Systems neuroscience research in recent years has been revolutionized by development of tools and techniques to aid in the understanding of neural circuits. Many of the more difficult questions relate to complex processes such as perception of stimuli, formation of memories in learning, or alterations in behavior based on social contexts.
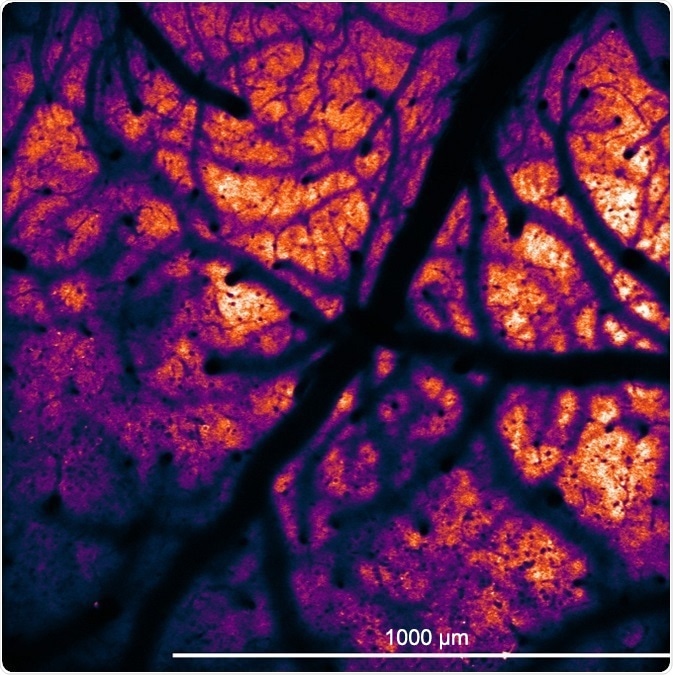
Large FOV of GCaMP6 neurons in transgenic mouse.

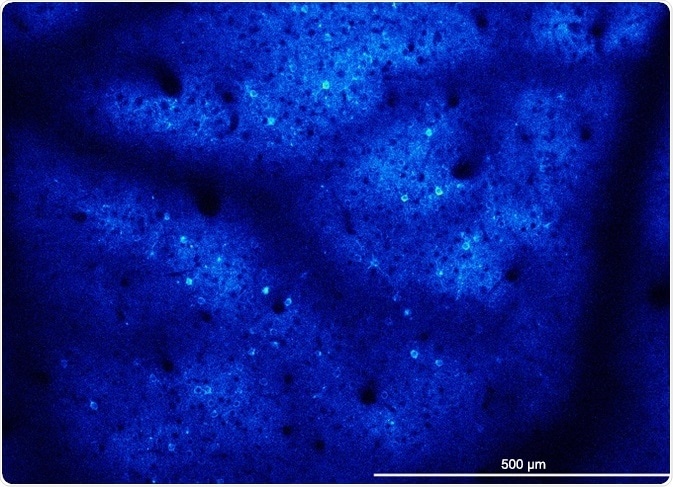
GCaMP6-expressing neurons firing in neocortex of a transgenic mouse captured at 15 fps. Image and video courtesy of Adam Packer, Robert Lees, and James Rowland (Packer lab, University of Oxford).
To study these higher-level problems, methods needed to be developed that were compatible with alive, awake, and behaving animal models.1,2,3 One of the technologies that has been instrumental in this effort has been 2-photon or multiphoton fluorescence imaging. The adoption of 2-photon microscopy in neuroscience has allowed for in-vivo imaging deep into highly light-scattering tissue, such as within a mouse brain. Coupled with fluorescent probes, such as the family of genetically-encoded calcium indicators like GCaMPs, scientists could now image and record neural activity with cellular resolution in real-time.
More accurate measurement of neuron action potentials is becoming possible as fluorescent probes improve (such as with genetically-encoded voltage indicators). And as scientists begin to interrogate more complicated circuits, microscopy innovation also needs to advance. Fast xyz scanners and large field-of-view sizes, such as is possible with the Bruker Ultima 2Pplus multiphoton workstation, enable capture of circuit dynamics that span larger distances in the brain. To prove hypotheses on the function of individual circuits, the advent of optogenetic probes,4 allowing for light-activation of specific neurons, has been invaluable.
GCaMP6-expressing neurons in the neocortex of a transgenic mouse (JAX 024742). Courtesy of Adam Packer, Robert Lees, and James Rowland (Packer lab, University of Oxford).
Scientists (e.g., Packer et. al., 2015) have ingeniously used the channelrhodopsin probe and its 2-photon counterparts to induce activation of neural ensembles, demonstrating functional interactivity correlated with animal behavior.1 This all-optical feedback loop of recording activity and stimulating neurons correlated to animal behavior prompted the collaborative development of spatial light modulator (SLM) stimulation technology, commercialized in the Bruker NeuraLight 3D. The SLM confers the capability to target multiple neurons in three dimensions by injecting a hologram pattern of light into the sample. Subsequently, after activation of one set of neurons, the SLM properties can quickly be changed through software command, and a different set of neurons can be targeted.
As tools like SLMs become more refined, techniques like optogenetics become more accessible, and closed-loop imaging and stimulation experiments will push forward our understanding of how neurons at the cellular level contribute to function at the system level.
Acknowledgements
This article was written by Jimmy Fong, Bruker Product Manager, Multiphoton Microscopes.
References
- Packer AM, Russell LE, Dalgleish HWP, Häusser M (2015) Simultaneous all-optical targeted manipulation and recording of neural circuit activity with cellular resolution in vivo. Nature Methods 12:140-146.
- Bloem B, Huda R, Sur M, Graybiel AM (2017) Two-photon imaging in mice shows striosomes and matrix have overlapping but differential reinforcement-related responses. Elife 6
- Jennings JH, Kim CK, Marshel JH, Raffiee M, Ye L, Quirin S, Pak S, Ramakrishnan C, Deisseroth K. Interacting neural ensembles in orbitofrontal cortex for social and feeding behavior. Nature. 2019.
- Boyden ES, Zhang F, Bamberg E, Nagel G, Deisseroth K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat Neurosci. 2005 Sep;8(9):1263-8.
About Bruker Nano Surfaces and Metrology

Bruker’s suite of fluorescence microscopy systems provides a full range of solutions for life science researchers. Their multiphoton imaging systems provide the imaging depth, speed and resolution required for intravital imaging applications, and their confocal systems enable cell biologists to study function and structure using live-cell imaging at speeds and durations previously not possible. Bruker’s super-resolution microscopes are setting new standards with quantitative single molecule localization that allows for the direct investigation of the molecular positions and distribution of proteins within the cellular environment. And their Luxendo light-sheet microscopes, are revolutionizing long-term studies in developmental biology and investigation of dynamic processes in cell culture and small animal models.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.