This article is based on a poster originally authored by Yuhui Wang, Chuyue Yu, Qianqian Li, Mingshuo Zhang, Junwen Zhang, Jiawen Xu, Xuan Liu, Xiaolan Huang, Qiang Gao, Pengwei Pan, and Fang He.
Antibody-drug conjugates (ADCs) are potent cancer treatments that selectively deliver lethal payloads to tumors.
In vitro efficacy testing with organoids is central to the identification of new ADCs, as they can mimic the 3D structure of tumors and provide a more accurate model for examining ADCs' complex mechanism of action.
This study introduces an organoid-based ADC testing platform ideal for high-throughput screening. The researchers show its use in predicting ADC efficacy and safety using a case study focusing on human epidermal growth factor receptor 2 (HER2).
Methods
- Established over 150 PDX-derived organoid (PDXO) models, including cancer and normal organoids.
- HER2 expression was assessed by RNAseq, WES, Western Blot, and IHC staining.
- Cell viability was assessed in 14 PDXO models with high, moderate, or low HER2 expression levels to evaluate responses to four HER2-targeted ADC medicines.
- Trastuzumab deruxtecan (T-DXd), MedChemExpress, HY-138298A
- Trastuzumab emtansine (T-DM1), MedChemExpress, HY-P9921
- Trastuzumab duocarmazine (T-Duo), MedChemExpress, HY-P99547
- Trastuzumab monomethyl auristatin E (T-MMAE), MedChemExpress, HY-164992
- Safety tests were conducted on these medications using human intestine and colonic organoids (HIO/HCO) generated from normal tissues.
Diverse organoid model library for ADC drug R&D
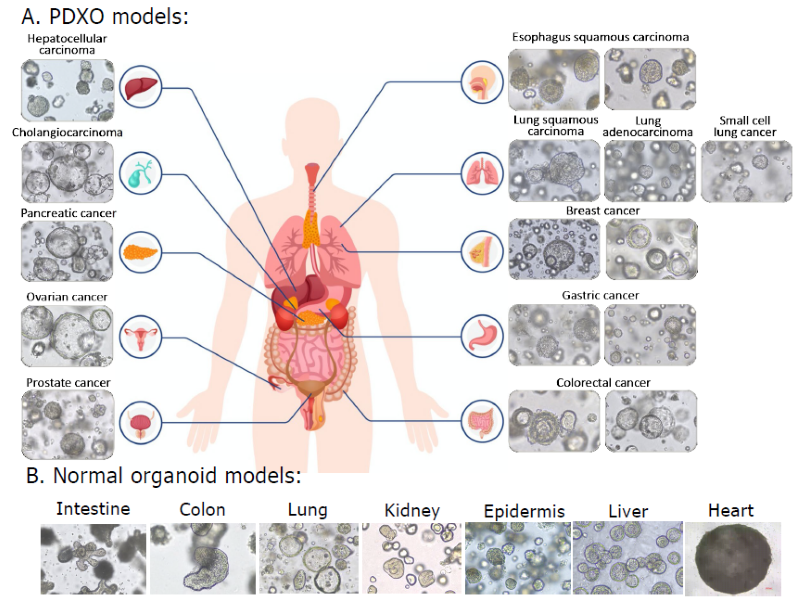
Organoid models. Image Credit: Pharmaron
Pharmaron's organoid collection has over 150 PDXO models generated from main tumor types (A) and several normal organoid models representing multiple organs (B). The biobank is constantly expanding with new elements.
Multifaceted characterization of PDXO models reveals HER2 expression heterogeneity
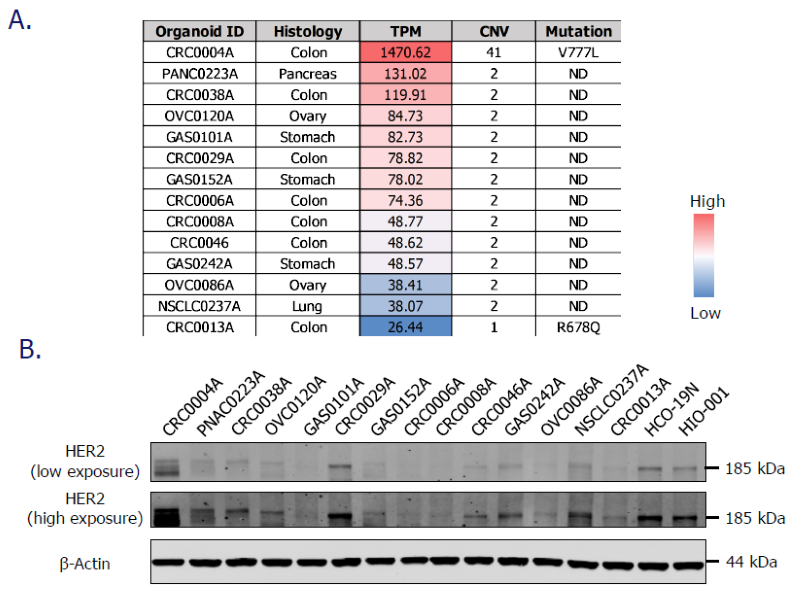
Assessment of HER2 expression status in PDXOs. Image Credit: Pharmaron
A. Analysis of transcriptional levels (transcripts per kilobase million, TPM), copy number variation (CNV), and point mutations of ERBB2 (HER2 coding gene) in PDXOs using RNAseq and WES data. The list is sorted by TPM values, from high (red) to low (blue).
