Drug development involves significant risk and expense. With average development costs topping $1 billion per approved medicine and stubbornly high failure rates, pharmaceutical leaders are increasingly relying on biomarkers to make better decisions and reduce drug development risk.
In this changing world, marked by rising demand to shorten timelines, lower costs, and increase success rates, biomarkers are swiftly progressing beyond their exploratory roots.
Protein biomarker research techniques are increasingly considered as critical assets throughout the drug development pipeline, from target selection and treatment evaluation to patient segmentation, thanks to significant technological developments.
When used correctly, biomarkers can make the research, development, and approval of medications more efficient, focused, and data-driven.1 This article captures the opinions of a live panel discussion that included:
- Dawn Waterworth, PhD – Executive Director, Immunology Translational Sciences, Johnson & Johnson Innovative Medicine, with extensive expertise in multiomics.
- Magnus Althage, PhD – Head of Translational Science Strategy, AstraZeneca R&D, specializing in human target validation, patient identification, and biomarker strategy.
- Jen-Chieh (Jay) Chuang, PhD – Director, Translational Biomarker Lead at Takeda Pharmaceuticals, focusing on liver diseases.
- Moderator: Ray Chen – Global Sr. Director, Strategic Accounts at Olink Proteomics (Part of Thermo Fisher Scientific).
Together, they shared practical insights into the strategic value of incorporating biomarkers early in the drug development and clinical trial design process to help identify the right drug targets, evaluate drug candidate efficacy and safety, investigate noninvasive endpoints, guide patient group segmentation, and make more informed decisions earlier – ultimately reducing drug development and clinical development timeline, cost, and risk.
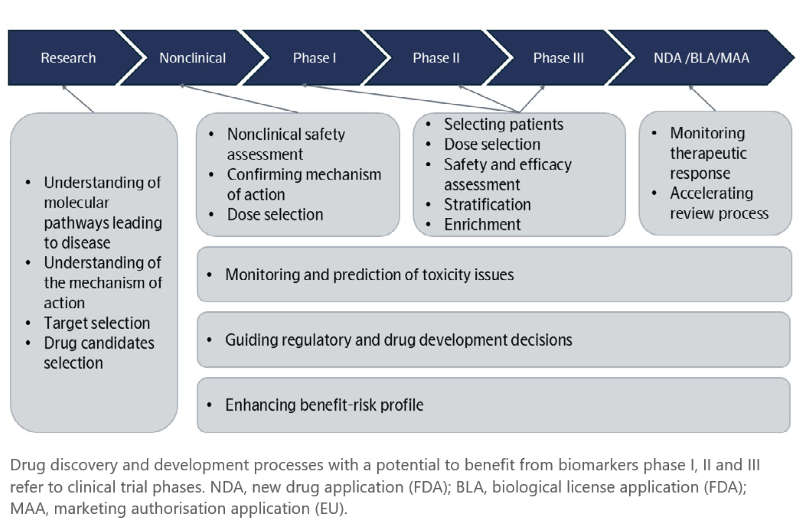
Figure 1. The potential application and benefits of biomarker utilization in various clinical trial phases1. Image Credit: Gromova, M., et al. (2020)
Theme 1 – Reducing risk at the source: How protein biomarker insights improve drug target selection
Poor target selection is a significant point of failure in drug development – many candidates advance only to fail later in development because the target's biological relevance and function in disease were not confirmed early on. These failures are more than just scientific defeats; they come at a high cost.
Late-stage clinical trial failures can cost up to $1.3 billion per medicine, deplete internal resources, and prevent potentially life-saving therapies from reaching patients.2
Improving target selection is more than good science; it is a strategic priority for reducing development risk and cost while accelerating the overall drug development process.
The panel discussion yielded a clear consensus: enhancing target selection requires first improving disease understanding. Protein biomarkers play a critical role in de-risking drug development by providing real-time, actionable insights that boost confidence in target selection and increase the likelihood of success.
High-throughput proteomic technologies like Olink enable the investigation of thousands of proteins across large cohorts, uncovering strong, biologically meaningful protein-disease relationships.
