The field of neurological disease research is rapidly evolving, with protein biomarkers advancing our understanding, improving diagnosis, and guiding treatment for disorders such as Alzheimer's disease (AD), Parkinson's disease (PD), and multiple sclerosis.
While cutting-edge proteomics technologies have enabled major advances in the field, there are still obstacles to turning discoveries into therapeutic applications. Bridging these gaps requires sharing significant lessons from both successes and failures as the profession moves toward precision medicine.
In an effort to contribute to this information exchange, this article presents key ideas from a recent panel discussion that brought together renowned academic and pharmaceutical specialists, covering areas such as:
- Identifying and utilizing biomarkers to diagnose and treat neurological disorders.
- Challenges, opportunities, and practical solutions for incorporating proteomics platforms into clinical research, including sample management and data interpretation.
- Collaborations and partnerships among academics, industry, and clinicians are necessary to advance.
- Future directions of proteomics and multi-omics in driving precision medicine in neurological disease studies.
State of the field: Neurological protein biomarker research
Neurodegenerative disorders
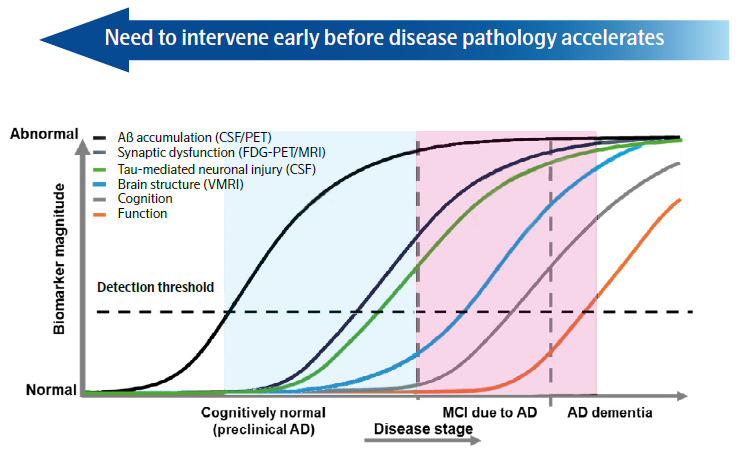
Figure 1. Graph representing biomarker magnitude across the AD disease progression continuum. Image Credit: Adapted from Sperling et al. Alzheimer’s Dement., 2011
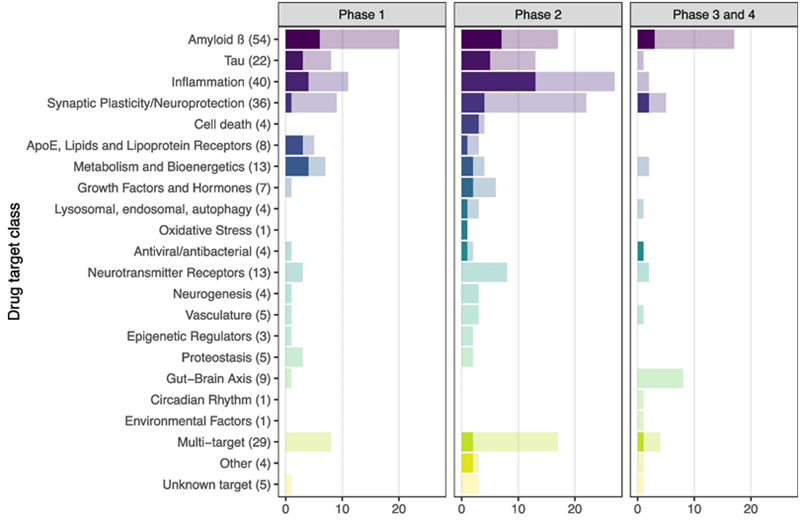
Figure 2. Number of trials with and without a fluid-based target engagement marker, by target class. Total number of trials (n= 272) was included in this analysis, darker shade indicates use of a fluid-based target engagement marker. Between brackets the total number of trials in that target class is listed. Image Credit: Oosthoek, Vermunt et al. Alzheimer Research & Therapy, 2024
Table 1. Intended uses for imaging, CSF, and plasma biomarker assays. 2024 AA revised criteria has expanded fluid biomarkers for diagnosis and staging of AD. Source: Jack CR Jr, et al. Alzheimer’s Dement., 2024
