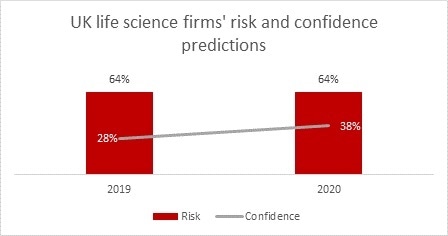
Brexit brings some serious challenges for UK life science businesses, most notably in terms of the regulation and funding of clinical trials, adding barriers to investment in the UK and threatening access to the latest medicines. However, the dominance of Britain as a life science destination in Europe means well over a third of sector leaders remain confident in the ability of their business to grow and prosper as they look ahead to 2020.
CNA Hardy Risk and Confidence research – May ‘19
The two risks which are growing fastest as UK life science leaders look to 2020 are economic, which is up 29%, and supply chain, which rises 17% as we look ahead to 2020.
Perhaps to a greater degree than many other sectors, life science faces particular economic risk exposure – changing government policy, pricing pressures, expanding demand for access to healthcare and to therapies, emerging digital and analytical capabilities are combining to push companies to explore new business models and new ways of working with customers – public and private.
Supply chain is also a fast-growing concern. The potential for divergent regulatory environments between the UK and the EU could also establish a non-tariff barrier requiring additional time, effort and therefore cost to resolve. The impact is likely to be significant on those operating with low-margin products (e.g. generics), but also on higher end pharma and medical device companies managing complex materials and technology flows expected to be particularly impacted.
Business leaders clearly believe the problems are surmountable with more feeling confident as they look ahead to 2020 than in 2019. But while it is possible to manage and mitigate supply chain risk, economic risk is a broader and more complex category of risk entirely. Insurers and brokers will need to pull out all the stops to support companies to manage their enhanced risk profile during this difficult period.