Since its inception in the late sixteenth century, light microscopy has provided biologists with ever increasing knowledge about microorganisms and the world beyond the scope of the human eye.
The light microscope has however always been limited by the laws of physics, which according to Ernst Abbes’ diffraction limit research in 1873[1] limits resolution to approximately the size of 200nm.
In 2014, following many years of painstaking research, three scientists won the Nobel Prize for Chemistry because they had defied the laws of physics and provided a technique to enable light microscopes to examine the structures inside cells beyond the diffraction limit. Super-resolution microscopy was born.
This new method allows images to be viewed with a higher resolution than the diffraction limit [2]. The development of super-resolved fluorescence microscopy was described by the Royal Swedish Academy of Sciences as “bringing optical microscopy into the nano-dimension” [2].
Super-resolution imaging describes a range of techniques that can examine structures at a sub cellular level. The structures need to be accurately labeled with fluorophores, which upon stimulation with a laser during imaging emit light inside the diffraction limit.
The most recent development allows for fluorophores to be switched on and off using a different laser light wavelength. This allows images of different areas to be accumulated without the fluorophores all emitting at once and confusing the image.
Super-resolution imaging
A whole range of ‘super-resolution’ fluorescence microscopy techniques have been developed to overcome the diffraction barrier [3]. These include:
- Stimulated emission depletion (STED) microscopy
- Reversible saturable optically linear fluorescence transitions (RESOLFTs) [4]
- Saturated structured-illumination microscopy (SSIM) [5].
Super-resolution fluorescence imaging permits dynamic processes to be investigated at resolutions of tens of nanometers in living cells, and as a matter of course numerous cellular structures have been imaged at sub-diffraction-limit resolution [3].
Fluorescence Photoactivated Localization Microscopy (FPALM)
FPALM [3] is a more recent development in super-resolution imaging. Here the technique uses photo-switchable fluorescent dyes or proteins. These may be activated by light at a different wavelength aside from the imaging light that excites fluorescence and deactivates the fluorophores.
By separating activation and imaging there is control over the fraction of molecules in the fluorescent state at a given time; this means that activated molecules are optically resolvable from each other and can be precisely localized.
By repeating the activation and imaging process the locations of fluorophores is mapped, allowing a super-resolution image to be constructed from these fluorophore locations to produce 3D images, even in live cells. The achievable resolution of these fluorescence microscopy techniques may in theory achieve molecular scale [3].
Applications
HIV transfer
Myeloid dendritic cells (DCs) are antigen presenting cells that play a huge role in sensing pathogens. It has however been suggested that HIV has ‘hijacked’ these cells to spread to CD4+Tcells and spread in a systemic fashion [6].
FPALM microscopy using a Vutara 200 super-resolution microscope (Bruker Corporation) was used as part of the investigation of this process. By using a biplane FPALM system, samples were imaged using 647nm and 488nm excitation lasers, respectively, and a 405nm activation laser.
Super-resolution microscopy revealed a close association of CD169 and HIV1 particles in surface accessible but deep plasma membrane invaginations, which effectively protect the virus from antigens [6].
Sarcomere change
Sarcomeres form the basic contractile units of cardiac myocytes, and some studies have revealed remodeling of sarcomeric proteins in several diseases, including genetic defects and heart failure.
This study used FPALM super-resolution microscopy as one method to investigate the remodeling of sarcomeric α-actinin in two different models of heart failure [7].
Isolated sarcomere cells from animal models were examined using a Vutara SR 200 microscope using a 647nm excitation laser and a 405nm activation laser. Resolutions of 20nm in the X-Y plane and 50nm in the Z direction were obtained.
The images obtained by super-resolution microscopy showed irregular α-actinin deposits in a longitudinal direction in sarcomeres from failing hearts indicating that repair of heart tissue does occur but not in such a regular efficient fashion [7].
A virus at work
The infection life-cycle mechanism of RNA viruses is fairly well known by microbiologists. However new super-resolution imaging FPALM methods were used in this study to image single virions infecting live cells and also filamentous virion fusion.
By using fluorescently labeled viral proteins and multiply labeled tetravalent RNA imaging probes (MTRIP) SR images were obtained of viral mechanism.
Using a Vutara SR 200 and single-molecule localization biplane FPALM technology, viral proteins and MTRIP probes were imaged using 647nm and 561nm excitation lasers, respectively, and a 405nm activation laser.
The total number of frames acquired per channel ranged from 2000 to 4000. The study demonstrated the utility of the MTRIP probe in studying viral mechanism and also how SR microscopy can help study this mechanism at sub-cellular resolution [8].
Ovarian cancer
Polymer-based anticancer therapeutics rely upon conjugates that will sustain a high concentration gradient of therapeutic compound between the vasculature and solid tumor. In this study, ovarian cancer was treated using hydroxypropyl methacrylamide (HPMA) copolymer carriers primed with Paclitaxel.
