The SPEAR UltraDetect™ pTau 217 biomarker assay kit is a specialized tool for researching Alzheimer’s disease (AD) using plasma samples. The assay requires the SPEAR UltraDetect™ Reaction Kit and is intended for research purposes only.
A breakthrough blood biomarker for AD pathology
pTau217, also called phosphorylated tau at threonine 217, is a specific blood-based biomarker for brain amyloid β pathology. When validated against CSF Aβ42/40 and PET imaging, the biomarker showed high specificity and sensitivity for identifying amyloid positivity compared with other plasma markers, such as Aβ42/40 or pTau181.
These properties make pTau 217 an ideal tool for non-invasive screening, early detection, and disease monitoring in clinical trials, diagnostic research, and longitudinal Alzheimer’s disease studies.
Unparalleled specificity with SPEAR UltraDetect pTau 217
The SPEAR UltraDetect pTau 217 assay has demonstrated high specificity in distinguishing between non-AD and AD patient samples. Its precision (quantifying 100 % of healthy samples above the fLLOQ) and sensitivity provide researchers with an improved tool for assessing blood-based samples in Alzheimer’s research.
A study comparing PET- and CSF-confirmed AD and non-AD samples showed that SPEAR UltraDetect pTau 217 exhibited a significant 5.41× mean fold-difference between groups and area under the curve of 0.984 for PET-confirmed samples and 0.989 for CSF-confirmed samples.
These results show that the SPEAR UltraDetect pTau 217 assay can differentiate between non-AD and AD patient plasma samples.
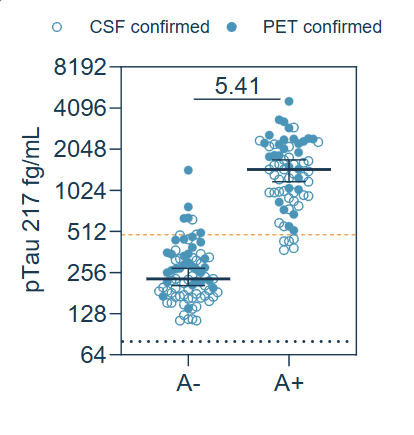
SPEAR UltraDetect pTau 217 fold-difference in AD and Control samples [1 µL diluted sample]. SPEAR UltraDetect pTau 217 testing on CSF-confirmed EDTA Plasma AD and non-AD (Control), including 48 non-AD and 40 AD. Amyloid PET confirmed EDTA plasma, including 33 Amyloid negative (Control) and 28 Amyloid positive (AD). Image Credit: Spear Bio
![SPEAR UltraDetect pTau 217 ROC for CSF-confirmed and PET-confirmed plasma samples [1 µL diluted sample]. SPEAR UltraDetect pTau217 ROC analyses on CSF-confirmed EDTA Plasma AD and non-AD (Control) including 48 non-AD and 40 AD. Amyloid PET confirmed EDTA plasma including 33 Amyloid negative (Control) and 28 Amyloid positive (AD)](https://www.news-medical.net/images/equipments/ImageForEquipment_7608_17706400771853239.png)
SPEAR UltraDetect pTau 217 ROC for CSF-confirmed and PET-confirmed plasma samples [1 µL diluted sample]. SPEAR UltraDetect pTau217 ROC analyses on CSF-confirmed EDTA Plasma AD and non-AD (Control), including 48 non-AD and 40 AD. Amyloid PET confirmed EDTA plasm, including 33 Amyloid negative (Control) and 28 Amyloid positive (AD). Image Credit: Spear Bio
Download the Datasheet for More Information
Assay specifications
Source: Spear Bio
| |
|
| MRD |
4X |
| Diluted Sample Volume |
1 µL |
| Raw Sample Volume to Machine |
10 µL |
| LOD (pg/mL) |
0.005 |
| fLLOQ (pg/mL) |
0.080 |
| Spike-recovery (% recovery) |
106 % |
| Linearity (% recovery) |
87 % |
| Intra-plate CV (AVG %) |
3.32 % |
| Inter-plate CV (AVG %) |
4.43 % |
| EDTA Plasma Quantifiability |
100 % |
FDA breakthrough device designation
In January 2025, the FDA awarded Breakthrough Device Designation to the SPEAR UltraDetect pTau 217 blood test, recognizing its value in advancing the diagnosis of Alzheimer’s disease. As an innovative blood test for pTau 217, the assay is considered a more accessible and less invasive alternative to conventional assay methods such as PET scans, Mass Spectrometry, and CSF analysis.
The test has been developed to detect pTau 217, which is a key biomarker for Alzheimer’s disease, and offers the high specificity and sensitivity required to measure even low concentrations in blood.
It supports more reliable, earlier disease diagnosis and thus has the potential to differentiate Alzheimer’s from other neurological diseases. This also enables patients to get access to early interventions and emerging treatments.
Scalable AD insights
Supporting Alzheimer’s Disease research at any scale
The SPEAR UltraDetect™ pTau217 blood test provides reliable and consistent data that correlates with established amyloid PET imaging and CSF biomarkers. This makes it suitable for characterizing biomarker changes in patients with mild cognitive impairment or monitoring treatment response in longitudinal studies. By minimizing invasiveness and complexity, the test is suitable for:
- Clinical trial endpoints and patient selection
- Multi-marker diagnostics (e.g., with neurofilament light or pTau231)
- Exploratory pipeline integration
- Preclinical therapeutic screening
Across samples, across studies
Recommended for research across sample types and research settings
The SPEAR UltraDetect™ pTau217 assay is developed for research purposes only and is optimized for use with plasma samples. Its high specificity makes it suitable for various research applications, including drug development studies and those involving memory clinic cohorts. It is an ideal alternative to ELISA, Mass Spectrometry, or PET-based methods in Alzheimer’s disease research, owing to its high analytical performance and its support in assessing Aβ positivity, biomarker changes, and cognitive decline.
Performance without tradeoffs
Designed for sensitivity and specificity without compromise
With its clean design, the SPEAR assay is ideal for scientists who require high performance with minimal complexity in the operational workflow.
Its homogenous format and two-factor authentication system verify each pTau217 molecule in a sample twice. This reduces false positives and ensures highly specific measurement of pTau217 levels in plasma, even in complex blood matrices.
The assay works with 1 µL of diluted samples, enabling repeated analysis of precious, scarce specimens, such as those collected in longitudinal neurodegeneration studies.
From bench to breakthrough
From evaluation to disease-modifying therapy development
SPEAR UltraDetect™ assay kit is suitable for researching biomarkers in Alzheimer’s disease, validating potential therapies, or studying the tau protein kinetics due to its specificity, sensitivity, and precision. Its user-friendly design makes it ideal for research labs of any size, accelerating discoveries from lab to clinical applications.