This article is based on a poster originally authored by Yinjie Li, Ce Zhong, Yufang Yan, Hong Liu, Zhanshan Chen, Jing Ma, Tong Wang, Ruixiu Wang, Yarou Pu, and Jingqi Huang.
Transgenic mouse models are important tools for understanding Alzheimer's disease (AD) pathophysiology and promoting treatment development. Selecting the appropriate AD mouse model is critical for accurately representing disease progression.
This work examined the pathogenic course of two traditional Aβ-driven models, APP/PS1 and 5xFAD, using behavioral assessment, histopathology, and genetic analysis.
Methods
- The researchers employed APP/PS1 mice (8-20 months) and 5xFAD mice (6-12 months) to study age-related pathological and molecular alterations in Alzheimer's disease models.
- The Morris Water Maze was used to measure spatial learning and memory deficiencies.
- Immunohistochemistry (IHC) of cortical and hippocampal tissues assessed Aβ deposition.
- Meso Scale Discovery (MSD) immunoassays were used to measure soluble and insoluble Aβ42 levels in brain homogenates, assessing age-related accumulation kinetics.
- Transcriptomic profiling was used to uncover model-specific pathway changes throughout development.
Age-related temporal patterns of cognitive deficits in APP/PS1 versus 5xFAD mice
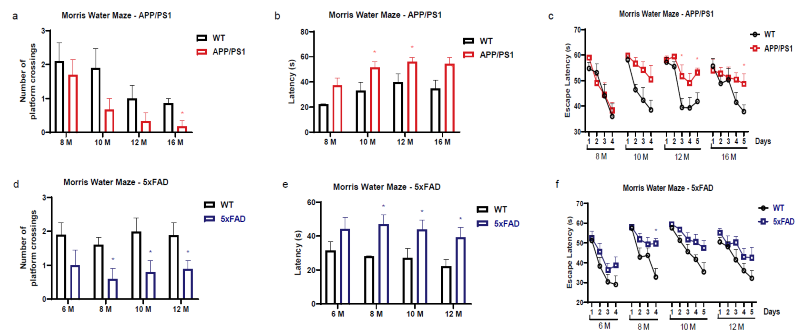
Spatial Learning and Memory Deficits in APP/PS1 and 5xFAD Mice Revealed by the MWM. Image Credit: Pharmaron
Performance of APP/PS1 and 5xFAD mice in the MWM task from 8 to 16 months of age, depicting (a, d) platform crossings, (b, e) escape latencies, and (c, f) learning curves over training periods. Data is reported as mean ± SEM for n = 6-10.
Differential age-related Aβ plaque accumulation progression in APP/PS1 and 5xFAD mice
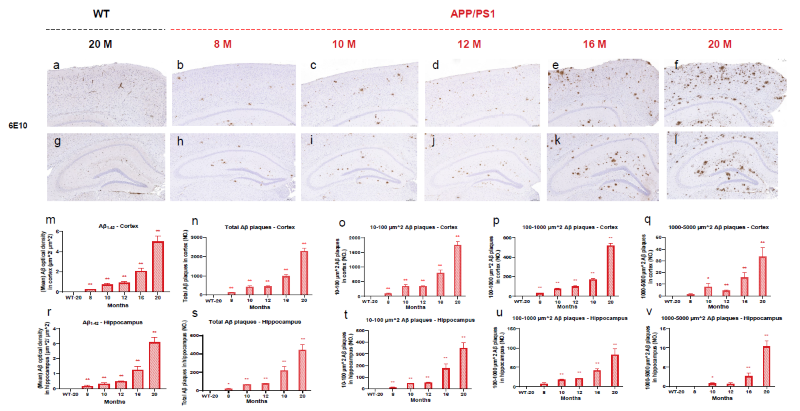
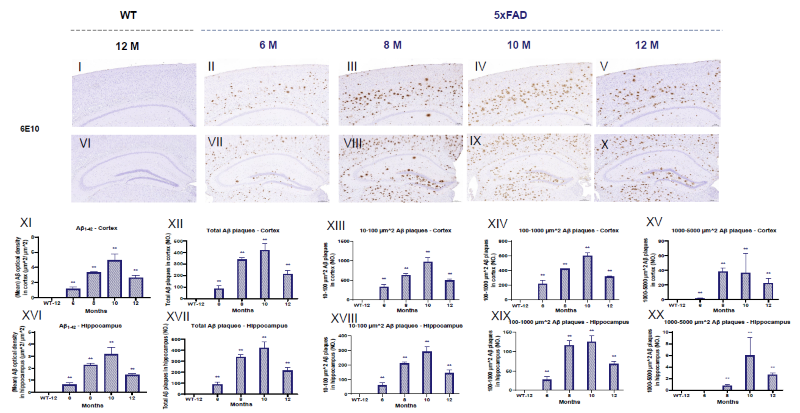
Cortex and Hippocampal Aβ Plaque Load in APP/PS1 and 5xFAD Mice via IHC. Image Credit: Pharmaron
Images of Aβ load in the cortex (a-f, I-V) and hippocampus (g-l, VI-X) of APP/PS1 and 5xFAD mice. Aβ plaques were quantified in the cortex (m, XI) and hippocampus (r, XVI) using optical density in μm2.
Stereological investigations show varying sizes (μm2) of Aβ plaques in the cortex (n-q, XII-XV) and hippocampus (s-v, XVII-XX) at different ages. The scale bar for the cortex is 100 μm. Scale bar for hippocampus = 200 μm. Data is presented as mean ± SEM (n = 3).
