This article is based on a poster originally authored by Karissa Cottier, Christian Smith, Devika Bhalerao, Nia Dawson, Logan Clemow, Seema Muranjan and Scott Heyward.
Liver Microphysiological Systems (MPS) with primary human hepatocytes (PHH) have been designed to better imitate the liver's physiological environment. These models are a promising tool for drug development; nevertheless, their use is often hampered by issues such as cost, throughput, and operational complexity.
Hepatic spheroids are particularly appealing among existing 3D liver models for ADME-tox applications due to their simplicity, scalability, and low cost.
The researchers used three lots of primary human hepatocytes to measure drug-induced liver injury (DILI) from known hepatotoxic medications or clearance of low-turnover chemicals in hepatic spheroids plated in conventional or microcavity (Elplasia) ultra-low binding plates.
Standard spheroid plates were constructed by plating 1500 cells/well in a Nunc ultra-low binding plate, while microcavity plates were created by plating 250 cells/microcavity (79/well) in a Corning Elplasia plate.
Spheroids formed on Day 5 of culture and maintained their morphology throughout the experiment. Spheroids were treated with acetaminophen (40-2.5 mM), valproic acid (50-3.13 mM), or diclofenac (1.2 mM-75 µM) for 72 hours to assess DILI. Cytotoxicity was measured using ATP content.
Both formats showed concentration-dependent hepatotoxicity. Standard plates had TC50 values of 3.46 mM for acetaminophen, 5.04 mM for valproic acid, and 303.5 µM for diclofenac. However, Elplasia plates had higher TC50 values for valproic acid (4.58 mM for acetaminophen, 18.19 mM for valproic acid, and 385.8 µM for diclofenac).
Separately, the ability of conventional and microcavity spheroids to metabolize low-turnover drugs risperidone and disopyramide was tested throughout a 7-day incubation period. Samples were collected daily, and parent depletion was determined by LC/MS/MS analysis.
Spheroids plated in standard plates did not exhibit significant parent depletion during the 7-day incubation period. Plating spheroids in microcavity Elplasia plates resulted in a ∼70 % depletion of risperidone and a 30 % depletion of disopyramide after a 7-day incubation, with donor-dependent clearance.
Taken together, these findings demonstrate that each spheroid configuration offers distinct challenges and benefits. While normal plates provide a cost-effective and physiologically appropriate DILI testing with fewer hepatocytes, microcavity Elplasia plates have higher sensitivity for detecting low-turnover chemicals.
These results highlight the significance of model selection based on specific experimental objectives and advocate the strategic use of hepatic spheroid platforms into drug discovery and safety pipelines.
Methods
Standard spheroid plating
Primary hepatocytes were thawed in spheroidal spin media and centrifuged at 100 x g for 10 minutes. To eliminate dead cells and debris, the supernatant was aspirated. The cell pellet was then resuspended in 5 mL of spheroid plating media. Cells were counted and diluted to 15,000 cells per mL.
To prevent edge effects during plating, 100 µL of spheroid plating medium was applied to each inner well and 200 µL of sterile water to the perimeter wells. To plate cells in a 96-well Nunclon™ Sphera™ Dish (ultra-low binding), 100 µL of cell suspension was added to wells holding 100 µL of spheroid plating mix (total volume: 200 µL).
Plates were centrifuged at 250 x g for 2 minutes before being placed in a 37 °C, 5 % CO2 incubator for 5 days to allow for spheroid formation.
Microcavity spheroid plating
Primary hepatocytes were thawed in spheroid spin media and treated as described above. After counting, cells were diluted to 197,500 cells/mL. To prevent edge effects during plating, 100 µL of spheroid plating medium was applied to each inner well, and 200 µL of sterile water was applied to the perimeter wells.
The plates were centrifuged at 250 x g for 2 minutes to evenly distribute the medium within each microcavity. To plate cells in 96-well Corning Microcavity Elplasia plates (79 microcavities per well, 250 cells per spheroid), 100 µL of cell suspension was added to wells with 100 µL of spheroid plating medium (200 µL total).
Plates were centrifuged at 250 x g for 2 minutes before being placed in a 37 °C, 5 % CO2 incubator for five days to allow for spheroid formation.
Drug treatment DILI assessment
Acetaminophen (80 mM), Valproic acid (100 mM), and Diclofenac (2.4 mM) were produced as 2X stocks in serum-free spheroid maintenance media and serially diluted (1:1 serial dilutions, 5 concentrations plus NT).
On day 5, 100 µL of media was withdrawn from each well in conventional and microcavity spheroid plates, leaving 100 µL containing cells. Spheroids were confirmed by removing the medium and adding 100 µL of each 2X substance.
Spheroids were treated with the chemicals for 72 hours. At the end of the incubation period, phase-contrast images were captured, and one representative image was selected for each compound and plate.
Cell viability assessment
The Cell Titer Glo® 2.0 (Promega®) was used to determine cell viability using ATP as a metric, following the manufacturer's protocol. The luminescence was measured with a Perkin Elmer Wallac 1420 Victor2 microplate reader. All luminescence measurements were normalized and expressed as a percentage of control.
