Oligonucleotide therapeutics have transformed the world of medicine over the last decade because of their ability to treat a wide range of indications, including previously untreatable targets that cause rare diseases. These oligonucleotides comprise a wide range of synthetically modified ribonucleic acid (RNA) or hybrids of RNA and deoxyribonucleic acid (DNA) that have been specially engineered to alter RNA and/or protein production by modifying a target RNA sequence.
As of March 2024, 20 oligonucleotide-based therapeutics had received commercial approval in the United States and the United Kingdom. This consists of one aptamer, 12 antisense oligonucleotides (ASOs), six small interfering ribonucleic acids (siRNAs), and a combination of single- and double-stranded polydeoxyribonucleotides. Oligonucleotides in development come in a variety of conjugated forms (e.g., GalNac, peptides, and lipids).
Challenges with oligonucleotide analysis
Reliable and robust bioanalytical procedures are critical for supporting preclinical and clinical studies on new oligonucleotides. Nonetheless, these compounds pose considerable analytical problems, including:
- High polarity and high negative charge, with molecular weights ranging from 5-7 kDa per strand. Non-specific binding and low sensitivity are among the method development obstacles, which are caused by large molecular weight, numerous charged species, and adduct formation
- Strong protein binding necessitates specialized procedures for optimal recovery. A popular strategy for extraction from biological matrix is to perform a liquid/liquid extraction (SLE) [AS1] with dichloromethane and a phenol/chloroform mixture. This is sometimes a complicated and difficult multi-step procedure
Case study: Analysis of a siRNA oligonucleotide in rat plasma
In the example below, Synexa illustrates how to obtain great recovery and repeatability while analyzing a siRNA oligonucleotide in rat plasma using the following technologies:
- Waters OligoWorks Kit
- Waters Acquity Premier C18 Oligonucleotide C18 column
- Waters iClass Acquity Premier UPLC with Xevo TQ-Absolute triple quad mass spectrometer
Background of the oligonucleotide
A siRNA having an antisense strand (AS) of 7354 Da and a sense strand (S) of 6185 Da. The mass spectrum on the TQ-Absolute revealed negative charged states of 5-10 for the AS strand and 6-11 for the S strand, with no indication of adduct formation.
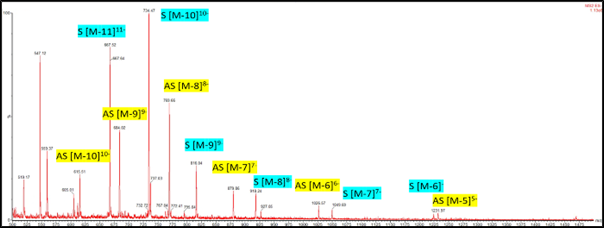
Image Credit: Synexa Life Sciences GmbH
System reproducibility
The UPLC system was configured with an Acquity Premier Oligonucleotide C18 column (1.7 µm, 2.1 × 50 mm). The aqueous and methanolic organic mobile phases both included HFIP, TEA, and EDTA. Mobile phases were made new every day.
Due to the system's unique "Premier" coating, which prevents possible interactions with metal ions, no passivation with strong acid was required prior to use. Multiple injections of an oligonucleotide solution provided instant reproducibility.
The table below depicts replicate injections of a non-extracted test sample with an internal standard (stable, labeled A strand). Peak area ratios had a precision of lt;2 % and peak areas had a precision of at least 3 %.
Source: Synexa Life Sciences GmbH
|
|
Peak Area |
IS Peak Area |
Area Ratio |
| Injection # |
1 |
107150 |
20689 |
5.179 |
|
2 |
105518 |
20743 |
5.087 |
|
3 |
103797 |
20254 |
5.125 |
|
4 |
108122 |
21527 |
5.023 |
|
Average |
106147 |
20803 |
5.103 |
|
S. Dev |
1900 |
530 |
0.066 |
|
CV % |
1.79 |
2.55 |
1.29 |
Optimized extraction from rat plasmaExtraction of the oligonucleotide from plasma proved difficult. Several extraction procedures were tested, including solid phase extraction (SPE) and LLE.[SD2] [AS3] Despite efforts to reduce non-specific binding during preparation, strong protein binding remained a significant issue, resulting in poor or uneven recovery.
Waters' introduction of the OligoWorks Kit provides an efficient solution to strong protein binding through the use of RapiZyme proteinase K digestion.
The kit includes all of the essential reagents as well as a mixed-mode SPE plate for a quick out-of-the-box solution with detergent-free digestion and an optimized SPE process with eluents that can be directly injected into the mass spectrometer.
By employing this protocol, remarkable recovery and reproducibility from rat plasma were attained.
