Filariasis refers to a group of diseases caused by parasitic worms, which are transmitted by blood feeding insects.
Two species infect the lymphatic and blood systems causing lymphatic filariasis, which in some people can lead to gross swelling of the limbs and 'elephantiasis'.
The other major filarial disease is onchocerciasis or 'river blindness'. This parasite lives in the skin causing intense and troublesome itching and dermatitis and parasite migration into the eyes can progress to blindness.
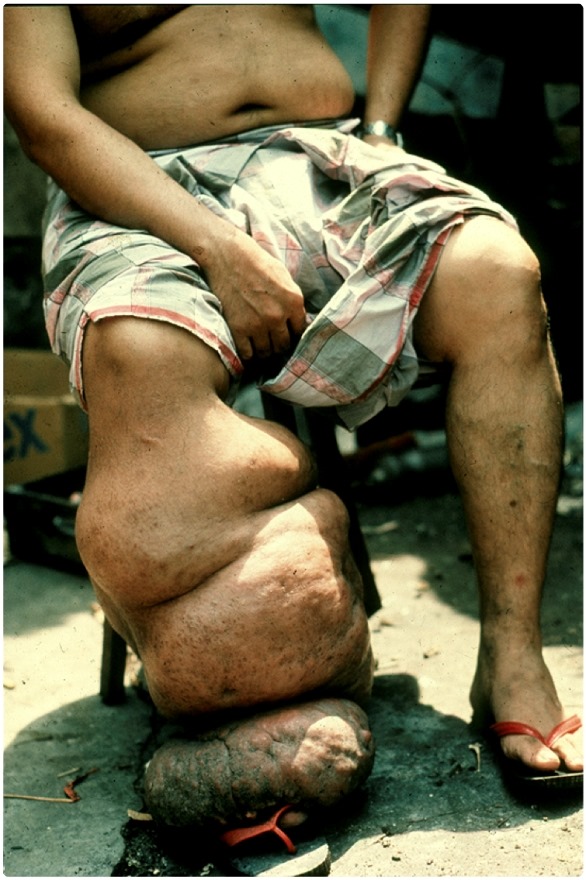
How many people are affected by these parasitic worm infections and in what countries are they based?
Lymphatic filariasis is estimated to cause 120 million cases in 83 countries, with India and Sub-Saharan Africa carrying the majority of the global burden.
Onchocerciasis affects 37 million people in 34 countries mostly in Africa, with smaller foci in southern and central America.
What research has currently been done on human filariasis and why is there a need for further research?
The key discovery that lead to this work was finding that the parasites host a bacterial symbiont called Wolbachia, which lives inside of the worm's cells and is essential for the growth, fertility and survival of the worms.
This lead us to test whether simple antibiotics might be effective as drugs to clear the bacteria from the worms, leaving them without their essential symbiont.
Much to our surprise and delight this approach worked very well and was even better than current drugs used to treat these diseases.
A course of the antibiotic doxycycline could effectively cure infections of both lymphatic filariasis and onchocerciasis and was even able to reverse clinical disease.
The need for further research is that the current treatment requires a 4-6 week course of doxycycline, which cannot be given to children or pregnant women.
Although this can be used to treat individual patients, in a ‘test & treat’ approach, it is not suitable for widespread community based treatments.
We need to find drugs, which work in the same way to clear the symbiont, but can do so in a treatment course of 7 days or less and be safe for children and in pregnancy.
This would then provide a safe curative treatment that could be deployed through existing mass drug administration strategies to dramatically shorten elimination programme timeframes.
Liverpool School of Tropical Medicine has recently received US$ 10 million from the Bill & Melinda Gates Foundation to continue their research on human filariasis. How do you plan to use this grant?
The grant is a continuation of our A·WOL (Anti-Wolbachia) programme launched in 2007.
The new grants will fund two programmes running in parallel: ‘A·WOL II Macrofilaricide Drug Discovery’ will progress six new chemical series, selected for their potency and drug-like features for ‘hit-to-lead’ optimization to select the best clinical candidate; ‘A·WOL II Macrofilaricide Drug Development’ will take the best registered and re-purposed drugs from our screening campaign to optimise the best combination of drugs, which can then be tested in field trials with our partners in Ghana and Cameroon.