Low-turnover compound treatment
Disopyramide and risperidone were produced as 2X stocks (0.6 µM) in spheroidal maintenance medium. On day 5, 100 µL of media was withdrawn from each well in conventional and microcavity spheroid plates, leaving 100 µL containing cells.
Spheroids were confirmed by removing the medium and adding 100 µL of each 2X substance. The time=0 samples were promptly terminated in an equal volume of cold methanol, and the wells were refilled with sterile water to prevent evaporation.
Clearance samples were taken at 24, 48, 72, and 144 hours, and parent compound depletion was measured by LC/MS/MS and standardized to time=0 to determine % remaining over time.
Results
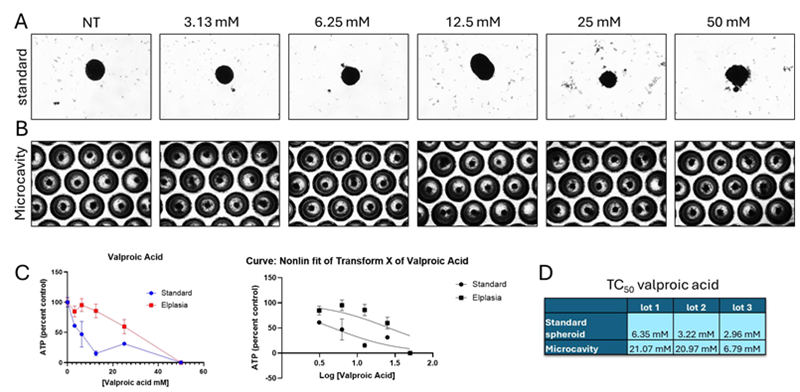
Figure 1. Valproic acid toxicity. Image Credit: BioIVT
Representative pictures following 72 hours of drug treatment on (A) standard and (B) microcavity spheroid plates. The presence of debris, an uneven form, and blurred borders all indicated visible toxicity.
(C) Toxicity was concentration-dependent in both plate types. Concentrations were log=converted, and nonlinear regression was used to calculate the TC50 in three distinct batches for each plate type.
(D) The three lots had equal TC50 values for both regular and microcavity Elplasia plates. Compared to lots 1 and 2, lot 3 had a much lower TC50 in Elplasia plates. The statistical analysis of the nonlinear regressions revealed a substantial difference between the curves for standard and Elplasia plates.
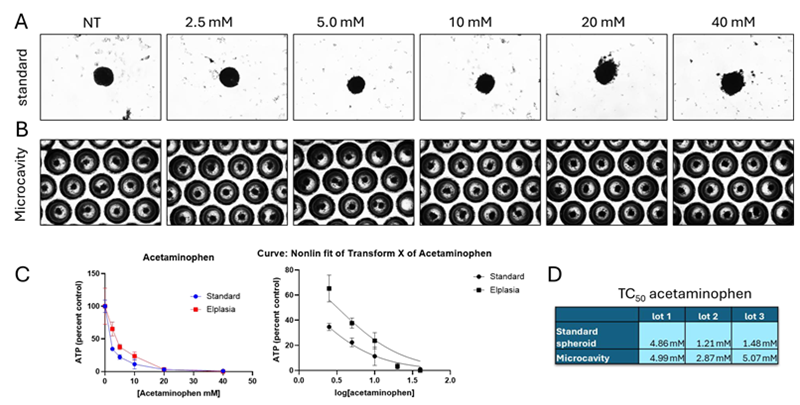
Figure 2. Acetaminophen toxicity. Image Credit: BioIVT
Representative pictures following 72 hours of drug treatment on (A) standard and (B) microcavity spheroid plates. The presence of debris, an uneven form, and blurred borders all indicated visible toxicity.
(C) Toxicity was concentration-dependent in both plate types. Concentrations were log converted, and nonlinear regression was used to calculate the TC50 in three distinct batches for each plate type.
(D) The three lots had equal TC50 values for both regular and microcavity Elplasia plates. In lot 3, the TC50 of the regular and Elplasia plates differed significantly.
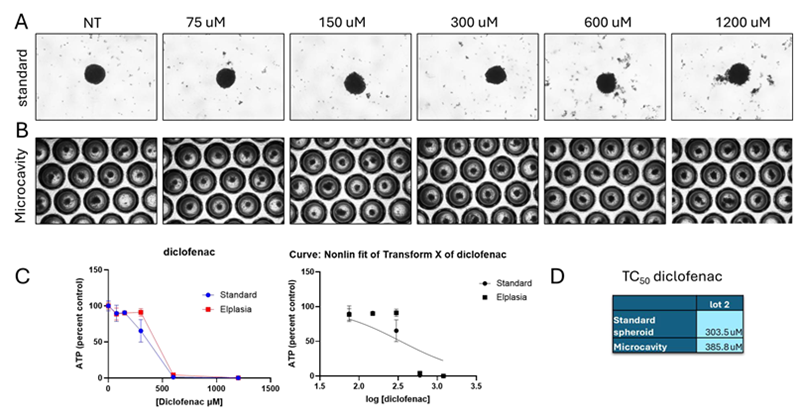
Figure 3. Diclofenac toxicity. Image Credit: BioIVT
Representative pictures following 72 hours of drug treatment on (A) standard and (B) microcavity spheroid plates. The presence of debris, an uneven form, and blurred borders all indicated visible toxicity.