B. The protein level of HER2 in PDXOs was measured using a Western Blot test.
PDXOs possess histopathological consistency with the original PDX tissues
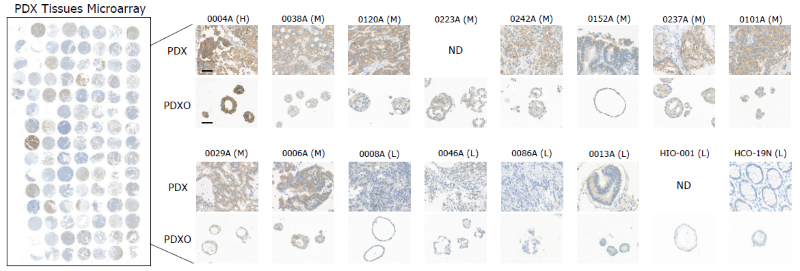
Immunohistochemical staining of HER2 in PDX and PDXOs. Image Credit: Pharmaron
The IHC staining pattern revealed a significant level of HER2 expression between PDXOs and their associated PDX tissues. Scale bar: 50 μm. H denotes high, M indicates moderate, L indicates low, and ND indicates not detected.
Organoids are good translational models for evaluating ADC efficacy and toxicity
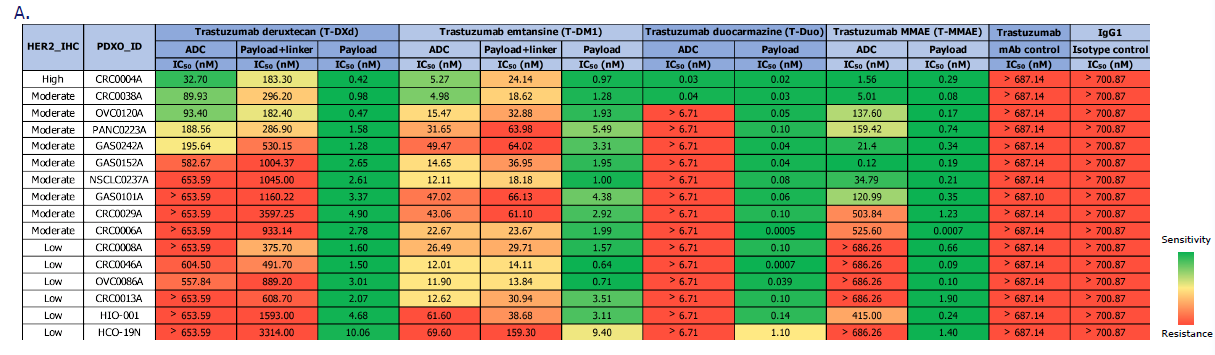
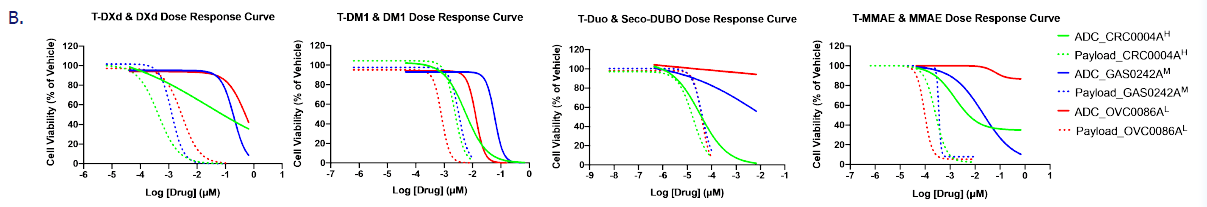
ADC drug screening results in organoid models. Image Credit: Pharmaron
A. CellTiter-Glo test to assess the cytotoxicity of four HER2-targeting ADCs and their payloads in PDXOs and standard HIO/HCO models.
B. Cell viability curves for CRC0004A (HER2High, green lines), GAS0242A (HER2moderate, blue lines), and OVC0086A (HER2low, red lines) for four ADCs (solid lines) and payloads (dashed lines).
Conclusion
- Model Sensitivity and ADC Specificity: The ADC screening demonstrated that organoids exhibited varied sensitivity to both HER2 expression levels and different ADC medicines. Typically, high (or moderate) HER2 expression was associated with enhanced sensitivity to ADC therapy, whereas low expression indicated resistance. In contrast, the payloads exhibited high, unbiased cytotoxic action against all animals.
- Therapeutic Window: Normal intestinal organoid models were insensitive to ADC treatment, indicating a low risk of GI side effects and a favorable therapeutic window.
- Summary: The organoid-based ADC screening platform provides a quick and dependable approach to early high-throughput screening, pharmacology, and efficacy research, bridging the gap between in vitro and in vivo studies.
Yuhui Wang1, Chuyue Yu1, Qianqian Li1, Mingshuo Zhang1, Junwen Zhang1, Jiawen Xu1, Xuan Liu1, Xiaolan Huang1, Qiang Gao1, Pengwei Pan1, Fang He*
1in vitro Biology, Pharmaron (Beijing) Co., Ltd, Beijing, China
*Corresponding author: [email protected]
About Pharmaron
Pharmaron is a premier R&D service provider for the life sciences industry. Founded in 2004, Pharmaron has invested in its people and facilities and established a broad spectrum of research, development, and manufacturing service capabilities throughout the entire drug discovery, preclinical, and clinical development process across multiple therapeutic modalities, including small molecules, biologics, and CGT products. With over 17,000 employees and operations in China, the U.S., and the U.K., Pharmaron has an excellent track record of delivering R&D solutions to its partners in North America, Europe, Japan, and China.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.
Last Updated: Mar 19, 2026