Super-resolution (SR) fluorescence microscopy encompasses a variety of techniques that can exceed the diffraction limit – the point at which molecular nanostructures can be distinguished from one another.
Prior to the development of SR microscopy techniques, conventional wide-field microscopy was unable to distinguish nanostructures that were smaller than 200 nm. This was because they were unable to go beyond the diffraction limit. Through the use of SR, highly-resolved, dynamic imaging was able to be produced, enabling the interactions between biomolecular structures and cellular architecture to be observed in a way that was previously unachievable.
An introduction to super-resolution microscopy of living cells
SR techniques rely on the switching on and off of fluorescent markers. With conventional wide-field microscopy all markers are switched on at the same time, causing overlapping and blurred imaging to be produced. By being able to control the photoswitching of molecular markers, SR techniques can produce super-resolved images.
A variety of SR techniques exist but can be divided into two categories:
- Ensemble techniques
- Single molecule-based techniques
While single molecule-based techniques rely on the localization of individual molecules, ensemble techniques rely on the response of emitters to excitation.
Viewing super-resolution cells in real time
Ensemble Techniques
Stimulated Emission Depletion (STED)
Developed by the 2014 Chemistry Nobel Prize winner, Stephen Hell, the STED technique makes use of two diffraction patterns; one to excite emitting molecules and one to de-excite these emitters. The technique requires two lasers to do this. STED has a similar setup to that of a confocal microscope.
In comparison to other microscopy techniques, STED is able to produce video-rate imaging and works well with a variety of markers. STED has even been used to achieve less than 10 nm resolution in studies to observe fluorescent markers in diamond.
However, an issue that arises with the use of STED is that photobleaching may occur. By and large, higher resolutions are associated with the occurrence of more photobleaching. This inevitably makes it more challenging to garner repeat measurements.
Structured Illumination Microscopy (SIM)
An adapted wide-field microscope is used in the SIM technique to excite molecules within a sample. The process of excitation in SIM occurs using pattern illumination (typically a striped pattern is used.). Due to the interaction between the sample and the excitation pattern, images with moiré fringes (large scale interference patterns) are produced. These images can be processed and reconstructed to produce a high-resolved final image. In comparison to wide-field microscopy, the resulting image has a two-fold improvement in resolution.
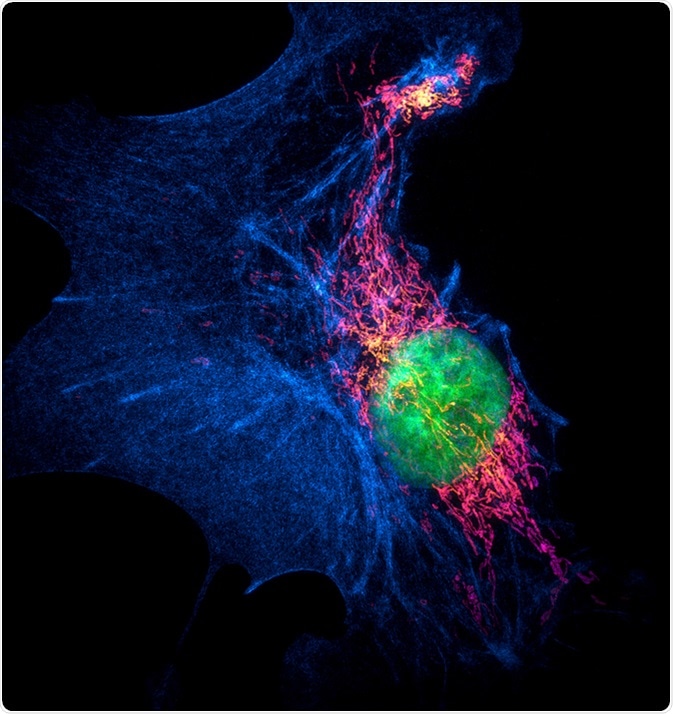
Structured illumination microscopy (SIM) images of a bovine pulmonary artery endothelial cell stained with fluorescent dyes for mitochondria, actin, nucleus. Maximum intensity projection of a z-stack. Image Credit: Micha Weber / Shutterstock
Single Molecule-Based Techniques
Photoactivated Localized Microscopy (PALM)
PALM is an SR which uses fluorescent proteins as markers in order to allow the molecular architecture of various cellular organisms to be resolved. The technique requires thousands of frames to be compiled to create a super-resolved image.
The imaging process begins by activating individual molecules using ultraviolet light, enabling them to fluoresce. The activated molecules are mapped and ultimately bleached by the ultraviolet light. The process is repeated until all the unique coordinates of all the molecules within a group are determined. This results in a high spatial resolution but a low temporal resolution as the process is generally slow.
PALM has been enhanced to allow 3D, multicolor super-resolved imaging to be produced. Biomolecular interactions and the structural details of cellular organisms have been observed using this technique.
Stochastic Optical Reconstruction Microscopy (STORM)
STORM is a highly regarded SR technique, it enables high resolution images to be produced using simple optical instrumentation. The setup that STORM is based on is the total internal reflectance fluorescence (TIRF) system. This is advantageous as TIRF is commercially available and economical.
The technique makes use of photoswitchable molecules, transitioning them between non-fluorescent and fluorescent states. The transition process operates in a random manner whereas in other SR techniques, like STED, they are more controlled.
Like PALM, STORM has the capability to produce, multicolor and 3D imaging.
The Future of SR Microscopy
The full potential of SR microscopy is yet to be reached. The field is relatively new, and the applications of these techniques have not been fully explored. With further improvements and advancements, SR will surely have a massive impact on the future of biomedical research.
Further Reading
Last Updated: Aug 23, 2018