The rapid advancement and widespread use of immune checkpoint inhibitors for cancer therapy purposes have revolutionized the way in which the disease is treated. As immunotherapy becomes a more viable treatment option for cancer patients, it is crucial that healthcare providers have a clear understanding of both the advantages and disadvantages associated with immune checkpoint inhibitor therapy.
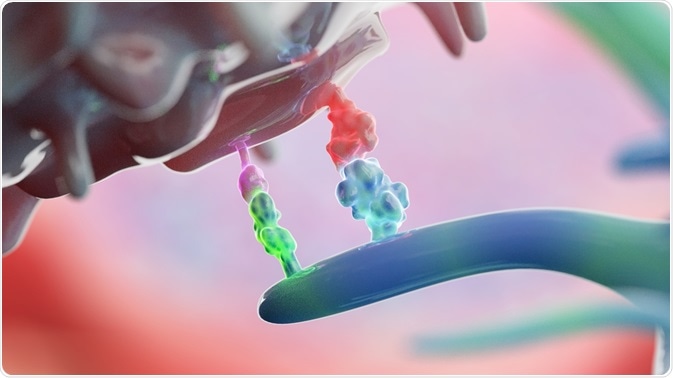 Image Credits: Alpha Tauri 3D Graphics / Shutterstock.com
Image Credits: Alpha Tauri 3D Graphics / Shutterstock.com
What are immune checkpoints?
Immune checkpoints are naturally present throughout the immune system and are primarily responsible for regulating the immune response. The activation of immune checkpoints occurs when proteins found on the surface of immune cells like T cells, interact with partner proteins that are present on the surface of other cells. This interaction ultimately causes the checkpoint protein to bind with the partner protein, thereby preventing any immune response from occurring.
What are checkpoint inhibitors?
Unfortunately, several cancer cells are associated with checkpoint proteins that are present on their surface. As a result, the immune system is unable to recognize these cancer cells as foreign, thereby allowing for these types of cancers to continue growing.
In an effort to overcome this innate response, several immune checkpoint inhibitors have been developed for use as immunotherapy drugs. By preventing the checkpoint proteins on immune cells from binding to those present on the surface of cancer cells, immune cells can remain active and capable in their ability to destroy cancer cells.
Advantages
Several different types of cancers have been found to respond well to treatment with immune checkpoint inhibitors, some of which include breast, bladder, cervical, colon, liver, lung, skin, stomach, and rectal cancer.
The two most notable classes of checkpoint inhibitors that have been used for the management of cancer include cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1) ligand inhibitors. Some of the advantages associated with certain immune checkpoint inhibitors include commercial availability, greater survival time and high potency against cancer cells.
Disadvantages
Despite the advantages associated with immunotherapy by checkpoint inhibitors, there are serious disadvantages that can be associated with this type of cancer treatment.
Side effects
Although immune checkpoint inhibitors are generally well-tolerated, their use is associated with several adverse effects including fatigue, nausea, vomiting, diarrhea, and dermatological reactions. The occurrence of severe immune-related adverse events (irAEs) has also been reported following the treatment of cancer patients with certain checkpoint inhibitors.
In fact, such reported irAEs have led to hospitalizations and the need for patients to receive supplemental treatments for treating these effects. Furthermore, such severe irAEs can also lead to permanent health effects, such as hypophysitis, diabetes, and neurological issues.
The tumor microenvironment
Despite the impressive clinical efficacy that has been demonstrated by certain checkpoint inhibitors in the treatment of various cancers, several research studies have found that the immunosuppressive tumor immune microenvironment (TIME) is responsible for preventing many patients from experiencing such benefits.
For example, the TIME of hepatocellular carcinoma (HCC) has been widely studied for its ability to coexist and successfully interact with immune cells to promote its metastatic growth throughout the body.
In addition to allowing HCC cancer progression, the TIME, which can be characterized by an abundance of immunosuppressive cells including tumor-associated macrophages, neutrophils, and myeloid-derived suppressor cells (MDSCs), also limits the efficacy of immunotherapy, particularly that by immune checkpoint inhibitors.
Conclusion
Overall, immune checkpoint inhibitor therapy has demonstrated its unique ability to treat highly malignant and devastating forms of cancer that were previously untreatable. In an effort to overcome some of the inherent limitations of this type of immunotherapy, several current research endeavors are investigating how combination therapies between checkpoint inhibitors and other forms of antineoplastic treatment may benefit patients for years to come.
Sources
- “Immune Checkpoint Inhibitors” – National Cancer Institute
- Spain, L., & Larkin, J. (2016). Weighing up the pros and cons of immune checkpoint inhibitors in the treatment of melanoma. Immunotherapy 8(6). DOI: 10.2217/imt.16.6.
- McLoughlin, K. C., Brown, Z. J., Shukla, Y., & Shukla, V. (2018). Promise and Pitfalls of Immune Checkpoint Inhibitors in Hepato-Pancreato-Biliary Malignancies. Discovery Medicine.
- Hodi, F. S., O’Day, S. J., McDermott, D. F., Weber, R. W., Sosman, J. A., et al. (2010). Improved Survival with Ipilimumab in Patients with Metastatic Melanoma. The New England Journal of Medicine 363; 711-723. DOI: 10.1056/NEJMoa1003466.
- Sosa, A., Lopez Cadena, E., Olive, C. S., Karachaliou, N., & Rosell, R. (2018). Clinical assessment of immune-related adverse events. Therapeutic Advances in Medical Oncology. DOI: 10.1177/1758835918764628.
- Lu, C., Rong, D., Zhang, B., Zheng, W., Wang, X., et al. (2019). Current perspectives on the immunosuppressive tumor microenvironment in hepatocellular carcinoma: challenges and opportunities. Molecular Cancer 18(130). DOI: 10.1186/s12943-019-1047-6.
Further Reading
Last Updated: Mar 16, 2020