This article is based on a poster originally authored by Alexis T.P. Ngo, Acilegna G. Rodriguez, Jian-Lu Chen, Zachariah J. Builta, Xuexiang Zhang, and Mark S. Warren.
The central nervous system (CNS) contains various potential targets for pharmacological research. Dysfunction in CNS pathways can cause a number of neurological disorders, including Alzheimer's and Parkinson's disease, as well as psychiatric disorders like depression, schizophrenia, and bipolar disorder.
Developing treatments involves not only developing molecules that are mechanistically effective, but also figuring out how to get those molecules across the blood-brain barrier (BBB), making the brain one of the most unreachable organs for therapeutic treatments.
In addition to physical barriers at the BBB, a number of efflux transporters, including BCRP (ABCG2) and P-gp (ABCB1), work together to inhibit drug intake.
Despite the existence of efflux transporters, the BBB has a large number of uptake transporters, notably OATP1A2 (SLCO1A2), which may provide a mechanism to carry medicines across the barrier.
To see if OATP1A2 uptake might overcome efflux by BCRP and P-gp, the researchers employed OPTI-EXPRESSION™ technology to create MDCK-II cells expressing OATP1A2, BCRP, and P-gp individually and in multi-transporter models.
This assay technique has the benefit of allowing analysis of uptake into polarized MDCK-II cells from the apical side (where OATP1A2, BCRP, and P-gp are all expressed) and apical to basolateral (A→B) transcellular transport through the cell monolayer.
The researchers employed these devices to examine a wide range of medicines that are known to be BCRP and P-gp efflux transporter substrates. Pravastatin uptake was 3.9-fold higher in cells expressing both OATP1A2 and P-gp compared to cells expressing only P-gp, with 9.3-fold higher A→B transcellular transport.
P-gp decreased rosuvastatin uptake, while cells expressing both OATP1A2 and P-gp had 4.1-fold more transcellular transport than cells expressing solely P-gp.
Triptans demonstrated comparable results, with both Almotriptan and Zolmitriptan showing enhanced uptake and 5 to 6-fold increased A→B transcellular transport in cells expressing both OATP1A2 and P-gp compared to cells expressing only P-gp.
OATP1A2 also boosted Fexofenadine and Methotrexate absorption and transcellular transport by more than sixfold, even when P-gp was expressed.
These findings demonstrate that, whereas the efflux transporter P-gp inhibits the apical permeability of many medicines, coexpressing an apical uptake transporter such as OATP1A2 can partially overcome this efflux and boost brain permeability for substrates.
This study demonstrates that using multi-transporter models with uptake and efflux transporters could be a viable screening method for BBB penetration by CNS treatment candidates.
Methods
MDCK-II and MDCK-MDR1 cells were cultured in DMEM with low glucose and 10 % FBS.
Cells were seeded into Millipore 96-well insert plates (PCF, 0.4 μm). MDCK-II and MDCK-MDR1 cells were then transfected with one or more plasmids encoding OATP1A2, BCRP, or GFP (as a control) utilizing BioIVT's unique OPTIEXPRESSION™ technology, which enables consistent and successful in-situ transfection of polarized cell monolayers.
Assays were performed roughly 24 hours after transfection to allow transporter expression and localization in polarized cells. Transcellular tests were performed by administering test chemicals to the apical compartment in HBSS with 25 mM Bis-Tris at pH 6.0, whereas the basolateral chamber contained just HBSS.
Following a 60-minute incubation, samples were collected from both the original apical "donor" chamber and the opposite basolateral "receiving" chamber. The cells were then rinsed and solubilized with 50 % acetonitrile to determine intracellular substrate accumulation.
Substrates were applied to either the apical or basolateral chambers, while the adjacent chamber was left buffer-free. After a 5-minute incubation, cells were washed and solubilized with 50 % acetonitrile to determine substrate accumulation intracellularly.
Results

Figure 1. Assay Setup. Image Credit: BioIVT
MDCK-II or MDCKMDR1 cells are seeded into Millicell-96 cell culture insert plates. Following seeding, the cells polarize and form tight junctions, resulting in a cell monolayer with apical and basal membranes. Following transfection, transporters locate to the proper membrane.
Figure 2. Compounds tested. Image Credit: BioIVT
All of these substances are substrates for the uptake transporter OATP1A2 and one of the efflux transporters BCRP or P-gp.
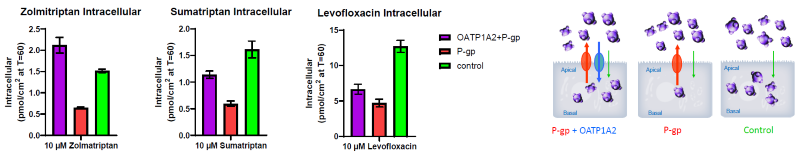
Figure 3. Uptake of Various Compounds Across the Apical Membrane. Image Credit: BioIVT
When chemicals are applied to the apical chamber, some will enter the cells via endogenous processes, as observed in control cells. When P-gp is produced, substrates are effluxed back into the apical chamber, reducing intracellular accumulation.
