Imines are one of several reactive groups, called functional groups, that are present in a compound. The mechanism by which the compound reacts is attributed to the properties of its functional group.
Imines are one such functional group. It is comprised of a carbon atom doubled-bonded to a nitrogen atom:
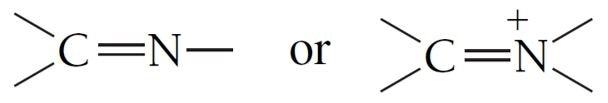
Figure 1. A pictorial representation of an imine.
It is sometimes called a Schiff base. They are formed from the reaction of two other functional groups; an amine (NH2) and an aldehyde (CHO) or a ketone (C=O). The latter two groups contain a carbon atom double bonded to a hydrogen. This is called a carbonyl group.
Imine Formation
The amine will attack the carbonyl group as the lone pair of electrons on the nitrogen atom is nucleophilic. Contrastingly, the carbonyl group is electron deficient due to the ability of oxygen to attract a bonding pair of electrons more strongly than carbon– a property called electronegativity.
One of the two shared pairs of electrons that constitute the double bond between C and O is transferred to O. Following a shift of the hydrogen atom attached to nitrogen (a process called tautomerization), the unshared electron pair on the nitrogen atom is shared with the electron-deficient carbon atom.
This forms the characteristic C=N double bond, and the oxygen is concurrently eliminated as water. The loss of the hydroxyl group is difficult, as the resultant hydroxyl ion is unstable. The ability to leave is enhanced in the presence of an acid.
This protonates the hydroxyl group, which transforms it into water, a considerably greater leaving group. Protonation increases the rate of the reaction by 17 orders of magnitude. The mechanism is illustrated in Figure 2.
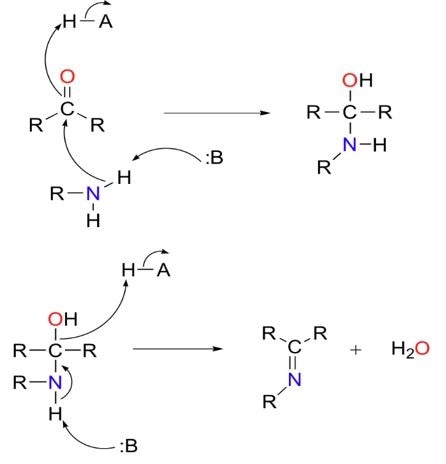
Figure 2 Imine formation. Mechanistically, imine formation is a two-step process. In step one (top) the amine attacks the carbonyl carbon. In step two (bottom), the nitrogen is deprotonated, and the oxygen of the carbonyl is eliminated as water.
Imine Hydrolysis
The reverse of imine formation is called imine hydrolysis, yielding the corresponding amine and carbonyl containing compound (aldehyde or ketone).
In this process, the imine undergoes an addition of water. Water requires an activation step before it can be added; a base (proton acceptor) pulls a hydrogen ion (proton) off the water molecule.
The resultant OH- (hydroxide ion) is an electrophic (electron pair donor); the lone pair on oxygen attacks the electron-deficient carbon of the imine group.
Carbon is electron-deficient as its electrons experience a pulling effect by the doubly-bonded nitrogen due to nitrogen’s higher electronegative effect.
Consequently, carbon can readily accept electrons, and is described as nucleophilic. Concurrently, one of the two shared pairs of electrons that constitute the double bond between C and N is replaced with a single bond to a hydrogen.
This hydrogen is provided by a base (proton donor) to the positively charged nitrogen of the imine. The attached hydroxyl group then loses another proton, through base attack; the lone pair that remains is used to form a double bond to carbon.
Concurrently, another acid provides a proton to the amine group to permit its elimination. This is illustrated in Figure 3.
Relevance of Imines in Biochemistry
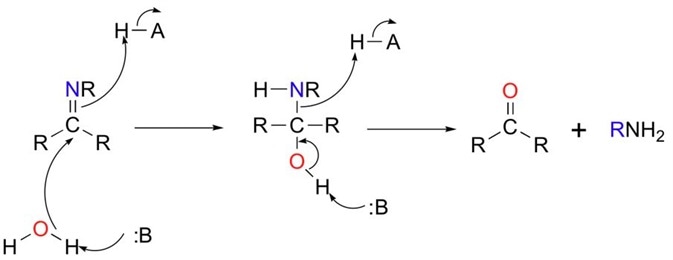
Figure 3 Imines can be hydrolysed (split with water) to give the starting aldehyde (or ketone) and amine. Note that the reaction is acid assisted.
Schiff base (imine) formation and hydrolysis are highly important in biological chemistry. Many enzymes employ imines as part of their reaction mechanisms. An important biological reaction is the formation of an imine bond between an amino acid carbonyl group and the amine of the amino acid lysine.
This is because they are highly reactive and can form adducts with other molecules. An adduct is the product of two molecules (in this case, one of which is the imine) that results in a single species. This adduction allows the molecule to experience the reactive effect of the imine. Imines are reactive due to their ability to act as electron ‘sinks’.
The nitrogen of the C=N bond is readily protonated, yielding a protonated imine. This serves as an electron-accepting group, which, when present in an adduct, can pull electrons away from the bonds in the attached molecule. This facilitates bond cleavage, which is an essential part of many metabolic reactions, for example, in glycolysis and amino acid metabolism.
Another crucial feature of imines, which has fuelled their biological relevance, is their ability to tautomerise, arranging the position of their proton and double bond, which also facilitates bond cleavage. The intermediate is called the iminium ion, and the resulting tautomer is called an enamine. This allows for regeneration of the imine and subsequent release of the adduct following hydrolysis.
Figure 3 Imine- enamine tautomerisation. Note, the intermediate is called the iminium ion. The reaction may be catalysed by an acid or a base (shown here). Enamines are the equivalent of enols in keto-enol tautomerism
Further Reading
Last Updated: Oct 30, 2018