This article is based on a poster originally authored by Diana Chiluiza and Courtney Noah.
Human peripheral blood mononuclear cells (PBMCs) are critical components of gene therapy and regenerative medicine research.1
PBMCs comprise a complex population of immune cells, including lymphocytes (CD4+ and CD8+ T cells), B cells, natural killer cells, monocytes that differentiate into macrophages, and dendritic cells, as well as a small number of stem cells.2,3
Leukopaks, derived from leukapheresis, are the preferred source of PBMCs. They are high in white blood cells and can reduce variability because they are derived from a single donor.
The conditions under which leukopaks and other biospecimens are stored and transported influence product quality, which is measured not only in terms of total cell counts and viability but also in immune cell population diversity.
Using fresh leukopaks as starting materials accelerates important research and development, particularly when the leukopak collecting and processing facilities are close together. However, this is not always achievable, and extended transit times may be necessary.4
Fresh leukopaks can be delivered locally in as little as 12 hours, but countrywide distribution takes 24 to 48 hours, and worldwide shipping takes much longer.
Previous research investigated the effects of temperature and duration on viability, morphology, phenotype retention, metabolism (pH, glucose, O2), mitochondrial DNA integrity, mitochondrial reactive oxygen species, membrane damage, and cytokine production in PBMCs and other cell lines.4,5
PBMC activation by lipopolysaccharide and heat-killed Staphylococcus aureus was mildly influenced by 24 hours of ambient shipping and totally eliminated after 48 hours.4
Shipping fresh rather than frozen items reduces cellular damage caused by osmotic pressure fluctuations during cryopreservation. Delays in product delivery might cause temperature variations in the leukopak, affecting viability, metabolism, and microtubule integrity.
Controlled shipping conditions that limit temperature fluctuations in leukopak shipments will help keep white blood cells undifferentiated, which is critical for the development of new therapies and the efficacy of existing treatments.
Proper storage and shipping conditions for cell and gene therapy products are critical to the design, repeatability, and reproducibility of research, development, product design, and treatment outcomes.
For this reason, the researchers investigated the circumstances of white blood cells in leukopak products. They measured cell concentrations, viability, and differential leukocyte counts (DLC) throughout five days of storage at ambient temperature and 4 °C to simulate shipping circumstances.
Methods
Three healthy donors were randomly chosen for leukapheresis. On the day of collection (0 hours), viability, CBC, and DLC were measured. From the leftover leukopak volume, 20 1.5 mL aliquots were deposited in 2 mL vials.
Ten vials were packed in room temperature shipping boxes (INTELSIUS PHT(15-25) 3.5 L 60-hr temperature boxes) and cold shipping boxes (NanoCool™ Cooling System 2-8 °C 1.05L shipping box).
Cold boxes were activated and quickly closed. ITAG Temperature Loggers (www.itag4.com) were placed inside each box, next to the leukopak samples, and programmed to read every one minute.
Each day, two vials were immediately taken from each box. One vial was utilized for CBC analysis, and another for PBMC purification and viability calculations using the standard Trypan Blue Exclusion assay.
Results
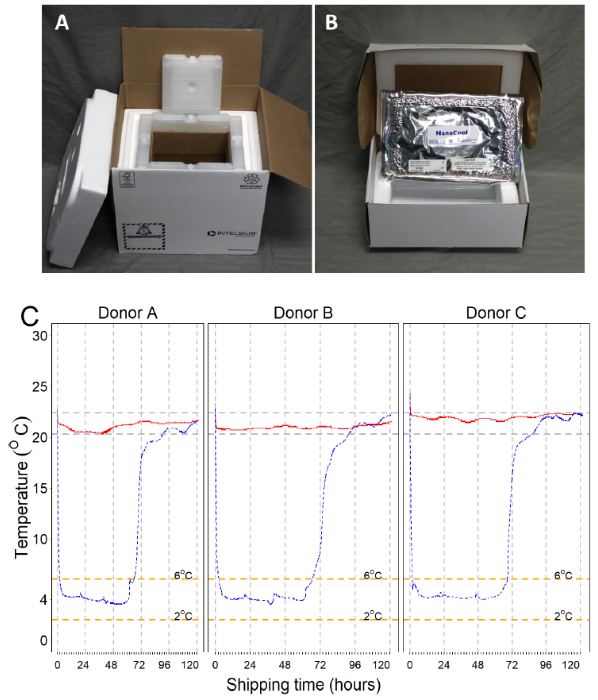
Figure 1. Ambient and cold boxes were used in the study, and temperature curves of the storage containers during the testing period. Image Credit: BioIVT
A. INTELSIUS PHT(15-25) 3.5 L 60-hour temperature box. B. NanoCool™ Cooling System 2 (8 °C, 1.05 L) delivery box. C. Temperature curves for room temperature and cold shipment boxes were recorded every minute for five days using ITAG temperature data loggers.
Ambient boxes maintained stable temperatures ranging from 20.3 °C to 22.4 °C throughout the trial (red lines), with a mean of 21.3 °C (SD 0.52).