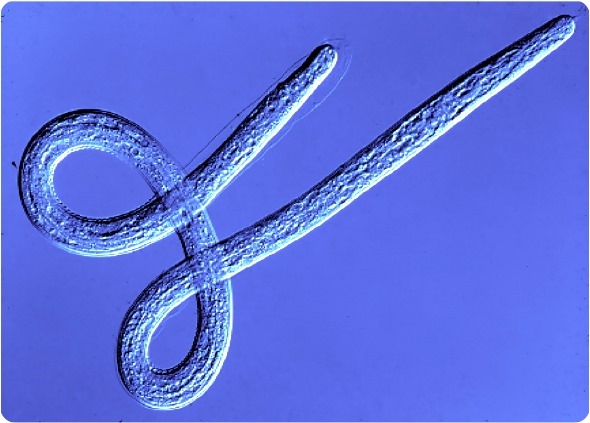
What methods will be used to determine the best clinical candidate from your portfolio of hundreds of drugs?
For ‘A·WOL II Macrofilaricide Drug Discovery’ we have developed a rational medicinal chemistry program around each of the six hit series. From the top six hits we have selected three templates for hit to lead optimization with an additional three back-up templates.
We are currently running a head to head evaluation of the three series with the intention of identifying the most promising template for final lead optimization.
For ‘A·WOL II Macrofilaricide Drug Development’ we will test the pharmacokinetics and dynamics of different drugs and use computer models to predict the best combinations and dosages.
How do you plan to progress these drugs and how long do you think this process will take?
When we have completed the head-to-head comparison of the best pre-clinical candidates, we will then progress these candidates though pre-clinical safety testing for new drugs and for registered drug combinations we will test the best combination in clinical trials, together with our A·WOL partners in the University of Bonn and Ghana.
What impact will your research have?
The A·WOL approach has already made an impact. The ‘Onchocerciasis Elimination Programme for the Americas’ (OEPA) has adopted doxycycline to foreshorten the time to elimination in a focus in Venezuela to achieve the Regional Elimination goal.
Also the use of doxycycline as new strategy for morbidity management of lymphatic filariasis has recently been endorsed by WHO.
Our hope is that further discovery and development of A·WOL drugs will make this approach feasible at a larger scale.
The WHO Road Map has very ambitious targets based on existing available preventive chemotherapy tools, yet, it is recognized that if there is to be a more rapid drive towards elimination and strategies to tackle the barriers of reduced efficacy of existing drugs and co-infection related adverse events, A∙WOL outcomes should be considered for deployment at the earliest opportunity.
How did LSTM manage to secure the latest grant from the Bill & Melinda Gates Foundation?
The Bill & Melinda Gates Foundation are committed to the development of a macrofilaricidal drug for onchocerciasis and LF.
The first A·WOL programme was successful at identifying a range of alternative drugs, with improved activity and better safety profiles than our existing drugs.
We also have a strong ‘proof-of-concept’ from an extensive series of clinical trials that this approach is effective and has an excellent safety profile.
We were also able to show that we can work effectively and efficiently as a consortium to deliver outcomes in a timely manner.
Together these features helped us to convince the Foundation that we have a good chance of success in achieving their goals.
Where can readers find more information?
Our website (http://www.a-wol.net/) contains further information and a number of scientific reviews in open access journals are listed on our website for more in depth information.
About Professor Mark Taylor
 Professor Taylor is Professor of Parasitology and Head of the Department of Parasitology at the Liverpool School of Tropical Medicine.
Professor Taylor is Professor of Parasitology and Head of the Department of Parasitology at the Liverpool School of Tropical Medicine.
He is Director of A·WOL, an international consortium of academic and industrial partners funded by the Bill & Melinda Gates Foundation to discover and develop new drugs against onchocerciasis and lymphatic filariasis.
His area of research is the filarial nematode diseases of humans. These include elephantiasis (lymphatic filariasis) and river blindness (onchocerciasis): two of the leading causes of global disability.
His research focuses on the role of Wolbachia-bacterial endosymbionts, which have evolved a mutualistic symbiosis with filarial nematodes that is essential for parasite development, fertility and survival.
Whilst the bacteria are beneficial for the nematode, their release into the host results in inflammatory immune activation that leads to adverse events following anti-filarial treatment and contributes to the pathogenesis of river blindness and elephantiasis.
His research team performs laboratory and field-based studies to exploit Wolbachia as a target for antibiotic therapy, providing a novel and effective macrofilaricidal treatment for filarial infection and disease.