(C) Toxicity was concentration-dependent in both plate types. Concentrations were log converted, and the TC50 for each plate type was calculated using nonlinear regression in a single lot. (D) Diclofenac was only tested in lot 2, and no variation in TC50 was seen between plate types.
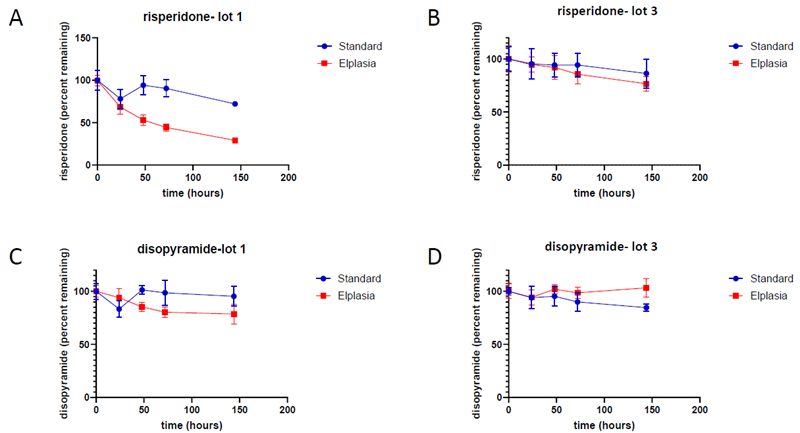
Figure 4. Low turnover compound clearance. Image Credit: BioIVT
Risperidone clearance over 144 hours (normalized to time=0). (A) In lot 1, risperidone was effectively digested in Elplasia plates (71 % depletion) but less so in conventional spheroid plates (28 % depletion).
(B) Risperidone was poorly digested in both plate types in lot 3 (23 % depletion in Elplasia and 13 % in standard). (C) Disopyramide was not removed efficiently in either plate type for lot 1 (Elplasia 21 % depletion, standard 4 % depletion) (D) or lot 2 (Elplasia no depletion, normal 17 % depletion).
Risperidone is mostly metabolized by CYP2D6 (∼77 %) and CYP3A4 (∼36 %) enzymes, while disopyramide is primarily metabolized by CYP3A4, emphasizing the significance of metabolic activity in lot selection for ADME research.
Conclusions
Human hepatic spheroids are a useful model for ADME-toxicity research.
- Standard spheroids provide a cost-effective and predictive approach for measuring drug toxicity in DILI investigations.
- When there are significant differences between parent and metabolite toxicity, using more robust models, such as microcavity Elplasia plates, can help prevent false toxicity profiling.
- Microcavity Elplasia plates allow spheroids to better metabolize low turnover chemicals compared to normal spheroid plates.
- Metabolic enzyme activity characterization data for key CYP450 enzymes should be considered while selecting lots for clearance experiments.
References
- Bell, C.C., et al. (2020). Functionality of primary hepatic non-parenchymal cells in a 3D spheroid model and contribution to acetaminophen hepatotoxicity. Archives of Toxicology, 94(4), pp.1251–1263. DOI: 10.1007/s00204-020-02682-w. https://link.springer.com/article/10.1007/s00204-020-02682-w.
- Cottier, K.E., et al. (2023). Micropatterned primary hepatocyte co-culture (HEPATOPAC) for fatty liver disease modeling and drug screening. Scientific Reports, 13(1). DOI: 10.1038/s41598-023-42785-9. https://www.nature.com/articles/s41598-023-42785-9.
- Knöspel, F., et al. (2016). In Vitro Model for Hepatotoxicity Studies Based on Primary Human Hepatocyte Cultivation in a Perfused 3D Bioreactor System. International Journal of Molecular Sciences, (online) 17(4), p.584. DOI: 10.3390/ijms17040584. https://www.mdpi.com/1422-0067/17/4/584?utm_source=researchgate.net&utm_medium=article.
About BioIVT
BioIVT, formerly BioreclamationIVT, is a leading global provider of high-quality biological specimens and value-added services. We specialize in control and disease state samples including human and animal tissues, cell products, blood and other biofluids. Our unmatched portfolio of clinical specimens directly supports precision medicine research and the effort to improve patient outcomes by coupling comprehensive clinical data with donor samples.
Our Research Services team works collaboratively with clients to provide in vitro hepatic modeling solutions. And as the world’s premier supplier of ADME-Tox model systems, including hepatocytes and subcellular fractions, BioIVT enables scientists to better understand the pharmacokinetics and drug metabolism of newly discovered compounds and the effects on disease processes. By combining our technical expertise, exceptional customer service, and unparalleled access to biological specimens, BioIVT serves the research community as a trusted partner in ELEVATING SCIENCE®.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.
Last Updated: Apr 24, 2026