After activating the NanoCool™ contents, the temperature within the cold boxes (blue lines) decreased from room temperature to below 6 °C in 2 hr and was maintained below 6 °C for around 2.7 days (mean 67 hr), returning to room temperature on day 3.
The average temperature for the crucial shipping period (below 6 °C and above 2 °C) was 4.2 °C (SD 0.5).
Up to 24 hours, viability is very constant and similar across the two temperatures (no significant variation in averages). At 24 hours, viability begins to decline rapidly in the room temperature groups, reaching an average of 75.4 % (SD 11.6) at 48 hours, whereas cold conditions maintained a viability of 97.3 % (SD 2.3).
The difference between the two circumstances is significant (p = 0.018). By 72 hours, viability in cold circumstances remained consistent (mean 96.6 %, SD 1.4), whereas viability in ambient conditions dropped considerably lower (mean 60.5 %, SD 13.2).
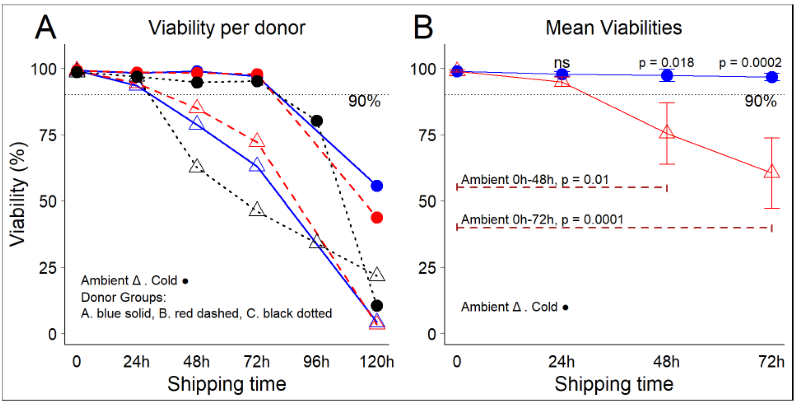
Figure 2. Temperature conditions influence the viability of freshly purified PBMCs during leukopak shipping. Image Credit: BioIVT
(A) PBMC viability by donor and shipment time. (B) Mean viabilities by temperature and shipping time. Error bars indicate one standard deviation from the mean. Using strong cellular starting materials in research and development is crucial for the success of innovative therapeutics.
The first quality control (QC) metric is the proportion of viable cells in a product volume, and industry standards recommend viabilities of ≥ 90 %. ANOVA was used for statistical analysis, with Tukey's Honest Significant Difference Test applied post hoc.
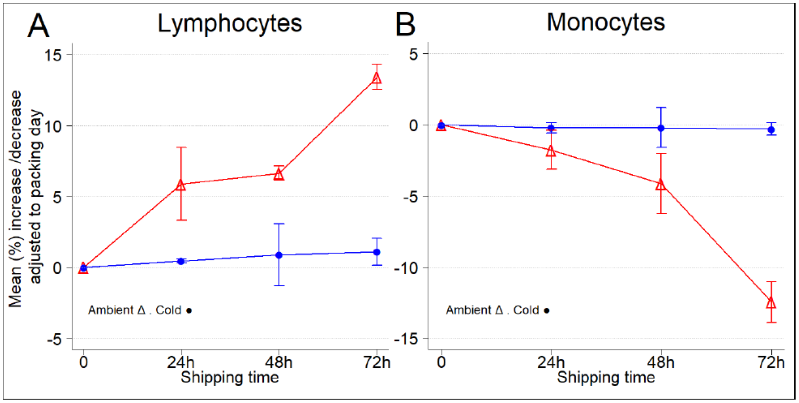
Figure 3. Temperature conditions (ambient versus 4 °C) influence the lymphocyte and monocyte composition of leukopaks during shipping. Image Credit: BioIVT
(A) Average increase/decrease in lymphocytes per shipping day, adjusted for collection day. (B) Average increase/decrease in monocytes per shipping day, adjusted for collection day. Error bars indicate one standard deviation from the mean.
Lymphocytes are the immune system's most essential cell types, which include CD4+ and CD8+ T cells, NK cells, and antigen-presenting CD19+ B cells.
Another essential component of the immune system involved in pathogen phagocytosis is monocytes, which differentiate into macrophages and dendritic cells. Every day, three separate leukopak samples were collected for CBC and DLC analysis.