| Intended use |
CSF |
Plasma |
Imaging |
| Diagnosis |
| A: (Aβ proteinopathy) |
– |
– |
Amyloid PET |
| T1: (phosphorylated and secreted AD tau) |
– |
p-tau217 |
– |
| Hybrid ratios |
p-tau181/Aβ42, t-tau/Aβ42, Aβ42/40 |
%p-tau217 |
– |
| Staging, prognosis, as an indicator of biological treatment effect |
| A: (Aβ proteinopathy) |
– |
– |
Amyloid PET |
| T1: (phosphorylated and secreted AD tau) |
– |
|
– |
| Hybrid ratios |
p-tau 181/Aβ42, t-tau/Aβ42, Aβ42/40 |
%p-tau217 |
– |
| T2: (AD tau proteinopathy) |
MTBR-tau243, other p-tau forms (e.g., p-tau205), nonphosphorylated mid-region tau fragments |
MTBR-tau243, other p-tau forms (e.g., p-tau205) |
Tau PET |
| N (injury, dysfunction, or degeneration of neuropil) |
NfL |
NfL |
Anatomic MRI, FDG PET |
| I (inflammation) Astrocytic activation |
GFAP |
GFAP |
– |
| Identification of copathology |
| N (injury, dysfunction, or degeneration of neuropil) |
NfL |
NfL |
Anatomic MRI, FDG PET |
| V vascular brain injury |
– |
– |
Infarction on MRI or CT, WMH |
| S a-synuclein |
aSyn-SAA |
|
|
Molecular subtyping – enabled by platforms such as the Olink Explore HT panel – has begun to reveal biologically meaningful subgroups within AD. These insights may help explain clinical variability and guide more targeted therapeutic strategies.
Pallavi Sachdev, MPH, PhD Executive Director and Head, Translational Medicine, Eisai Inc
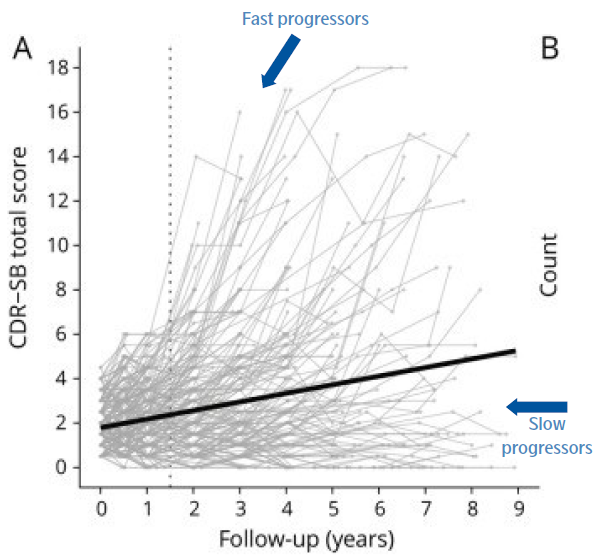
Figure 3. Individual trajectories show the heterogeneity of cognitive decline for an early AD cohort of 302 individuals, selected from the Alzheimer's Disease Neuroimaging Initiative database (ADNI). Clinical Dementia Rating scale–sum of boxes: the Alzheimer's Disease Assessment Scale–cognitive subscale (ADAS-Cog). The dotted vertical line presents scores at the 18-month time point. Image Credit: Adapted figure from Jutten RJ et al. Neurology, 2021
Multiple sclerosis
To address the biological complexity of multiple sclerosis, multiplex protein panels are being developed to capture the multifaceted nature of disease activity and tissue damage. These panels not only support pathophysiological understanding but also show promise for aiding in early diagnosis.