These proteomic insights, especially when combined with genomic data, offer a robust approach to target identification and validation, enabling more informed selection and prioritization, reducing the likelihood of late-stage attrition, and expediting progress toward clinical proof of concept.3
From the field: Expert insights
“We start by understanding the disease we want to treat, that defines everything else. Proteomics is just giving us a lot more in terms of identifying biomarkers that are really relevant to our processes.” Dawn Waterworth, PhD
“Traditionally, biomarker discovery was limited to analyzing one marker at a time, which constrained exploratory work and narrowed our view of disease biology. But with the advent of high-throughput proteomic platforms – capable of measuring thousands of proteins from just a small sample volume – we have unlocked the ability to explore the proteome at scale. This shift has opened the door to discovering novel, biologically relevant drug targets that were previously out of reach.” Magnus Althage, PhD
“We often hear about data-driven decision-making, but it is not just about having data – it is about the quality of the data and distilling that data into actionable insight. As multiomics technologies like proteomics evolve, generating massive amounts of information, the real value lies in translating those signals into knowledge that can guide smarter decisions.” Jen-Chieh (Jay) Chuang, PhD
Key biomarker-driven strategies for smarter drug target selection
1. Multiomics integration
Combining genomics, transcriptomics, and proteomics allows for a more comprehensive understanding of disease biology, exposing relationships between genes, transcripts, and proteins that single-omic techniques may overlook.
As Dawn Waterworth, PhD, pointed out, focusing simply on genomes may overlook the functional, real-time protein-level changes that drive disease, especially in varied settings.
When protein biomarkers are combined with genetic and transcriptome data, they provide a more detailed picture of biological pathways, which improves target finding precision.
The most remarkable proof comes from the largest population-scale proteogenomics effort, the UK Biobank Pharma Proteomics effort (UKB-PPP), where large-scale integration of proteomic and clinical data has identified new disease subtypes and improved prediction models for over 60 diseases.
This achievement emphasizes the importance of proteomics not only in disease mechanisms but also in diagnostics, guiding diagnostics development, detecting early efficacy or safety signals, improving trial inclusion criteria, supporting better decision making in overall drug development, and the clinical trial process.4
Table 1. How proteomics and genomics together provide a more complete view of disease understanding. Source: Olink®- Part of Thermo Fisher Scientific
| Precision Medicine Goals |
Genomics |
Proteomics |
| Find the right drug |
Genetic clues often indirect |
Proteins are the direct drug targets |
| Find the right patient |
Strong for rare conditions |
Enables broad stratification across diseases |
| Find the right time |
Genetic sequence is stable |
Protein levels are dynamic and time-sensitive |
2. Reverse translation
Reverse translation, or the process of using clinical trial results to drive upstream R&D decisions, has emerged as an effective method for decreasing development risk.
Magnus Altage, PhD, and Jay Chuang, PhD, noted that proteomic data from late-stage studies might indicate how a drug influences biological pathways in real patients, providing vital information about the mechanism of action, biological pathways, and prospective combination strategies.
This feedback loop not only enables improved early target selection, but it also helps to deprioritize non-performing prospects earlier. According to Jay Chuang, PhD, "a quick kill is already a success" – especially when it avoids costly downstream failures.
3. Maximize sample value
Sample availability in both early discovery and clinical trials is frequently limited, especially for invasive biopsies or unusual populations. The capacity to gain maximum insight from minimal input is critical.
Scalable, high-throughput technologies such as Olink enable researchers to characterize thousands of proteins from small-volume samples, resulting in richer datasets without depleting valuable material.
As Jay Chuang, PhD, noted, “Per unit of sample collected, if you can maximize the number of data points you acquire, you will have a better chance of generating better knowledge.”
This not only recognizes the importance of patient samples, but also allows researchers to address more complicated, system-level questions from the start.
Theme 2 – Reducing risk downstream: How biomarker insights support data-driven decision-making
In addition to improving drug target selection, protein biomarker insights are revolutionizing drug development and clinical trial design by allowing data-driven "go/no-go" decisions based on real-time efficacy and safety monitoring and educated patient segmentation.
