) reductase – an enzyme involved in the production of cholesterol.
Atorvastatin is used to reduce the levels of total blood cholesterol, low density lipoprotein (LD, Apoprotein (Apo)-B, and triglycerides. LDL is also referred to as "bad" cholesterol and Apo B is a marker for cardiovascular risk.
Atorvastatin also increases the levels of HDL cholesterol (good cholesterol) and helps prevent cardiovascular diseases. It is also approved for dysbetalipoproteinemia – a rare hereditary condition associated with elevated cholesterol and triglyceride levels.
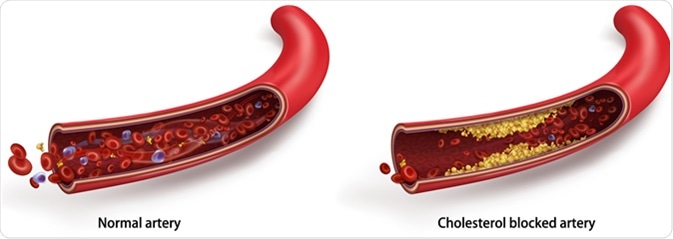
Normal blood flow. The accumulation of cholesterol in the blood vessels. Atherosclerotic plaque. Vector 3d illustration - Illustration Credit: Studiovin / Shutterstock
How Does Atorvastatin Work?
HMG-CoA reductase is a key enzyme involved in the production of cholesterol. The enzyme converts 3-hydroxy-3-methylglutaryl-coenzyme A to mevalonate, a precursor of cholesterol in the liver.
Atorvastatin inhibits HMG-CoA reductase from performing this conversion and hence disrupts the synthesis of cholesterol in the body. It also increases the number of LDL receptors on the cell; which leads to increased LDL uptake by the cells, and lowers the plasma LDL levels. Atorvastatin has a shape complementary to the active site of HMG-CoA reductase and also forms specific hydrogen bonds within the active site.
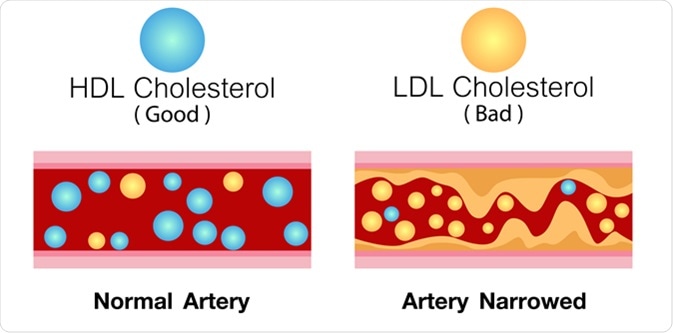
HDL cholesterol and LDL cholesterol in artery. Image Credit: Ching Design / Shutterstock
Pharmacokinetics and Pharmacodynamics of Atorvastatin
All drugs belonging to the drug class statins share a common mechanism of action; however, they differ in terms of their structures, pharmacokinetic profiles and efficacy. The drug’s chemical structure decides its water solubility, which ultimately influences the pharmacokinetic properties - absorption, distribution, metabolism and excretion.
Pharmacokinetics
Absorption: Atorvastatin is rapidly absorbed after oral administration. Maximum plasma concentrations occur within 1 to 2 hours and the extent of absorption increases with the increase in dose. Plasma concentrations of atorvastatin also depend on the population being treated. Higher concentrations of the drug are found in older adults (age 65 years or older), women and patients with chronic alcoholic liver disease.
Atorvastatin undergoes metabolism in the gastrointestinal mucosa and the liver before it reaches the systemic circulation. This phenomenon is termed as first-pass metabolism.
Food decreases the rate of drug absorption; however, it doesn’t interfere much with the extent of drug absorption. Changes in rate of atorvastatin absorption are not expected to have a clinically significant effect; hence atorvastatin can be taken with or without food.
Distribution: Atorvastatin has a volume of distribution of approximately 381 litres and the drug is =98% bound to plasma proteins.
Metabolism: Atorvastatin acid is extensively metabolized into its active derivatives in the gut and liver by chemical reactions such as oxidation, lactonisation and glucuronidation. These active derivatives account for almost 70% of HMG-CoA reductase inhibitory activity.
Excretion: Atorvastatin and its metabolites are eliminated by biliary secretion. Less than 2% of the drug is passed in urine. The elimination half-life of a drug is defined as the time required for the concentration of the drug in the body to be reduced to half. The elimination half-life of atorvastatin is approximately 14 hours, but the half-life of inhibitory activity for HMG-CoA reductase is 20 to 30 hours due to the contribution from the active metabolites of atorvastatin.
Pharmacodynamics
Atorvastatin and its active metabolites are all pharmacologically active. The liver is the main target organ of action, being the principal site of cholesterol synthesis and LDL clearance. The extent of LDL reduction depends on the dose of the drug rather than on the plasma drug concentration. Hence the individual dosage regimen should be based on the therapeutic response.
