Please can you introduce your position at UCL?
I'm Bernard Siow and I'm an MRI physicist working here at CABI - the Center for Advanced Biomedical Imaging. I also have a position at the Francis Crick Institute as head of the MRI facility there.
What is CABI?
CABI is a preclinical imaging laboratory where we have about ten modalities. We started off with MRI and we have a 9.4T high-field system.
We have expanded our modalities since then and now have PET and SPECT/CT nuclear imaging, bioluminescent confocal microscopes, the OPT - optical projection tomography, and the ICON-1T system.
Working with Compact MRI
What are you currently working on?
I'm mainly developing the imaging areas at the Francis Crick Institute at the moment, which is going to open later this year. There's quite a lot of work to be done to set up the lab and get it going and it is currently taking up a lot of my time.
In terms of research projects, what I'm really interested in is a technique called diffusion MRI. The beauty of using this technique is that it can give you information about the microstructure of tissue.
On a clinical scanner, the MRI voxel is probably about a millimeter in length, whereas the kind of spatial information that we want is a thousand times smaller than that, it’s about a micron in size.
So, how do we get spatial information on the scale of microns when the actual voxel is about a millimeter long? We use diffusion MRI, because we can look at the way water diffuses within the cell.
If you have an axon for example, which is a long, thin tube, water is relatively free to diffuse along it, but it is restricted when it comes to going across the axon.
By looking at different diffusion times, we can infer the size of the axon. In particular, axon diameter estimates are important in the diagnosis of many neurodegenerative diseases.
Another new area I’m looking at that is associated with microstructure is cell permeability, which is abnormal in many neurodegenerative diseases such as multiple sclerosis.
We hope to be able to use these sequences in many neurodegenerative diseases, as well as things like stroke and heart disease. It is going to be a useful tool that utilizes very interesting physics, and that's what I'm really interested in - using clever physics and MRI.
We're just starting on this project, and hopefully we'll be able to develop something fairly soon.
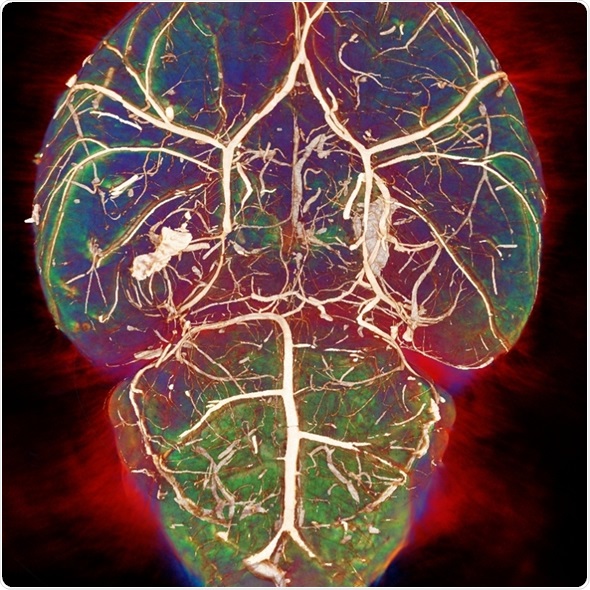
What is multimodality?
Each modality looks at tissue in a different way. For example, MRI has very good soft tissue contrast and there is a number of parameters that are associated with different pathologies.
For example, there are T1 and T2 changes in cancer and neurodegenerative diseases, whereas, with a modality like CT, there's less soft tissue contrast and it would be used more to look at things like bone structure. It uses a different type of contrast and produces images through a different physical mechanism.
By using different modalities, we can get much more information compared with using only one modality.
What other advantages are there of using a multimodality approach?
If you want to look at something that's very specific, then we have the option of doing that as well. There are limitations with each modality, and by having lots of modalities, we don't restrict ourselves so much. We can perform many different studies and we can look at a really wide range of diseases.
What are the main reasons why you use MRI?
The MRI has a very good soft tissue contrast. The parameters that I generally look at using MRI are called T1 and T2. These vary in different tissues, obviously, but also across different pathologies. MRI provides very high sensitivity to these pathologies, which can't be seen using other modalities.