Together, these developments represent a paradigm shift: proteomic biomarkers are no longer just exploratory tools; they are becoming core decision-making assets in clinical operations, providing critical information to optimize drug candidates and the clinical trial evaluation process, deciding when to discontinue underperformers, and dynamically refining development strategies.
From the field: Expert insights
“There are a lot of high-stakes decisions to make in development, and sometimes, an early ‘quick kill’ is already a success. Having efficacy and safety signals early, in real time, helps us make those calls with greater confidence.” Jen-Chieh (Jay) Chuang, PhD
“You want to start integrating biomarkers from the very beginning. The more you understand your disease and its biomarkers during discovery, the more informed you will be during development – they are two sides of the same coin.” Dawn Waterworth, PhD
“The most exciting frontier is in patient segmentation – using proteomic and genomic profiles to identify distinct subgroups we could not isolate before. It is a game-changer for matching the right patients with the right therapies, and it is where we will see the biggest impact in the near future.” Magnus Althage, PhD
Key biomarker-driven strategies for development risk mitigation
1. Early efficacy evaluation: Enabling fast, informed decisions
Proteomic biomarkers provide real-time insights into target engagement and pathway modulation – key indicators that a medicine is performing as intended, allowing development teams to quickly decide whether to advance or terminate a molecule. This "fail-fast" method saves time and resources by terminating ineffective programs early.
2. Patient segmentation: Matching therapies to individual molecular profiles
Traditional clinical features can hide genetic variability among patients. Proteomics can bridge these gaps by identifying distinct molecular signatures within seemingly identical populations, enabling more precise, effective, and tailored treatments 6, 8.
Dawn Waterworth, PhD, Jay Chuang, PhD, and Magnus Althage, PhD, all identified patient segmentation as a critical and exciting application of protein biomarkers that will alter drug development and clinical trial processes.
“Proteomics gives us the tools to finally tease apart the patient subsets we have always known existed. Now we can match therapies to the biology more precisely – and that is where we will see the biggest impact.”
3. Non-invasive endpoints: Reducing patient burden, improving recruitment
In several disease categories, particularly liver and kidney problems, clinical studies use invasive biopsies to determine endpoints. Protein biomarkers and comprehensive proteomic platforms such as Olink have the potential to improve these procedures by providing more accessible and dependable blood-based biomarkers.
Jay Chuang, PhD, emphasized this shift:
If we could move from biopsy-based to blood-based readouts, it would improve feasibility, safety, and patient recruitment – especially in chronic diseases where repeat biopsies are a barrier.
The ability to use biomarkers as surrogate endpoints also allows for speedier readouts, shorter trial durations, and increased patient desire to participate in studies, thus lowering development risk and cost.
Summary
- Smarter target selection through better disease understanding: Protein biomarkers provide valuable insights into disease biology, enabling teams to select more relevant therapeutic targets and avoid late-stage failures.
- Stronger discovery decisions with multiomics integration: Integrating proteomics, genomes, and transcriptomics can improve therapeutic target selection and validation by providing a more comprehensive understanding of disease mechanisms.
- Reverse translation turns trials into upstream insight: Using proteomic data from late-stage studies can improve understanding of disease causes, enable innovative medication combinations, and speed up "fail fast" decisions.
- Early go/no-go decisions with proteomic biomarkers: Proteomic biomarkers enable early decision-making by measuring target engagement and pathway activity in real-time, saving time and money.
- Improved trial success through patient segmentation: Proteomics identifies patient subgroups within clinically similar populations, enabling better trial design and precision medicine.
- Safer, scalable trials with non-invasive biomarker endpoints: Safer and scalable trials using non-invasive biomarker outcomes. Blood-based biomarkers as endpoints could eliminate the need for biopsies, lowering patient burden, increasing recruitment, and allowing for more frequent, cost-effective monitoring.
Closing thoughts
As biomarkers become more integrated into the drug development process, their role transitions from supportive to strategic.
Beyond target identification, biomarkers provide fit-for-purpose tests, such as target engagement and disease-associated indicators, which connect preclinical models to clinical results, providing greater evidence to support regulatory and investment decisions.