Differential soluble and insoluble levels of Aβ42 in cortex and hippocampus in APP/PS1 and 5xFAD
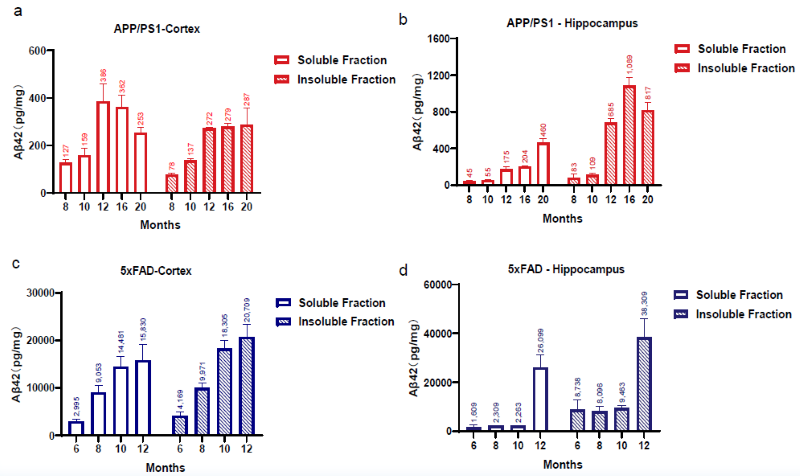
Quantification of Soluble and Insoluble Aβ42 in the Cortex and Hippocampus of APP/PS1 and 5xFAD Mice by MSD Assay. Image Credit: Pharmaron
Aβ42 levels in the cortex (a, c) and hippocampus (b, d) vary with age. Data is presented as mean ± SEM (n = 3).
Dynamic gene expression profiling in APP/PS1 and 5xFAD mice across four-stage aging
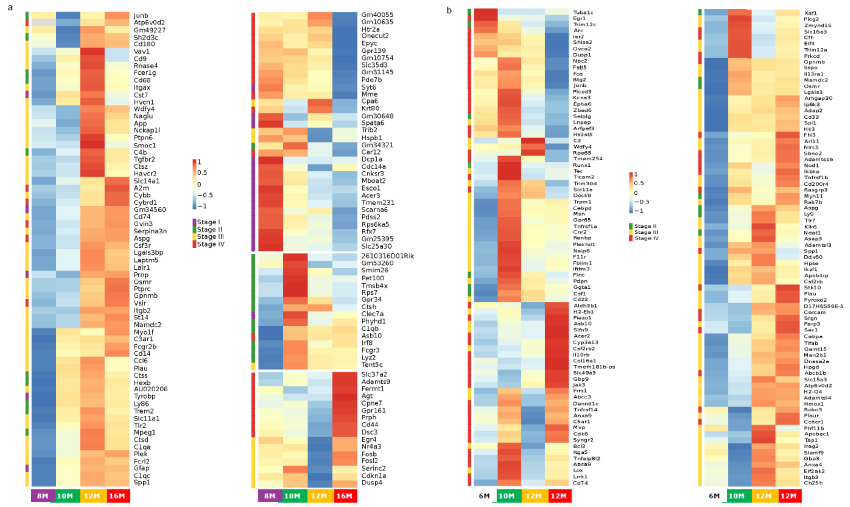
Heatmap of gene expression during progression of APP/PS1 and 5xFAD models. Image Credit: Pharmaron
Genes with significant differential expressions were clustered and classified into four stages according to the earliest time point of differential expression in (a) APP/PS1 and (b) 5xFAD mice. Each stage represents a single time point in cortical collecting.
Due to image size limits, genes from 5xFAD models were not presented in stage I. The Gamma-Poisson distribution was used to identify differentially expressed genes (DEGs). The fold change and adjusted p-value for DEGs were set to 2 and 0.05, respectively. n = 3.
Conclusion
- The findings demonstrate the usefulness of these Alzheimer's mouse models in various contexts.
- APP/PS1 mice are ideal for researching cognitive decline and the delayed formation of amyloid-beta (Aβ) plaques. This makes them useful for studying long-term mechanisms of illness.
- 5xFAD mice have severe memory and learning deficits due to Aβ plaque accumulation. These mice are ideal for early-stage research and high-throughput drug screening for Aβ buildup.
- Understanding these distinctions allows researchers to select Alzheimer's mouse models based on their research and treatment goals, improving the efficiency and relevance of preclinical drug development for the disease.
Yinjie Li, Ce Zhong, Yufang Yan, Hong Liu, Zhanshan Chen, Jing Ma, Tong Wang, Ruixiu Wang, Yarou Pu, Jingqi Huang*
*Corresponding author: Jingqi Huang, SVP Pharmacology; IACUC Chair, Pharmaron, Beijing, China
About Pharmaron
Pharmaron is a premier R&D service provider for the life sciences industry. Founded in 2004, Pharmaron has invested in its people and facilities and established a broad spectrum of research, development, and manufacturing service capabilities throughout the entire drug discovery, preclinical, and clinical development process across multiple therapeutic modalities, including small molecules, biologics, and CGT products. With over 17,000 employees and operations in China, the U.S., and the U.K., Pharmaron has an excellent track record of delivering R&D solutions to its partners in North America, Europe, Japan, and China.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.
Last Updated: Mar 19, 2026