Sponsored Content by PittconSep 12 2017
An interview with Nobel Prize Laureate Dr. Stefan W. Hell, Director at the Max Planck Institute for Biophysical Chemistry in Göttingen and Director at the Max Planck Institute for Medical Research in Heidelberg, conducted by April Cashin-Garbutt, MA (Cantab)
It was recently announced that you will be presenting the Plenary Lecture at Pittcon 2018. What will be the main focus of your lecture?
I will talk about the simple but fundamental idea that allowed breaking the diffraction barrier in fluorescence microscopy as well as about MINFLUX nanoscopy, the latest development in the field that for the first time provides true molecular resolution with visible light and standard objective lenses.
Stefan Hell on nanometer-scale microscopy in biophotonics
What was the key to breaking the diffraction barrier in a fluorescent life science microscope? How was the resolution-limiting role of diffraction overcome?
In the 20th century, light microscopes relied solely on focusing light in space for the separation (=resolution) of adjacent tiny features. Either they focused the illumination light as sharply as possible on the sample and/or the fluorescence light as sharply as possible on the detector.
As one cannot focus the light more sharply than to an extent given by diffraction, the resolution was limited to 200 nanometers. The key idea was to separate fluorescent features or molecules not by focusing but by transiently turning their fluorescence on and off so that features residing closer together than 200 nm could be distinguished by their sequential emission.
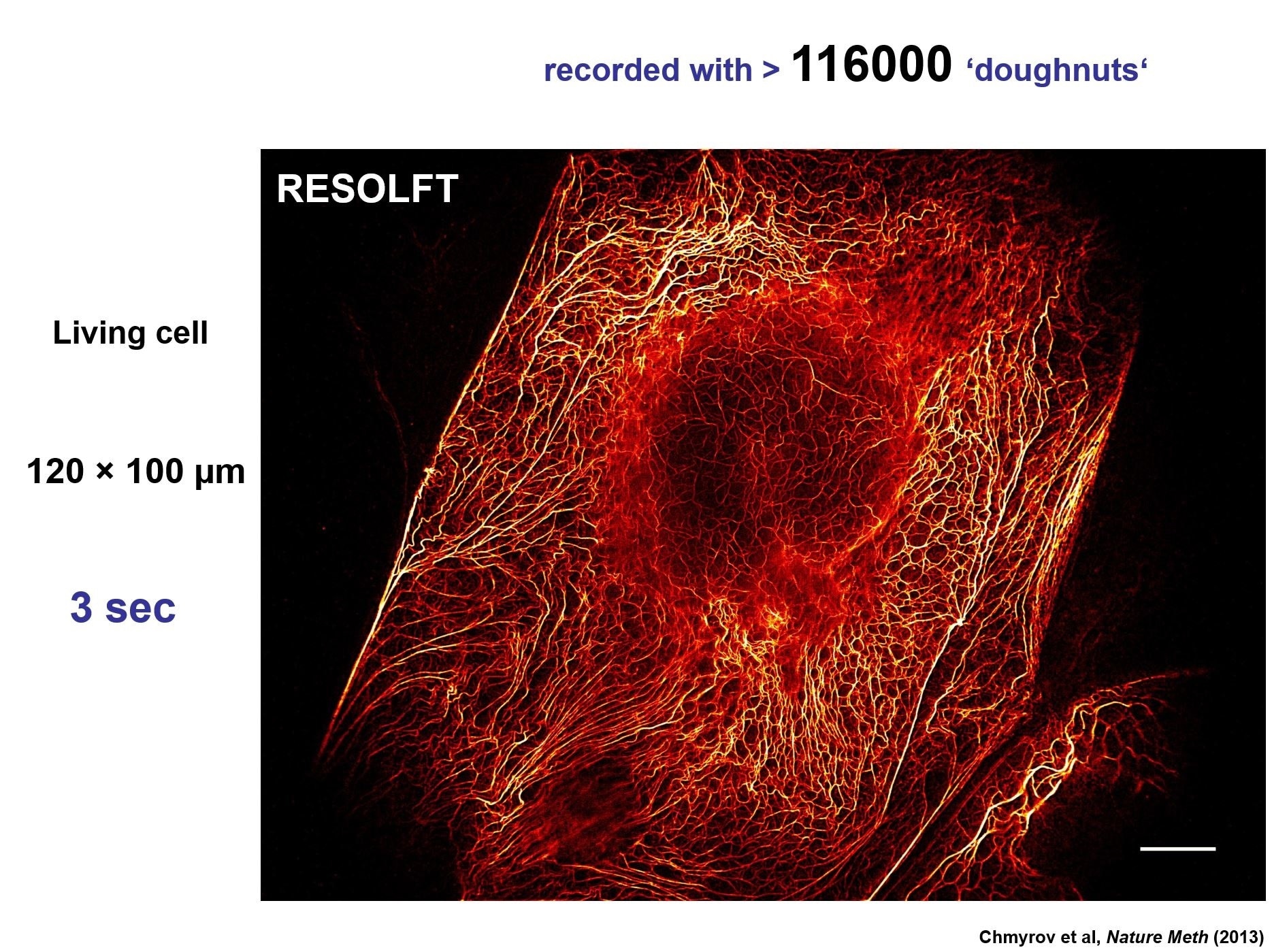 Credit: Chmyrov, A., et al. (2013). Nanoscopy with more than 100,000'doughnuts'. Nature methods, 10(8), 737-740. MPI Biophysical Chemistry, Göttingen, Germany.