When OATP1A2 is expressed in cells that simultaneously express P-gp, OATP1A2 substrates accumulate intracellularly, showing that OATP1A2 uptake can partially balance P-gp export.
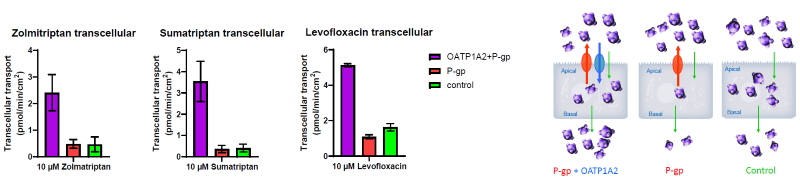
Figure 4. Transcellular Transport of OATP1A2 + P-gp Substrates Through the Cells and Across the Basolateral Membrane. Image Credit: BioIVT
When substrates are applied to the apical chamber, OATP1A2 promotes intracellular accumulation and transcellular transport across MDCK cells, despite P-gp efflux activity.
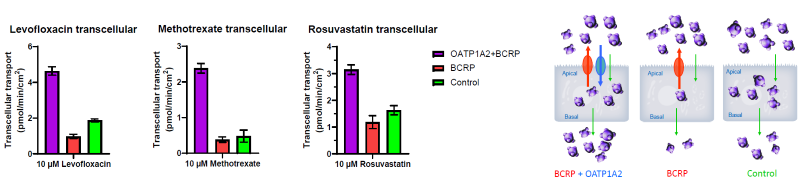
Figure 5. Transcellular Transport of OATP1A2 + BCRP Substrates Through the Cells and Across the Basolateral Membrane. Image Credit: BioIVT
Similarly, when substrates are added to the apical chamber, OATP1A2 promotes intracellular accumulation and transcellular transport across MDCK cells, despite BCRP's efflux activity.
Discussion and conclusions
A number of efflux transporters found at the blood-brain barrier (BBB) prevent many potential therapeutic medications from entering the brain.
P-glycoprotein (P-gp), breast cancer resistance protein (BCRP), and multidrug resistance-associated proteins (MRPs) are all efflux transporters that actively pump a variety of small molecules from brain endothelial capillary cells back into the bloodstream, limiting CNS exposure and potentially reducing the efficacy of many pharmacological agents.
Several drugs are known to be able to penetrate the BBB, with many employing active uptake transporters such as OATP1A2. Several substances known to be substrates of both OATP1A2 and at least one of the efflux transporters BCRP and P-gp were evaluated in MDCK-II cells that expressed uptake, efflux, or both transporters.
Despite being P-gp substrates, zolmitriptan, sumatriptan, almotriptan, and levofloxacin demonstrated higher transcellular transport in cells expressing OATP1A2 plus P-gp than in cells expressing P-gp alone.
Rosuvastatin, Pravastatin, Levofloxacin, and Methotrexate all showed higher transcellular transport in cells expressing OATP1A2 and BCRP compared to cells expressing BCRP alone.
Because compounds such as triptans and statins can cross the BBB, these multi-transporter models may help forecast which novel medication candidates will be able to enter the CNS.
With the push to limit the usage of animal models in research, effective non-animal models such as these MDCK cells coexpressing several uptake and efflux transporters may be very valuable.
In addition to OATP1A2, the BBB expresses a variety of additional uptake transporters, including organic cation transporters such as OCTs and OCTN2, monocarboxylate transporters such as MCT1, and amino acid transporters like LAT1.
Similar multi-transporter models may be easily built with OPTI-EXPRESSION™ technology to see if drug candidates could harness those transporters to overcome efflux and perhaps obtain access to the brain.
Designing compounds to take advantage of uptake transporters, such as OATP1A2, may aid in overcoming the limiting effects of efflux transporters at the BBB, allowing potential therapeutic medicines to reach their destinations in the CNS.
These multitransporter models could be useful in screening medication candidates to identify those with a higher likelihood of crossing the BBB.
About BioIVT
BioIVT, formerly BioreclamationIVT, is a leading global provider of high-quality biological specimens and value-added services. We specialize in control and disease state samples including human and animal tissues, cell products, blood and other biofluids. Our unmatched portfolio of clinical specimens directly supports precision medicine research and the effort to improve patient outcomes by coupling comprehensive clinical data with donor samples.
Our Research Services team works collaboratively with clients to provide in vitro hepatic modeling solutions. And as the world’s premier supplier of ADME-Tox model systems, including hepatocytes and subcellular fractions, BioIVT enables scientists to better understand the pharmacokinetics and drug metabolism of newly discovered compounds and the effects on disease processes. By combining our technical expertise, exceptional customer service, and unparalleled access to biological specimens, BioIVT serves the research community as a trusted partner in ELEVATING SCIENCE®.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.
Last Updated: Apr 23, 2026