Protein-based medicines are increasingly prevalent as a crucial category of contemporary therapeutics. The presence of subvisible particles (SbVPs) in these pharmaceuticals is a critical quality attribute (CQA) that is strictly regulated due to possible risks to product stability, quality, bioavailability, and patient safety.
Nonetheless, characterizing SbVPs using conventional optical techniques is difficult. The low refractive index of proteinaceous SbVPs complicates detection, limiting the capacity to guarantee that the CQA requirements established in compendial techniques and published in recognized pharmacopeias are satisfied.
A recent “Stimuli to the Revision Process” article from the US Pharmacopeia (USP) emphasizes the necessity for orthogonal techniques, including Flow Imaging Microscopy (FIM), to distinguish silicone oil droplets (SiOPs) from protein aggregates.1
Even though light obscuration (LO) is the leading compendial approach for quantifying subvisible particulate matter in parenteral pharmaceutical products, it frequently fails to identify these low-contrast particles. In contrast, FIM captures high-resolution images for direct measurement, offering in-depth morphological characterization to facilitate accurate differentiation of particle types.
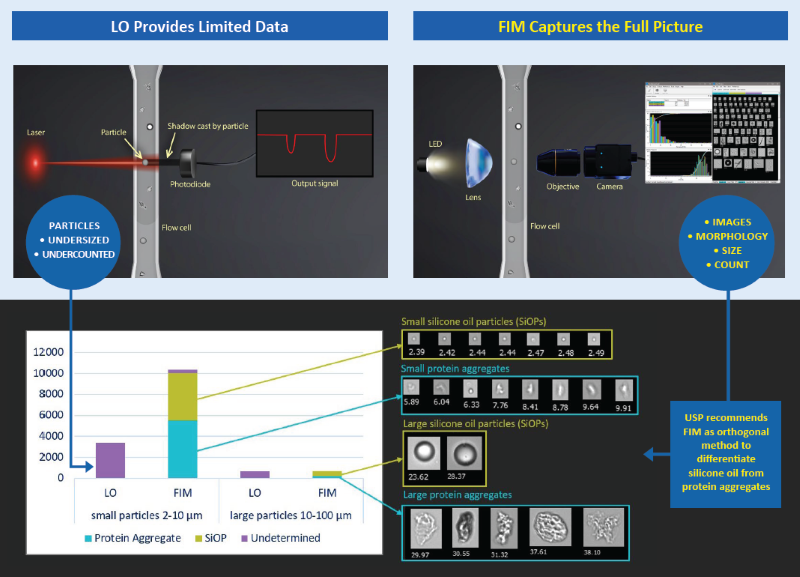
Image Credit: Yokogawa Fluid Imaging Technologies, Inc.
Addressing the regulatory and technical gaps in compendial approaches
The recent Stimuli article underscores persistent obstacles in characterizing SbVPs in protein treatments per USP <787>, in parenteral medicinal products per USP <788>, and more generally in medicinal injections and ophthalmic solutions per USP <1788>.
LO, a standard optical approach and the compendial analytical method, quantifies particles by identifying the amount of light they obstruct as they move through a laser, using the size of casted shadow to evaluate particles. However, LO is inefficient at identifying low-contrast particles, including protein aggregates and silicone oil particles (SiOPs), owing to their aforementioned low refractive index.
In addition, given that prefilled syringes (PFSs) lubricated with silicone oil have emerged as the favored primary container for biologics, it is essential to evaluate if and how SiOPs interact with medicinal proteins. These interactions may impact stability, aggregation behavior, and general product quality – an obstacle that LO cannot overcome on its own.
The study: Differentiating silicone oil droplets from protein aggregates using FIM
This research demonstrates the benefit FIM brings to conventional LO processes by offering the visual and quantitative evidence needed to close these gaps.
FlowCam, an FIM device, is used to demonstrate how morphological information can be employed to successfully count, measure, and classify SiOPs separately from proteinaceous SbVPs. By incorporating FlowCam into the analytical process, producers obtain the in-depth insights necessary to determine the primary causes of particle formation and create effective mitigation approaches when pharmaceutical products surpass compendial limits.
Download the full article to learn more

References & further reading
- Yokogawa Fluid Imaging Technologies, Inc. (2026). Webinar | USP | Addressing Subvisible Silicone Oil Droplets – Industry Challenges, Analytical Strategies, and USP’s Rationale for a New General Informational Chapter. (online) Available at: https://www.fluidimaging.com/webinar-usp-addressing-subvisible-silicone-oil-droplets.
About Yokogawa Fluid Imaging Technologies, Inc.
At Yokogawa Fluid Imaging Technologies our mission is to bring clarity and focus to the study of subvisible particles in the life sciences. With our FlowCam technology, we are committed to developing innovative hardware and software to monitor Earth’s water bodies and drinking water supply for hazardous algae, to help make vaccines and injectable medicines safer, and to improve product quality in a variety of applications.
FlowCam instruments combine the benefits of digital imaging, flow cytometry, and microscopy into a single solution - Flow Imaging Microscopy (FIM). Beyond traditional particle sizing and counting, FIM's image-based analysis allows for comprehensive characterization of subvisible API aggregates and contaminants in biopharmaceuticals, mammalian cells, microplankton, emulsions, and advanced materials. FlowCam instruments have been deployed in over 50 countries, supporting research, development, and environmental monitoring in the life sciences, materials research, and industrial applications.
In the video below you will learn about flow imaging microscopy and how FlowCam works to provide count, size, characterization, and digital images of subvisible particles.
FlowCam Explainer Video - What is Flow Imaging Microscopy?
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.