Drugs that Interact with Atorvastatin
The following are some important drug interactions of atorvastatin:
Cytochrome P450 3A4 (CYP 3A4) Inhibitors
Atorvastatin is metabolized by cytochrome P450 3A4; therefore, the concomitant use of drugs that inhibit CYP 3A4 can result in increased concentrations of atorvastatin in the blood. Caution should be exercised when co-administering atorvastatin with clarithromycin, protease inhibitors, and itraconazole, which are drugs that possess strong CYP 3A4 inhibitory action.
Cytochrome P450 3A4 Inducers
Efavirenz and rifampin are drugs with cytochrome P450 3A4-inducing activity; thus concomitant administration of atorvastatin with these drugs can lead to increased metabolism of atorvastatin and reductions in the plasma concentrations of atorvastatin.
Organic Anion Transporting Polypeptide 1B1 (OATP1B1) Inhibitors
OATP1B1 is an uptake transporter, responsible for the hepatic uptake of drugs from the blood.
Atorvastatin and its metabolites are substrates of the OATP1B1 transporter. Drugs such as cyclosporine are inhibitors of the OATP1B1 and cause inhibition of hepatic uptake of atorvastatin. This ultimately leads to increased bioavailability of atorvastatin which results in higher levels of the drug in the body. This leads in turn to an increased risk of muscle-related atorvastatin toxicity known as myopathies. Thus, the co-administration of atorvastatin with cyclosporine must be strictly avoided.
Gemfibrozil
Gemfibrozil is a lipid-modifying drug that belongs to the class of fibrates. Co-administration of atorvastatin with gemfibrozil leads to decreased metabolism of atorvastatin. This leads to an increased concentration of atorvastatin and a higher risk of myopathies.
Niacin
The use of atorvastatin with high doses of niacin increases the risk of rhabdomyolysis – a serious condition characterized by the breakdown of skeletal muscle tissue.
Digoxin
Co-administration of atorvastatin and digoxin leads to increased concentration of digoxin in the blood. Though the exact mechanism responsible for this effect is not known, a study by Boyd RA et al., found that atorvastatin inhibits the secretion of digoxin into the intestinal lumen. Patients taking digoxin should be monitored regularly when atorvastatin therapy is initiated.
Colchicine
The co-administration of atorvastatin with colchicine can lead to the increased bioavailability of atorvastatin, predisposing to the occurrence of myotoxicity such as rhabdomyolysis. Atorvastatin is a P-glycoprotein substrate, and colchicine is a P-glycoprotein inhibitor; combining the two modifies the pharmacokinetics of the drug by increasing the bioavailability and organ uptake of atorvastatin.
Oral Contraceptives
Atorvastatin may increase the blood concentrations of oral contraceptives such as norethindrone and ethinyl estradiol.
Grapefruit Juice and Atorvastatin
Consuming grapefruit or grapefruit juice can increase the plasma concentrations of atorvastatin, which, in turn, increases the potential for adverse reactions. This happens because grapefruit contains furanocoumarins that inhibit CYP 3A4 – the enzyme responsible for the metabolism of atorvastatin. The effect is prominent when the juice is consumed in excess of 1.2 liters per day.
Common Side Effects of Atorvastatin
Myopathies
Myopathies have been reported frequently in patients taking atorvastatin. Myopathies are a group of conditions affecting the skeletal muscle, and are characterized by muscle pain, muscle tenderness or muscular weakness. Elevated creatine phosphokinase levels are a prominent sign of myopathies.
Rhabdomyolysis, a life-threatening syndrome, may occur in severe cases of myopathy. Rhabdomyolysis is a complex condition caused by the breakdown of skeletal muscle fibers and the leakage of these proteins into the circulation, which may disrupt normal renal function, among other effects.
One common factor predisposing to the development of myopathy and rhabdomyolysis is the combination of atorvastatin with medications that increase the plasma concentration of atorvastatin. Advanced age (> 65), uncontrolled hypothyroidism, and renal disease are some other risk factors for the development of myopathies.
Strategies helpful for the management of myopathies include dose reduction, temporary cessation of the therapy or switching to another statin.
Liver Enzyme Abnormalities
Atorvastatin can also cause alterations in liver enzymes. Liver enzyme tests should be performed before initiating therapy; the enzyme levels should also be monitored regularly during the course of therapy. If serum transaminases are found to be more than three times the upper limit of normal, the dose of atorvastatin should be reduced or the drug discontinued. Tiredness, loss of appetite, dark urine, yellow discoloration of skin and abdominal pain are some common symptoms of liver derangement
Joint pain, pain in the extremities, digestive problems, cold, insomnia and urinary tract infections are some other frequent adverse effects observed in patients taking atorvastatin.
Due to its propensity to cause serious adverse effects, atorvastatin is contraindicated in individuals with active liver disease, hypersensitivity to atorvastatin or any of its components, pregnant women and lactating mothers.
Further Reading
Last Updated: Sep 19, 2019