Ahmed Abdelhak, MD Assistant Professor of Neurology, University of California San Francisco
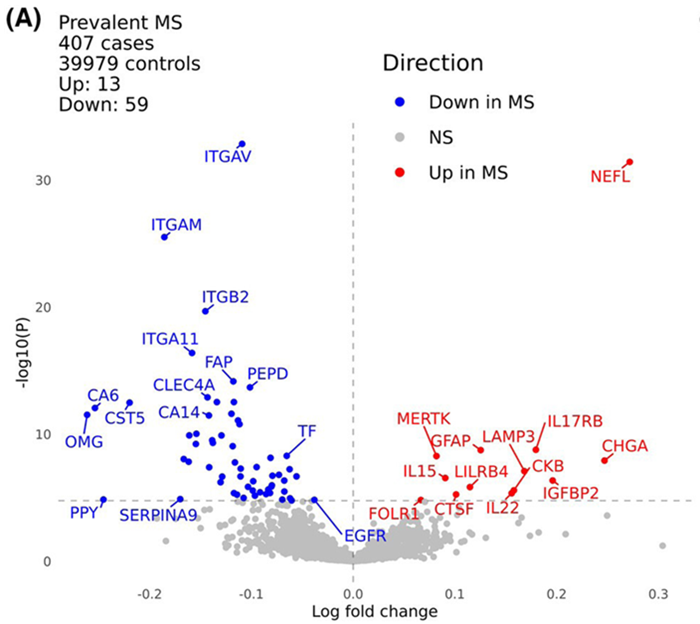
Figure 4. Plasma proteomic analysis of multiple sclerosis. Volcano plot displaying differences in plasma levels of proteins measured with Olink proteomics between UK Biobank participants with (n = 407) and without (n = 39,979) MS at the time of sample collection. The x-axis indicates the log-fold change of the protein (values above 0 indicate proteins present at higher levels in the MS cohort, while those with values below 0 are present at higher levels in the control cohort). The y-axis indicates the negative log of the p-value for each protein (higher values indicate a more statistically significant result). Proteins surpassing a Bonferroni-corrected threshold of 5 % are shown in color; other results are shown in gray. Image Credit: Jacobs et al., Annals of Clinical and Translational Neurology, 2024
Which pathways hold novel insights for biomarker discovery in neurological diseases?
Understanding the biological pathways represented by a specific marker is an important aspect of successful biomarker development. Without this context, it is impossible to determine if a biomarker represents illness onset, progression, or response to treatment.
As neurology advances toward more robust, clinically meaningful fluid biomarkers, various biological pathways have emerged as attractive areas of research.
Addressing the challenge of characterizing the early stages of AD and other dementias will depend heavily on studying neuroinflammatory mechanisms, particularly the roles of astrocytes and microglia. These glial cells are increasingly understood to be active participants in disease progression and elucidating the timing and nature of their involvement may yield biomarkers that reflect early or preclinical disease states.
Lisa Vermunt, MD, PhD Assistant Professor, Neurochemistry Laboratory, Amsterdam UMC
Olink’s PEA™ platform can support your protein biomarker research journey
Olink's product portfolio, powered by breakthrough PEA technology, provides a comprehensive solution for biomarker research. It assists researchers from exploratory studies to clinical translation by enabling the simultaneous measurement of thousands to single digit number of analytes using a single, uniquely scalable platform.
Olink's portfolio comprises a variety of pre-configured and configurable panels, a simple, wash-free process, and the lowest sample volume required among multiplex immunoassay platforms.
About Olink®- Part of Thermo Fisher Scientific
Olink’s mission is to accelerate proteomics together with the scientific community, to understand real-time biology and gain actionable insights into human health and disease. Our innovative solutions deliver highly sensitive and accurate protein quantification, giving scientists the power to investigate complex biological processes with precision.
One platform. Endless possibilities.
Explore up to 5,400 proteins with high specificity, transparent data, and the flexibility to answer any research question. Meet the next-generation proteomics platform trusted by the scientific community, from small academic research teams through to leading pharma companies.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.