Designing a protocol for trials on participants from an elderly population can be a challenging task due to the necessity to adapt to the specific needs of this population.
This study outlines the main difficulties faced when recruiting from an elderly population as well as detailing the primary considerations required to overcome when designing a conducting a clinical study in a food ingredient.
The sponsor was a clinical stage Health Ingredients company that develop food ingredients with clinically proven health benefits across a range of populations with varying health conditions.
Challenges and objectives
The product under investigation supported immune function and was believed to enhance resistance to ARTI. The goals of this study were:
- To test the effect of the product under investigation on volunteer’s resistance to ARTI evaluated using validated questionnaires.
- To assess the effect of the product on some secondary variables such as doctor/hospital visits or any infection that arises during the treatment period.
- To investigate the effect of the product on some exploratory variables such as inflammatory markers or immune cell activity.
In vitro studies as well as other clinical trials were performed. The sponsor required a trusted partner with the ability to develop and conduct a vigorous study as the ultimate step towards establishing a food ingredient for inclusion in dietary supplements, functional foods, and Foods for Specific Medical Purposes (FSMP).
The main hurdle was the recruitment of a sizeable population that showed ARTI symptoms during the winter session.
How Atlantia’s solution helped
Atlantia’s world-leading clinical research team worked in close alliance with the Sponsors’ researchers to develop and deliver the objectives of the study.
Atlantia possesses an extensive wealth of experience when performing clinical studies in elder populations, holding a large database of volunteers, in good compliance with the protocol requirements.
Their knowledge of GCP located Atlantia as the primary research site, making sure that additional sites met compliance with those standards.
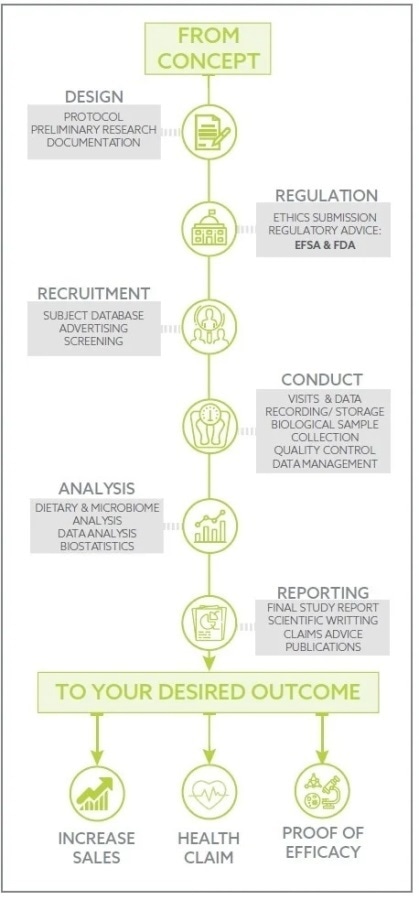
Image Credit: Atlantia Clinical Trials
About the study
This study was a double-blind, multi-center, placebo-controlled, randomized, parallel study. There were four visits throughout the 10 month duration of the study. The population was divided into three sets: two had two different treatment doses, low dose and high dose and the final one received a placebo.
Subjects meeting the eligibility criteria post-screening were randomized on a 1:1:1 basis to facilitate equal distribution of low dose, high dose, and placebo amongst the various geographical locations (Ireland and UK).
Other eligibility criteria included the intention to be vaccinated or having received vaccination for influenza, cohort of malnourished & risk of malnourishment, respiratory disease (COPD & Asthma) cohort, and various elderly age groups (65-69 years, 70-74 years, 75-79 years, 80-84 years and 85 years and over).
An interactive web response system utilizing a minimization protocol was used for randomization. The minimization protocol for randomization is a method that best maintains balance in prognostic factors by assigning subjects to the appropriate treatment group.
Even at small sample sizes, it is a suitable method and for those with numerous prognostic variables.
As well as the data points collected during the visits, Atlantia developed an app mimicking a daily diary to continuously collect data points.
The app recorded information related to the consumption of the product, medications, adverse events and doctor visits. It was also effective for tracking whether a subject had an ARTI by using the Jackson questionnaire.
If any of the subjects met certain criteria on the Jackson questionnaire, they were informed to complete the WURSS-24 and take their temperature until symptoms from the ARTI had subsided.
If a subject recorded an ARTI, an alert was sent to the site via the app, and a home visit was arranged where a health care professional carried out an examination and if a positive diagnosis of Acute Respiratory Tract Infection was delivered, certain procedures were followed.
In Atalantia’s main facility in Cork, 350 participants were successfully recruited. In total, 193 subjects per group completed the entire study (withdraw rate= 3.5%). In terms of PP and ITT populations from approximately 200 per group, all of them were assessed, but not all of them were assigned to the PP group.
Future plans and return on investment
The sponsor was extremely satisfied with Atlantia’s management of the trial, from initial development to how it was conducted. They identified the role of the company as Principal Site in this research, ensuring the sponsors’ best interest. The recruitment of participants also exceeded the expectations of the sponsor.
About Atlantia Clinical Trials
Atlantia Clinical Trials Ltd is a CRO that specializes in conducting studies in foods, beverages and supplements for companies world-wide that want to scientifically validate their functional ingredients to support an: EFSA (European Food Safety Authority) Health Claim; FDA (Food & Drug Administration) Structure Function Claim; or General Product Marketing Claim.
Atlantia works with world leading scientists (among the top cited 1% internationally, in the areas of digestive health and functional foods) at the: APC Microbiome Institute in University College Cork, Ireland; Teagasc, Moorepark, Ireland and recognized centers of excellence globally.
Atlantia runs and operates its own clinic sites and conducts all studies to ICH-GCP standard (International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use - Good Clinical Practice).
Its team includes physician experts in digestive health, mental health (psychological stress and cognition), cardiovascular health, sports performance, metabolic disease, bone health, immune health and healthy aging.
The clinical team also includes project managers, research nurses, nutritionists, certified sports trainers and lab researchers.
Atlantia manages all elements from protocol design, placebo manufacture, recruitment, and study execution, to sample and data analysis, statistics and report/dossier preparation to provide a service which is technically, scientifically and clinically superior.
The clinical studies cover a broad spectrum of functional food and beverage categories, such as dairy, cereal, probiotic, different protein forms, infant-specific foods, vitamins/minerals, plant or marine extracts and medical foods.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.