Elastin is an extracellular matrix protein that lends elasticity and resilience to tissues such as the arteries, lungs, tendons, skin, and ligaments. Elastic fibers have two components, one of which is encoded by the ELN gene.
This protein has a high proportion of hydrophobic amino acids like glycine and proline, forming mobile hydrophobic domains. ELN is located on chromosome 7 of the human genome. Alternative splicing of the ELN gene results in the formation of at least 11 tropoelastin isoforms in humans.
Elastin is synthesized from the precursor molecule tropoelastin. Many molecules of tropoelastin are linked together to form the larger elastin complex.
Each molecule of tropoelastin has 36 domains arranged in a random coil. It has alternating hydrophobic and hydrophilic domains encoded by separate regions of the gene.
The hydrophilic domains contain lysine residues that are important for crosslinking the tropoelastin molecules during the formation of the elastin fiber.
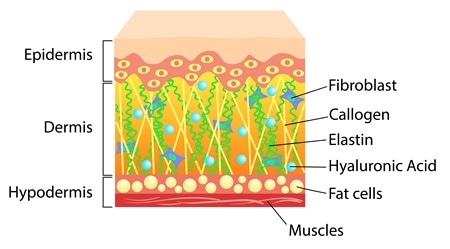
Skin cross section. Image Credit: Elh / Shutterstock
Elastin in Tendon
Tendons are made of fascicles with an interfascicular matrix (IFM). Some tendons can store energy and those that do are more elastic than positional tendons. Some examples of energy-storing tendons are the equine superficial digital flexor tendon and the human Achilles tendon.
With aging, the IFM of tendons becomes stiffer and less resistant to fatigue. These tendons are thus more prone to injury. Whereas the overall elastin content in tendon is low, IFM is rich in elastin.
In a study investigating elastin in tendon and the effects of aging on elastin content, it was found that elastin provides the IFM with an elastic recoil property which is necessary for storing energy, and with aging there is a decrease in elastin content and more disorganization within energy-storing tendons.
Elastin: the rubber band of life | Naren Vyavahare | TEDxGreenville
Elastin in Arteries
Elastin is one of the major structural components of the extracellular matrix of the blood vessel wall.
When elastin degrades, it releases elastin-derived peptides (EDP) called elastokines. These peptides have effects on other cells, including endothelial cells, monocytes, and smooth muscle cells, through interactions with the elastin receptor complex.
It is believed that EDPs play an important role in age-related vascular disease. Accumulations of EDP have been shown to promote hyperglycemia and insulin resistance in mice. Vascular aging can also lead to hemostasis dysregulation and blood clotting.
Elastin in Lungs
The alveoli, or air sacs, of the lungs are like little balloons that fill up with air during breathing. Elastin provides pressure on the balloons to prevent hyperinflation and maintain proper lung function. If the elastin is damaged, such as occurs due to inflammation caused by inhaled irritants in the case of emphysema, pressure in the alveoli is lost and airflow is reduced. The lungs may overinflate, making them vulnerable to further damage.
Elastin in Skin
Elastin and collagen are the main structural protein components of the skin. Elastin is about a thousand times more flexible than collagen. It lends elasticity to the skin tissue.
Damage or inflammation of the elastin in skin can lead to breakdown or disease of the skin.
Hereditary Diseases
Deletions and mutations of elastin-encoding genes are associated with some inherited diseases. Those include:
- Marfan syndrome: The cross-linking of aortic elastin is reduced leading to increased fragility of the vessel
- Buschke-Ollendorff syndrome: Non-cancerous growths of elastic fibers (and sometimes of collagen) are widespread throughout the body
- Pseudoxanthoma elasticum: Elastic tissue in the body is mineralized, with deposits of calcium occurring in the tissue
Further Reading
Last Updated: Jul 19, 2023