Systemic treatments for psoriasis in oral or injectable form are typically recommended for patients with insufficient response to topical treatments and phototherapy.
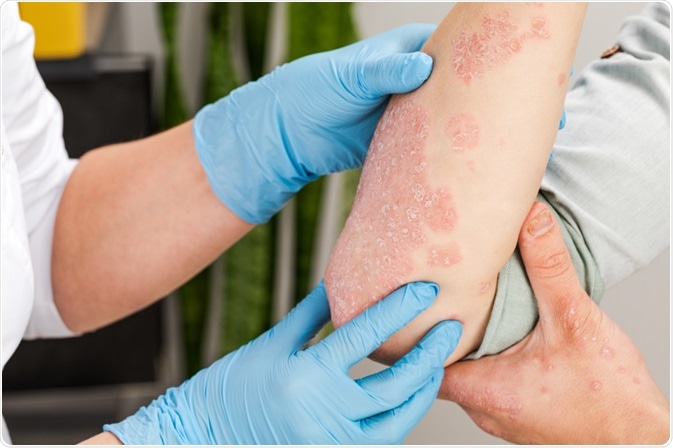
Image Credit: Fuss Sergey / Shutterstock
Cyclosporine
Cyclosporine is an immunosuppressant medication that inhibits the activity of immune cells and slows the growth of skin cells responsible for symptoms of psoriasis. It typically provides some relief within a few weeks and reaches a stable level of control within three to four months.
Administration with cyclosporine may increase the risk of renal dysfunction, skin cancer and other health conditions. The adverse effects associated with systemic cyclosporine for patients with psoriasis are not well known. For this reason, the current guidelines for the duration of treatment are not clear.
Methotrexate
Methotrexate is an antimetabolite medication that may be prescribed for adults with severe psoriasis or psoriatic arthritis. It helps to reduce symptoms of psoriasis due to its action to suppress the immune system and inhibit the inflammation associated with the condition. Methotrexate can be taken orally or by injection once a week.
An improvement can usually be noted within three to six weeks of treatment initiation, but it may take up to six months for the full benefits of the medication to become evident.
There is a significant risk of liver damage for patients taking methotrexate and approximately 1 in 200 patients experience reversible cirrhosis. Other side effects may include nausea, fatigue, headache and sensitivity to sunlight.
Soriatane (acitretin)
Soriatane is a retinoid or vitamin A derivative medication available in an oral formulation to be taken daily for patients with psoriasis. The exact mechanism of action is not known, but it is thought to be beneficial in the treatment of psoriasis because it helps to control skin cell multiplication.
Many patients find that their symptoms of psoriasis initially worsen with Soriatane treatment, but the skin lesions have some improvement after two to four months. It is less effective as a stand-alone therapy and is usually recommended in combination with other treatments for severe cases of psoriasis.
Alternatively, isotretinoin is another oral retinoid medication that is sometimes used for psoriasis treatment.
Other Oral Systemic Medications
There are various other oral treatments that are sometimes used in the treatment of psoriasis, including:
- Antimalarial medications
- Hydroxyurea
- Isotretinoin
- Mycophenolatemofetil (Cellcept)
- Nonsteroidal anti-inflammatory drugs (NSAIDs)
- Sulfasalazine (Azulfidine)
- 6-Thioguanine
- Otezla (apremilast)
Biologic Response Modifiers
Biologic medications are a newer class of drugs that are administered by intravenous injection or infusion. Their action is targeted towards specific proteins that affect the immune system and inflammatory response, thereby reducing the risk of side effects in comparison to traditional treatment options.
Tumor necrosis factor-alpha (TNF-alpha) blockers work by reducing the TNF-alpha in the body, reducing the inflammation associated with psoriasis. Drugs in this class include:
- Cimzia (certolizumab pegol)
- Enbrel (etanercept)
- Humira (adalimumab)
- Remicade (infliximab)
- Simponi (golimumab)
Stelara (ustekinumab) is another type of drug that selectively targets the interleukin-12 and interleukin-23 proteins, which reduces inflammation and improves symptoms of psoriasis.
Systemic Drug Warnings
Due to the high potentiality of adverse effects when taking systemic treatment for psoriasis, it is essential to monitor the concentration and any toxicity with regular blood and liver function examinations. The lowest dose possible to achieve the desired effect and control of symptoms should be used.
The majority of systemic treatments used for psoriasis may cause harm if taken by women who are pregnant or planning to become pregnant and should, therefore, be avoided.
A rebound effect can be expected if patients stop systemic treatment for psoriasis with a flare-up of symptoms. For this reason, the medications should slowly be withdrawn to reduce the rebound effect.
References
Further Reading
Last Updated: Feb 25, 2023