Credit: Chmyrov, A., et al. (2013). Nanoscopy with more than 100,000'doughnuts'. Nature methods, 10(8), 737-740. MPI Biophysical Chemistry, Göttingen, Germany.
What impact has the ability to image the interior of transparent samples, such as living cells and tissues, at the nanoscale had on life sciences?
Scores of scientific studies in neurobiology, cell biology, and in many other areas of science have been carried out with fluorescence nanoscopy alias superresolution microscopy.
As the microscopes are becoming compact, easy-to-use, and less expensive, virtually all life science laboratories around the world will have to take advantage of the superior resolution in order to keep up in their respective fields.
In my view, every (single) beam scanning confocal microscope should have a STED option. Alternatively, any epifluorescence microscope can be easily upgraded to a confocal STED system.
Breaking the Diffraction Barrier
Breaking the Diffraction Barrier from AZoNetwork on Vimeo.
What improvements in instrumentation would you like to see in the future?
Instrumentation will become rugged, reliable, and economic. As a matter of fact, the latest versions of STED microscopy provide state-of-the-art multicolor resolution < 30 nm with a compact module smaller than the size of shoe box. The price tag is less than a fifth of early systems. Moreover, the module fits onto literally any modern epifluorescence microscope, no matter if upright or inverted.
Have we reached the absolute limit yet? Do you think it will be possible to see features of the molecules, such as symmetries?
The latest edition of fluorescence nanoscopy, MINFLUX, has indeed attained molecular scale (~1 nm) spatial resolution. This is the ultimate limit, because in fluorescence microscopy the ultimate resolution limit is ultimately given by the fluorescent tags themselves, which act as proxies for the biomolecules to be seen.
Yes, I can imagine that one should see molecular symmetries using STED and related techniques, however, that will be an entirely different area of application.
What are the main challenges that still need to be overcome?
Given that MINFLUX has attained the ultimate limit, I believe that increasing the imaging speed, i.e. minimize the time to resolve molecules with highest resolution will be a major research thrust.
Other aspects will be sample compatibility and the quest for improved labelling techniques. Labels need to be small and minimally disturbing. Lots of progress has been made in these fields, but clearly more work needs to be done.
How do you think the imaging speed can be improved?
Concepts that require fewer emitted photons to attain the same resolution, such as MINFLUX, can be optimized further in order to attain maximum i.e. true molecular (1 nm) resolution with a minimal number of detected fluorescence photons, say < 30 detection events. Such an effective use of fluorescence photons will speed up nanoscale imaging enormously.
What impact do you think super resolution microscopy will have on neuroscience?
Just imagine a microscope that provides molecular scale resolution with minimal invasion. I think the impact of such a tool will be enormous. For example, we should be able to fully unravel the distribution of proteins at the synapse and also see many of the relevant molecules in action.
Where can readers find more information?
The most recent review of fluorescence nanoscopy applications is by S.Sahl, S.W. Hell, S. Jakobs in Nat Rev Mol Cell Bio (2017).
Information about the basic principles of fluorescence nanoscopy (superresolution) are described in my Nobel lecture: "Nanoscopy with focused Light (Nobel Lecture)" Angew. Chem. Int. Ed. 54, 8054-8066 (2015)
About Dr. Stefan W. Hell
Stefan W. Hell is a physicist recognized for his pioneering research in far-field optical nanoscopy, also known as super-resolution microscopy.
After studies in Heidelberg (PhD in 1990) and postdoctoral work at the European Molecular Biology Laboratory, Hell laid out the principle of STED microscopy while on a research fellowship in Turku, Finland (1994).
The underlying idea, namely of discerning molecules at subdiffraction length scales by transiently preparing a subset of them in a non-signaling state, underlies all the practical diffraction-unlimited super-resolution fluorescence microscopy concepts to date.
For these achievements, Hell has received numerous awards. In 2014 he shared the Kavli Prize in Nanoscience and the Nobel Prize in Chemistry. Hell is a director at the Max Planck Institute for Biophysical Chemistry in Göttingen, and at the Max Planck Institute for Medical Research in Heidelberg (both Germany).
About Pittcon
 Pittcon® is a registered trademark of The Pittsburgh Conference on Analytical Chemistry and Applied Spectroscopy, a Pennsylvania non-profit organization. Co-sponsored by the Spectroscopy Society of Pittsburgh and the Society for Analytical Chemists of Pittsburgh, Pittcon is the premier annual conference and exposition on laboratory science.
Pittcon® is a registered trademark of The Pittsburgh Conference on Analytical Chemistry and Applied Spectroscopy, a Pennsylvania non-profit organization. Co-sponsored by the Spectroscopy Society of Pittsburgh and the Society for Analytical Chemists of Pittsburgh, Pittcon is the premier annual conference and exposition on laboratory science.
Proceeds from Pittcon fund science education and outreach at all levels, kindergarten through adult. Pittcon donates more than a million dollars a year to provide financial and administrative support for various science outreach activities including science equipment grants, research grants, scholarships and internships for students, awards to teachers and professors, and grants to public science centers, libraries and museums.
Visit pittcon.org for more information.