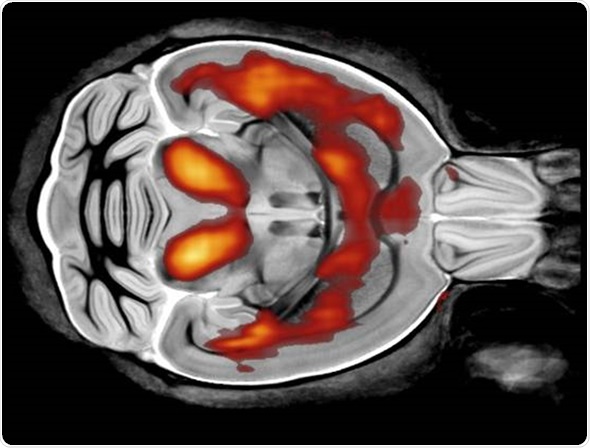
What are your thoughts on low-field systems?
When I first heard of these low-field systems, I was dubious as to how they would work and fit in, but I'm actually quite pleasantly surprised working with the ICON system and low-field systems.
Traditionally, people think that the SNR in a low-field system is much lower than in a high-field system and that, at best, there's a linear relationship between the two.
One would probably expect about ten times less SNR using a low-field system. However, there are ways of using the scanner to improve that SNR. We can adjust the scanning parameters, because at that particular field strength, the T1s and T2s literally take advantage of those parameters.
There is also a hardware aspect to it. You can have different geometry coils that actually beat the SNR, as opposed to in the high-field systems, where this is not possible due to the direction of the magnetic field in permanent magnet systems.
What are the main benefits of working with a desktop system?
You don't need three rooms to run the machine and the fringe field is within the actual machine itself, meaning you don't need to take out your keys.
In terms of health and safety, the desktop system is a lot easier to work with and is a very nice, compact system. You can just sit it in the corner of a lab and you don't really need specific equipment.
You also don't need to make too many changes to a building so that you can house an MRI scanner. Aside from the cost of actually buying the machine, a lot of the cost involved with a high-field system is actually that of changing the building to house the machine, whereas you don't need to do those sorts of things for the ICON.
The Center for Advanced Biomedical Imaging at UCL
What do you think the future holds?
That's a very interesting question. To be honest, I don't think that 1T low-field systems will completely replace the high-field systems. The high-field systems still beat on SNR, on time and so forth.
However, there is a place for the ICON, because it takes up much less space compared with a 9.4T system. For a 9.4T system or any cryogenically cored system, you need three big rooms. You need a scanner room, an equipment room (which has to have strict environmental controls) and a control room - you need a separate room to control the scanner.
It needs building services such as cold water supplies and if those go down, your scanner doesn't work. Also, you need a big air conditioner to keep all of the equipment cool and, again, if that goes down, the scanner doesn't work.
There are many environmental controls and building issues that you need to think about. All of this compares to the desktop system, which can simply sit in the corner of most biomedical labs. It doesn't require such stringent environmental controls, nor does it need a cold water supply. It can sit in with other modalities or it can sit in with other labs.
I think that's probably where the niche will be, in areas where you can't fit a big 9.4T scanner or the council costs are too high. Additionally, the running costs of the ICON are very low compared with the 9.4T scanner. These are all advantages for the ICON.
Image credit: Photoacoustic imaging group, UCL
You’re mainly working on developing the Francis Crick Institute at the moment. Please can you tell us more about this new centre?
The Crick Institute is a collaboration between six organizations: the Wellcome Trust, Cancer Research UK and the Medical Research Council and also three high quality educational establishments – UCL, Imperial, and King's College, London.
It's a big centre that is being built just behind the British Library in King's Cross. It will eventually employ about 1,500 scientists. The idea is to have a very open and collaborative environment where there will be lots of exchange of ideas between the various research groups that will be working there.
In addition, we want to bring in collaborations from the original founding partners - those six organizations, as well as from the wider academic community, industry and the public. Public engagement will be one of the key areas that the Crick will be hoping to pursue.
It is going to be very interdisciplinary. We'll be looking at cell work and preclinical work across a number of fields such as cancer research, cardiac research, neurodegenerative diseases and infectious diseases, to name just a few examples.