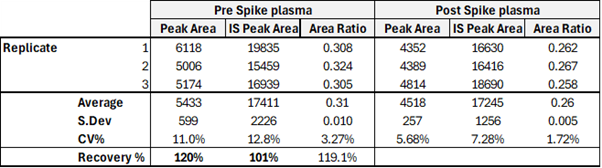
Recovery of sense-stranded siRNA from rat plasma after SPE OligoWorks extraction
Source: Synexa Life Sciences GmbH
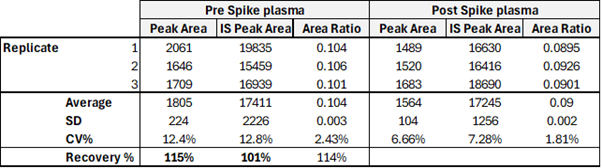
Recovery of antisense stranded siRNA from rat plasma after SPE OligoWorks extraction
Source: Synexa Life Sciences GmbH
Next steps in oligonucleotide bioanalysis
Building on these findings, Synexa wants to improve the oligonucleotide bioanalysis capabilities by validating the technique in accordance with M10 recommendations and achieving a lower limit of quantification (LLOQ) in the low-ng range for plasma samples.
To increase productivity, the Waters Andrew + Pipetting Robot will be used to automate and simplify extraction procedures. Furthermore, Synexa wants to broaden the use of the OligoWorks Kit for oligonucleotide extraction from diverse tissues.
Advanced oligonucleotide analysis at the GLP/GCP facility
Synexa operates two validated iClass Acquity systems at the GLP/GCP facility in Macclesfield, both equipped with TQ-Absolute mass spectrometers that enhance oligonucleotide analysis capabilities. This technique has high sensitivity in electrospray negative mode, reduces adduct formation, and eliminates the need for system passivation with acid, resulting in simplified operation.
The standardized procedures allow for quick method development at a minimal cost, covering a wide range of oligonucleotide treatments. Furthermore, crucial reagents may be obtained directly from the vendor, ensuring constant quality.
The approach is designed for maximum efficiency, with a quick 60-minute digestion followed by SPE direct injection into the LC-MS/MS instrument. This technique achieves high recovery with minimal matrix effects, improving data reliability and repeatability.
Why LC-MS/MS?
Compared to alternative methods, LC-MS/MS provides a simpler, quicker, and more economical approach for developing oligonucleotide assays. Method development durations are shortened, and costly, specialized reagents are not needed.
Adduct formation, limited sensitivity, and protein binding are only a few of the difficulties that have been overcome. Early in the drug development process, findings can be obtained rapidly, and turnaround times are short. It will be easy to switch to fully validated assays to assist regulatory PK studies.
References
- Vinjamuri, Pan amp; Peng. J Pharmaceutical Sciences. 2024 Jul: 113(7):1749-1768 The clinical potential of l-oligonucleotides: challenges and opportunities - PMC
- Liu, A., et al. (2022). Bioanalysis of Oligonucleotide by LC–MS: Effects of Ion Pairing Regents and Recent Advances in Ion-Pairing-Free Analytical Strategies. International Journal of Molecular Sciences, 23(24), p.15474. DOI: 10.3390/ijms232415474. https://www.mdpi.com/1422-0067/23/24/15474.
- Yuan, L., Dupuis, J.-F. and Mekhssian, K. (2023). A Novel Hybridization LC-MS/MS Methodology for Quantification of siRNA in Plasma, CSF and Tissue Samples. Molecules, (online) 28(4), p.1618. DOI: 10.3390/molecules28041618. https://www.mdpi.com/1420-3049/28/4/1618.
About Synexa Life Sciences GmbH
Synexa Life Sciences is a biomarker and bioanalytical lab CRO, specializing in the development, validation and delivery of a wide range of complex and custom-designed assays.
With a team of over 200 staff across three global laboratory locations; Manchester, Turku (Finland) and Cape Town, we provide innovative solutions to support our customers to achieve their clinical milestones.
Our main areas of expertise include biomarker identification and development, large and small molecule clinical bioanalysis, (soluble) biomarker analysis (utilizing MSD, LC-MS/MS, ELISA, RIA, fluorescence and luminescence-based technologies), cell biology (including flow cytometry, ELISpot and Fluorospot) and genomic services to support clinical trials and translational studies.
We pride ourselves on our deep scientific expertise and ability to tackle complex problems, translating them into robust and reliable assays to support clinical trial sample analysis.
Since 2019, Synexa has been backed by Gilde Healthcare, a specialized healthcare investor. Synexa, improving the quality of human health through innovative biomarker and bioanalytical solutions.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.