Paclitaxel is a hydrophobic mitotic inhibitor with a powerful anti-cancer effect. It belongs to the group of taxanes (together with docetaxel), which are potent cytotoxic diterpenes derived from yew trees. Paclitaxel is active against a spectrum of malignancies that are generally considered to be refractory to conventional chemotherapy.
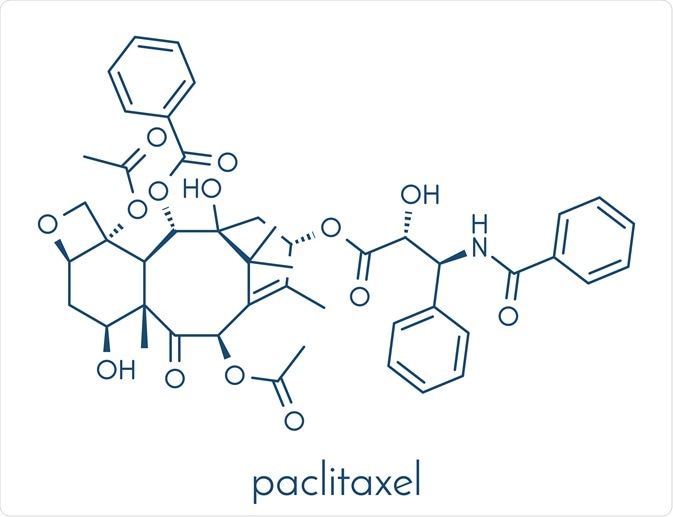 Image Credit: StudioMolekuul/Shutterstock.com
Image Credit: StudioMolekuul/Shutterstock.com
The drug was initially discovered as part of a National Cancer Institute program in 1962, where thousands of plant extracts were screened in order to find natural products that might treat cancer. A crude extract from the bark of the Pacific yew tree Taxus brevifolia, slow-growing and rare evergreen found in the forests of the Pacific Northwest, has been shown effective against many tumors in preclinical trials.
In recent years its activity was studied extensively by a vast number of clinical trials and the drug has become an irreplaceable part of various chemotherapeutic regimens. Paclitaxel inhibits cell division and chromosome segregation by targeting tubulin and stabilizing microtubules.
Structure and nomenclature
Paclitaxel is in a form of whitish crystalline powder which is highly lipophilic, thus very insoluble in water. It contains a heptadecane (17-carbon) skeleton with a molecular weight of 853.9 Da and a melting point around 216 ºC. The antitumor activity of the drug is mainly due to the side chain, A ring, oxetane ring and C2 benzoyl group. The drug was approved by the US Food and Drug Administration (FDA) and marketed in 1992.
Semisynthetic paclitaxel analog docetaxel derived from Taxus baccata species (a European yew) contains a hydroxyl functional group on carbon 10 (where paclitaxel has an acetate ester) and a tert-butyl carbamate ester on the phenylpropinate side chain (instead of benzyl amide). Both of those properties make docetaxel more soluble in water.
In 2005, the FDA approved a nanoparticle paclitaxel (also known as nab-paclitaxel). This form of the drug showed better effectiveness and lesser toxicity when compared to original paclitaxel; one of the reasons for the latter is that a highly toxic solvent does not have to be used anymore.
Range of activity
Paclitaxel is an effective anticancer agent against ovarian, lung, breast, prostate, liver, gastric and pancreatic cancer. This drug has also shown beneficial effects on melanoma, head and neck squamous cell carcinoma, anaplastic thyroid carcinoma and some other types of malignant tumors.
Although the list of cancers targeted by paclitaxel is still expanding, its main role to date is in treating metastatic carcinoma of the ovary, non-small cell lung carcinoma, metastatic breast cancer and as a second-line agent in AIDS-related Kaposi's sarcoma.
Production of IL-12 in tumor bearing hosts is also one of the effects of paclitaxel, which can in turn down-regulate the tumor growth significantly. The drug also upregulates functional IL-6 expression in ovarian cancer cells by employing multiple signaling pathways. In addition, glycolysis can be reduced via specific mechanisms.
This drug can also act as an anti-proliferative agent. Stent-based elution of paclitaxel considerably reduces neointimal hyperplasia, angiographic restenosis and the need for repeated revascularization in patients undergoing percutaneous coronary interventions.
Paclitaxel is currently being studies for other diseases that require stabilization of microtubules and the avoidance of angiogenesis and cell proliferation. One example is psoriasis, one of the most baffling and persistent skin disorders. As there is an ongoing research of paclitaxel as a potential treatment for Alzheimer's or Parkinson's disease, the magnitude of the drugs' usefulness could be even bigger.
References
Further Reading
Last Updated: May 24, 2021