Goodpasture syndrome is a condition characterized by renal and pulmonary symptoms due to the presence of anti-GBM antibodies.
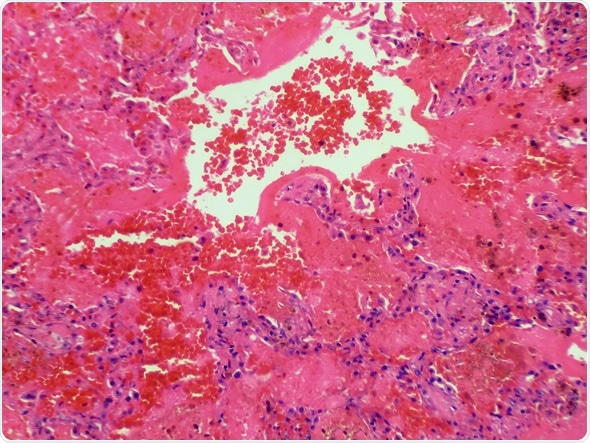
Goodpastures syndrome is a rare autoimmune disease in which antibodies attack the lungs and kidneys. Bleeding lung tissue is shown here. Magnification 200x - Image Copyright: Jubal Harshaw / Shutterstock
Symptoms
Goodpasture syndrome is first manifested as non-specific fatigue, nausea and vomiting, and a feeling of weakness. Patients may also report loss of appetite.
This may be followed, sometimes very rapidly, by pulmonary manifestations. These may precede or occur simultaneously with renal involvement, and include breathlessness, coughing and hemoptysis.
Hemoptysis used to occur in almost 70% of patients, and only in smokers. It often presented months or years before any symptom or sign of kidney disease arises. The falling prevalence of smoking may be responsible for the declining frequency of this manifestation of Goodpasture syndrome. However, chest infection or fluid overload can also precipitate pulmonary hemorrhage and hemoptysis. It may range from episodes of mild bleeding to catastrophic hemorrhage. It is the primary cause of death in the early phase of the illness.
Renal symptoms include:
- Hematuria
- Foamy urine
- A burning sensation during urination
- Rapidly developing pallor
- Edema of the legs or other areas of the body
- Hypertension
- Sudden onset of oliguria or anuria in some patients
Goodpasture syndrome may develop quickly over days or weeks and even insidiously in some cases.
Diagnosis
The diagnosis of Goodpasture syndrome may involve the following tests, based on the signs and symptoms:
Urine Testing
A urine sample is collected in a special container and analyzed for the presence of protein, blood, and bacterial growth through culture, if indicated - this may be done by dipstick testing. The presence of damaged red cells or of significant proteinuria indicates renal damage. Other findings include the presence of white cells, granular casts and red cell casts.
Blood Testing
As in all cases where autoimmune disease is suspected, the first screening will include:
- Full blood count
- A metabolic panel to rule out other causes of kidney damage
- Markers of acute inflammation such as CRP
- HLA typing
- Serologic tests for antinuclear antibodies, antiDNA antibodies, cryoglobulins, ANCA and anti-GBM antibodies
Besides specific anti-GBM antibodies, ANCA testing is also positive in many patients, especially anti-myeloperoxidase (MPO) or p-ANCA antibodies.
Chest X-rays
Pulmonary symptoms are evaluated by a chest X-ray used to visualize any lung abnormalities.
Renal Biopsy
A renal biopsy involves removing a small sample of tissue from the kidney for microscopic evaluation to assess the presence, extent and type of abnormalities in the tissue. This can pinpoint the type of lesion, confirm the diagnosis and guide the prognosis. Biopsy is guided by ultrasound imaging or CT imaging and is performed under local anesthesia. Typical crescentic areas of glomerular proliferation are seen in more than half of the glomeruli. Immunofluorescent (IF) staining of the tissue shows the classical lines of antibody deposited on the GBM.
An important proviso in the diagnosis of Goodpasture syndrome is that it is based upon the typical signs and symptoms of pulmonary and renal damage, in association with markedly raised levels of a specific autoantibody. A demonstration of serological positivity is not enough in the absence of clinical features of the disease. This is because even healthy individuals may show the presence of autoantibodies, as may others who have non-immune disease.
Demonstration of Autoantibody to the GP Autoantigen
The autoantigen specific to Goodpasture syndrome is the NC1 domain of the alpha-3 chain of the type IV collagen that makes up the basement membrane of the glomeruli and the pulmonary alveoli. ELISA or IF are used to detect the presence of anti-GBM autoantibodies. Western blotting is used to resolve doubts in higher centers.
However, anti-GBM antibodies in serum may be absent if the only clinical feature is diffuse alveolar hemorrhage. In such patients, immunofluorescence of lung tissue will show the presence of the characteristic IgG deposits on the basement membrane of the alveoli. In a few patients, IgA or IgM is found instead.
References
Further Reading
Last Updated: Jan 2, 2023