As part of the observations, 3D stochastic optical reconstruction microscopy (STORM) was used with a Vutara SR-200 fluorescence microscope (this method is also known as FPALM [3]). STORM images were obtained of the movement of fluorescently labeled drug conjugate FITC-P-Cy5.
These images visualized the cellular endocytosis of intact conjugates and also the diffusion of the drug model from lysosomes into the cytoplasm [9]. The results were encouraging for the development of second generation ovarian cancer treatment.
Commercially available super-resolution systems
The Vutara super-resolution microscopes are among the fastest on the market, and are ideal for a range of advanced image applications. Super-resolution microscopy has been elevated to a new level by Bruker with the Vutara 352.
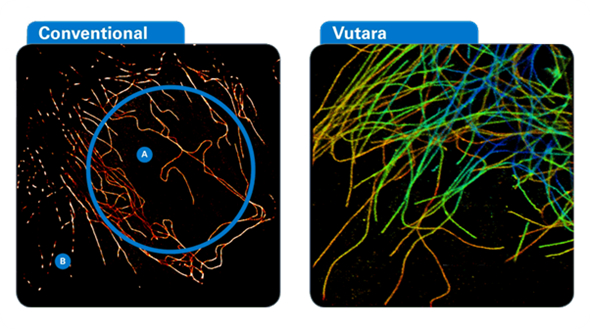
Equipped with ResEnhanced™ technology, the microscope allows simultaneous four-color super-resolution imaging, including a 750 nm excitation laser. The Vutara has 1000 mW lasers, sCMOS detection, and with high-speed GPU and CPU processing, it can provide >3000 fps image acquisition, allowing live-cell imaging and the ability to ‘freeze frame’ cellular activity at nanoscale dimensions.

References
- Abbe, E. (1873). "Beitrage zur Theorie des Mikroskops und der mikroskopischen Wahrmehmung". Archiv für Mikroskopische Anatomie (in German) 9: 413–420.
- Chang, Kenneth (8 October 2014). "2 Americans and a German Are Awarded Nobel Prize in Chemistry, http://www.nytimes.com/2014/10/09/science/nobel-prize-chemistry.html?_r=0
- Bo Huang, Mark Bates, and Xiaowei Zhuang, Super resolution fluorescence microscopy, Annu Rev Biochem. 2009; 78: 993–1016.
- Hell SW. Far-field optical nanoscopy. Science. 2007;316:1153–1158
- Gustafsson MGL. Nonlinear structured-illumination microscopy: wide-field fluorescence imaging with theoretically unlimited resolution. Proc. Natl. Acad. Sci. USA. 2005;102:13081–13086
- H. Akiyama, N-G. PinaRamirez, M.V.Gudheti, S. Gummuluru, CD169-Mediated Trafficking of HIV to Plasma Membrane Invaginations in Dendritic Cells Attenuates Efficacy of Anti-gp120 Broadly Neutralizing Antibodies , PLOS Pathogens, DOI:10.1371/journal.ppat.1004751 March 11, 2015
- J. G. Lichter, E. Carruth, C. Mitchell, A. S. Barth, et al., Remodelling of the sarcomeric cytoskeleton in cardiac ventricular myocytes during heart failure and after cardiac resynchronization therapy, The Journal of Molecular and Cellular Cardiology, July 2014Volume 72, Pages 186–195
- E. Alonas, A. W. Lifland, M. Gudheti, D. Vanover, et al., Combining Single RNA Sensitive Probes with Subdiffraction-Limited and Live-Cell Imaging Enables the Characterization of Virus Dynamics in Cells, ACS Nano, 2014, 8 (1), pp 302–315
- R.Zhanga, J.Yanga, M. Simaa, Y. Zhoua, and J. Kopečeka, Sequential combination therapy of ovarian cancer with degradable N-(2-hydroxypropyl)methacrylamide copolymer paclitaxel and gemcitabine conjugates , PNAS August 19, 2014 vol. 111 no. 33 12181-12186
About Bruker Nano Surfaces and Metrology

Bruker’s suite of fluorescence microscopy systems provides a full range of solutions for life science researchers. Their multiphoton imaging systems provide the imaging depth, speed and resolution required for intravital imaging applications, and their confocal systems enable cell biologists to study function and structure using live-cell imaging at speeds and durations previously not possible. Bruker’s super-resolution microscopes are setting new standards with quantitative single molecule localization that allows for the direct investigation of the molecular positions and distribution of proteins within the cellular environment. And their Luxendo light-sheet microscopes, are revolutionizing long-term studies in developmental biology and investigation of dynamic processes in cell culture and small animal models.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.