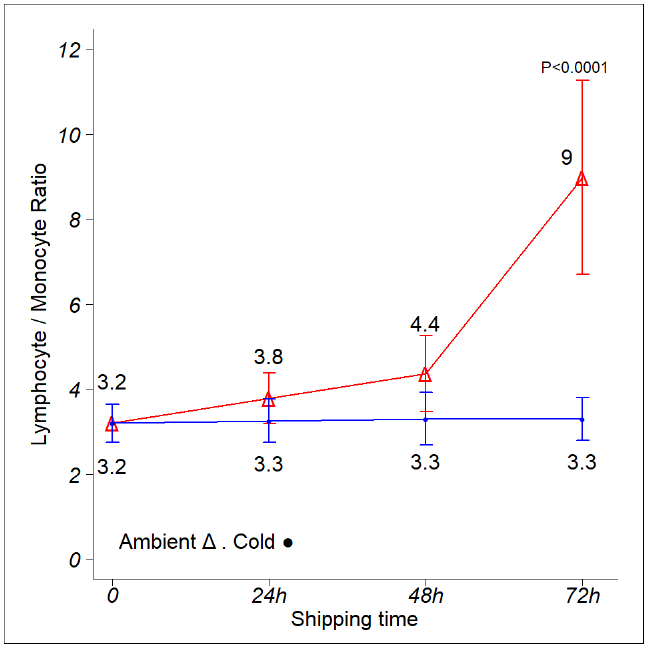
Figure 4. Effects of time and temperature on lymphocyte to monocyte ratios. Image Credit: BioIVT
Shipping circumstances were replicated by storing boxes at ambient temperature and 4 °C. Three leukopak samples were analyzed independently. Every day, CBC and DLC samples were collected, and the Lym/Mon ratio was computed for each donor, shipping time, and storage temperature.
The data points on the graph represent the mean Lym/Mon ratio determined during the study. The Lym/Mon ratio remained steady in cold boxes for the first 72 hours, then rapidly increased as the temperature increased (Fig. 1C and data not shown).
On ambient shipping, the Lym/Mon ratio increases gradually over the first 48 hours before quickly increasing at 72 hours. Error bars indicate one standard deviation from the mean. ANOVA with a post-hoc Tukey’s Honest Significant Difference Test.
Conclusion
This study compared how temperature fluctuations during shipping affect freshly collected leukopaks at ambient (about 21 °C) and cold (4 °C) temperatures.
Ambient boxes were stable at 21 °C for five days, while cold boxes acclimatized to 4 °C for approximately 72 hours before returning to ambient temperatures. PBMC viability remained above 95 % for 72 hours in cold transportation before dropping to 80 % at 96 hours.
In contrast, ambient shipping resulted in a quick drop to 75 % after 48 hours. Despite a minor viability fall to 94 % at 24 hours in ambient shipping, there was a noticeable rise in lymphocytes and a decrease in monocyte percentages, particularly at 48 and 72 hours.
Ambient shipping is only appropriate for same-day local destinations; cold shipping is strongly advised for longer periods of time and when probable transportation delays may negatively impact cell health. Further research comparing shipping methods and evaluating cell activation could yield useful information.
BiolVT utilizes NanoCool™ Cooling System shipping containers to ensure cell product quality for up to 72 hours. Even with overnight shipment, it is strongly recommended that low temperatures be used for maximum cell health. By following these instructions, researchers can avoid unanticipated changes in leukopak cell populations and testing failures.
References
- Kobayashi, D.T., et al. (2012). Evaluation of Peripheral Blood Mononuclear Cell Processing and Analysis for Survival Motor Neuron Protein. PLoS ONE, 7(11), p.e50763. DOI: 10.1371/journal.pone.0050763. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0050763.
- Abbas AK, L. A. Cellular and molecular immunology. vol. 5 (Saunders, Philadelphia, 2005).
- Jerram, A., et al. (2021). Effects of storage time and temperature on highly multiparametric flow analysis of peripheral blood samples; implications for clinical trial samples. Bioscience Reports, (online) 41(2), p.BSR20203827. DOI: 10.1042/BSR20203827. https://portlandpress.com/bioscirep/article/41/2/BSR20203827/227854/Effects-of-storage-time-and-temperature-on-highly.
- Johnson, R.K., et al. (2022). Peripheral blood mononuclear cell phenotype and function are maintained after overnight shipping of whole blood. Scientific Reports, 12(1). DOI: 10.1038/s41598-022-24550-6. https://www.nature.com/articles/s41598-022-24550-6.
- Kim, D.-W., et al. (2013). Overnight Storage of Blood in ACD Tubes at 4 °C Increases NK Cell Fraction in Peripheral Blood Mononuclear Cells. Annals of Clinical & Laboratory Science, (online) 43(3), pp.267–273. Available at: https://www.annclinlabsci.org/content/43/3/267.full.
About BioIVT
BioIVT, formerly BioreclamationIVT, is a leading global provider of high-quality biological specimens and value-added services. We specialize in control and disease state samples including human and animal tissues, cell products, blood and other biofluids. Our unmatched portfolio of clinical specimens directly supports precision medicine research and the effort to improve patient outcomes by coupling comprehensive clinical data with donor samples.
Our Research Services team works collaboratively with clients to provide in vitro hepatic modeling solutions. And as the world’s premier supplier of ADME-Tox model systems, including hepatocytes and subcellular fractions, BioIVT enables scientists to better understand the pharmacokinetics and drug metabolism of newly discovered compounds and the effects on disease processes. By combining our technical expertise, exceptional customer service, and unparalleled access to biological specimens, BioIVT serves the research community as a trusted partner in ELEVATING SCIENCE®.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.
Last Updated: Apr 22, 2026