Sponsored Content by PittconJan 26 2016
Please can you give an overview of single-molecule optical nanoscience?
Single fluorescent molecules provide a local nanometer-sized probe of complex systems. We can measure the motion of the single molecule, use them to achieve imaging on a scale down to 20 nanometers, or we can infer aspects of the behavior of the object under study by the details of the light that is emitted.
Understanding Cellular Processes using Single Molecule Studies
Understanding Cellular Processes using Single Molecule Studies from AZoNetwork on Vimeo.
Why is this area particularly exciting to the molecular biology and biochemistry communities?
Because cells are filled with many tiny machines like enzymes, proteins, DNA, and RNA, which have their own jobs to do all at the same time, much would be missed if we watched them all at the same time. Observing individuals provides much more access to the molecular mechanisms at work.
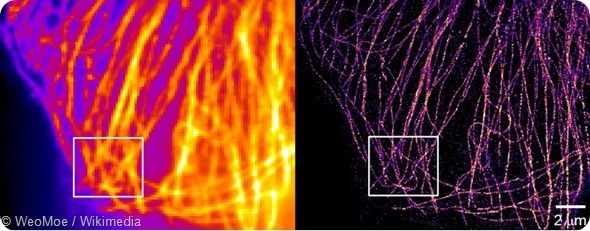
How do single-molecule studies allow the exploration of hidden heterogeneity in complex systems, such as biomolecules in cells?
When millions of molecules are measured at the same time, the measurement is termed an “ensemble average”, that is, the measurement is a mean over all the molecules. This single number obscures heterogeneity, which is the possibility that some have one value, and others have another value.
With single-molecule measurements, however, we can measure a quantity molecule by molecule, to see if they are all equivalent or not.
In what ways can single-molecule measurements provide new information?
This is a huge question. When single molecules are used for super-resolution imaging beyond the optical diffraction limit, then we can see structures that could not be observed before.
For example, we can see the shape of sub-diffraction-sized aggregates of amyloid proteins in a cell, or researchers can see previously unknown bands on actin filaments. When a single DNA locus, a single RNA molecule, or a single protein are observed, we can see their motion and determine if they are moving freely, restricted by some interaction, or if they are bound to another protein.
It was recently announced that you will be presenting the upcoming Wallace H. Coulter Lecture at Pittcon 2016. What will be the main focus of your lecture?
How Optical Single-Molecule Detection in Solids Led to Super-Resolution Nanoscopy in Cells and Beyond.
More than 25 years ago, single molecules were first detected optically in the Moerner lab at IBM Research in the course of explorations of fundamental limits on optical storage. Far from being only an esoteric effect at low temperatures, the attainment of this ultimate limit of sensitivity led to the ability to detect single molecules in solution, solids, crystals, and even cells; moreover, it became possible to control the single molecules as well.
Single emitting molecules are like tiny light sources which can used to light up an extended structure when combined with a way to control the concentration of molecules that are “on” at a given moment.
This strategy has recently enabled “super-resolution” imaging through the work of Eric Betzig and others, which means overcoming the resolution limit imposed by the wavelength of light of about ~200 nm in the visible. Now it is possible to record detail all the way down to 10’s of nm and below.
Essentially, images which were fundamentally blurry and out of focus are now sharp so that structures which could not be observed before are now visible. This has led to many advances in cell biology and microscopy by allowing structures and behavior to be seen for the first time, both in normal and in diseased cells.
What Pittcon Can do for You
What Pittcon Can do for You from AZoNetwork on Vimeo.
Please can you outline your involvement in the development of new photoswitchable single-molecule fluorophores? How will this enable further single-molecule imaging in cells?
In 1997, we found that single molecules of green fluorescent protein blink and can also be optically controlled as to when they would leave a dark state. We use the blinking and switching to achieve super-resolution, because these physical effects can be used to maintain the concentration of emitters at a low level so that a series of frames of a fluorescence movie allows the nanoscale location of the individual molecules to be determined.
In addition, small organic molecules are very useful for this kind of imaging because they can provide more photons before photobleaching. Working with synthetic collaborators, we have identified several fluorophores which can be “turned-on” by light, and others that can be turned on by an enzyme.
What do you think the future holds for single-molecule studies and cell biology?
There is so much more to learn about the behavior of individual proteins and oligonucleotides in cells, and so much to be done to improve the methods whereby we extract information from single molecules, particularly in the three-dimensional environment of the cell.
Colleagues Celebrate Nobel Laureate W.E. Moerner
Where can readers find more information?
There are several reviews at http://web.stanford.edu/group/moerner/sms_gen.html
About Professor W. E. Moerner
W. E. (William Esco) Moerner, the Harry S. Mosher Professor of Chemistry and Professor, by courtesy, of Applied Physics at Stanford University, conducts research in physical chemistry and chemical physics of single molecules, single-molecule biophysics, super-resolution imaging and tracking in cells, and trapping of single molecules in solution.
His interests span methods of precise quantitation of single molecule properties, to strategies for three-dimensional imaging and tracking of single molecules, to applications of single-molecule measurements to understand biological processes in cells, to observations of the photodynamics of single photosynthetic proteins and enzymes.
He has been elected Fellow/Member of the NAS, Royal Society of Chemistry, American Academy of Arts and Sciences, AAAS, ACS, APS, and OSA.
Major awards include the Earle K. Plyler Prize for Molecular Spectroscopy, the Irving Langmuir Prize in Chemical Physics, the Pittsburgh Spectroscopy Award, the Peter Debye Award in Physical Chemistry, the Wolf Prize in Chemistry, and the 2014 Nobel Prize in Chemistry.
About Pittcon
 Pittcon® is a registered trademark of The Pittsburgh Conference on Analytical Chemistry and Applied Spectroscopy, a Pennsylvania non-profit organization. Co-sponsored by the Spectroscopy Society of Pittsburgh and the Society for Analytical Chemists of Pittsburgh, Pittcon is the premier annual conference and exposition on laboratory science.
Pittcon® is a registered trademark of The Pittsburgh Conference on Analytical Chemistry and Applied Spectroscopy, a Pennsylvania non-profit organization. Co-sponsored by the Spectroscopy Society of Pittsburgh and the Society for Analytical Chemists of Pittsburgh, Pittcon is the premier annual conference and exposition on laboratory science.
Proceeds from Pittcon fund science education and outreach at all levels, kindergarten through adult. Pittcon donates more than a million dollars a year to provide financial and administrative support for various science outreach activities including science equipment grants, research grants, scholarships and internships for students, awards to teachers and professors, and grants to public science centers, libraries and museums.
Visit pittcon.org for more information.