This comprehensive biomarker paradigm enables research teams to make more informed go/no-go decisions, reducing risk, shortening timelines, and increasing the likelihood of successful therapeutic advances.3
Ray Chen concluded, “The question is no longer whether biomarkers can improve drug development and clinical trial design, it is how we scale their value across every decision.”
References and Further Reading
- Gromova, M., et al. (2020). Biomarkers: Opportunities and Challenges for Drug Development in the Current Regulatory Landscape. Biomarker Insights, 15, p.117727192097465. DOI: 10.1177/1177271920974652. https://journals.sagepub.com/doi/10.1177/1177271920974652.
- DiMasi, J.A., Grabowski, H.G. and Hansen, R.W. (2016). Innovation in the pharmaceutical industry: New estimates of R&D costs. Journal of Health Economics, 47, pp.20–33. DOI: 10.1016/j.jhealeco.2016.01.012. https://www.sciencedirect.com/science/article/abs/pii/S0167629616000291?via%3Dihub.
- Wagner, J.A. (2008). Strategic Approach to Fit-for-Purpose Biomarkers in Drug Development. Annual Review of Pharmacology and Toxicology, 48(1), pp.631–651. DOI: 10.1146/annurev.pharmtox.48.113006.094611. https://www.annualreviews.org/content/journals/10.1146/annurev.pharmtox.48.113006.094611.
- Carrasco-Zanini, J., et al. (2024). Proteomic signatures improve risk prediction for common and rare diseases. Nature Medicine, (online) pp.1–10. DOI: 10.1038/s41591-024-03142-z. https://www.nature.com/articles/s41591-024-03142-z.
- Sun, B.B., et al. (2023). Plasma proteomic associations with genetics and health in the UK Biobank. Nature. DOI: 10.1038/s41586-023-06592-6. https://www.nature.com/articles/s41586-023-06592-6.
- Zannad, F., et al. (2022). Effect of empagliflozin on circulating proteomics in heart failure: mechanistic insights into the EMPEROR programme. European Heart Journal, 43(48), pp.4991–5002. DOI: 10.1093/eurheartj/ehac495. https://academic.oup.com/eurheartj/article/43/48/4991/6676779?login=false.
- Åkesson, J., et al. (2023). Proteomics reveal biomarkers for diagnosis, disease activity and long-term disability outcomes in multiple sclerosis. Nature Communications, (online) 14(1). DOI: 10.1038/s41467-023-42682-9. https://www.nature.com/articles/s41467-023-42682-9.
- Parra, E.R., et al. (2024). Multi-omics analysis reveals immune features associated with immunotherapy benefit in squamous cell lung cancer patients from Phase III Lung-MAP S1400I trial. Clinical Cancer Research: An Official Journal of the American Association for Cancer Research. (online) DOI: 10.1158/1078-0432.CCR-23-0251. https://aacrjournals.org/clincancerres/article/30/8/1655/742118/Multi-omics-Analysis-Reveals-Immune-Features.
- Li, M., et al. (2023). Cerebrospinal fluid immunological cytokines predict intracranial tumor response to immunotherapy in non-small cell lung cancer patients with brain metastases. OncoImmunology, 13(1). DOI: 10.1080/2162402x.2023.2290790. https://www.tandfonline.com/doi/full/10.1080/2162402X.2023.2290790.
- Jia, X., et al. (2023). Identification and multicentric validation of soluble CDCP1 as a robust serological biomarker for risk stratification of NASH in obese Chinese. Cell Reports Medicine, 4(11), pp.101257–101257. DOI: 10.1016/j.xcrm.2023.101257. https://www.sciencedirect.com/science/article/pii/S2666379123004342?via%3Dihub.
About Olink®- Part of Thermo Fisher Scientific
Olink’s mission is to accelerate proteomics together with the scientific community, to understand real-time biology and gain actionable insights into human health and disease. Our innovative solutions deliver highly sensitive and accurate protein quantification, giving scientists the power to investigate complex biological processes with precision.
One platform. Endless possibilities.
Explore up to 5,400 proteins with high specificity, transparent data, and the flexibility to answer any research question. Meet the next-generation proteomics platform trusted by the scientific community, from small academic research teams through to leading pharma companies.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.