This article is based on a poster originally authored by Qiuyuan Yang, Kaiqiang Hu, Lei Liu, Tingting Guo, Haiyan Gu, Pengwei Pan, and Fang He.
The study aimed to unravel the underlying processes that explain the variable efficacy of Dabrafenib, a potent inhibitor of the BRAF V600E mutation, across different cancer types.
Methods
The Cell Titer-Glo (CTG) test was used to evaluate Dabrafenib's potency across a wide range of cell lines, with a focus on skin and myeloid cancer cells.
To uncover the molecular cause behind Dabrafenib resistance, the researchers used bioinformatics analysis, examining high-throughput sequencing data from public databases and linking Dabrafenib potency to specific gene expression profiles.
They then conducted a comprehensive investigation of 19 BRAF V600E-mutation cancer cell lines to determine their sensitivity to Dabrafenib.
Finally, synergistic effect analysis was used to evaluate the putative genes involved in Dabrafenib resistance.
Results
The CTG assay results showed that skin cancer cell lines were sensitive to Dabrafenib, while myeloid cancer cell lines exhibited significant resistance to the medication.
Bioinformatics analysis revealed that the PI3K signaling pathway was significantly elevated in myeloid cancer cell lines, particularly compared with skin cancer cell lines.
Among the PI3K family, PIK3CG expression levels differed the most between skin and myeloid cancer cell lines, a finding corroborated by clinical evidence.
While there was no substantial variation in PI3K signaling pathway expression levels between sensitive and resistant cell lines with the BRAF V600E mutation, PIK3CG expression remained strongly linked with Dabrafenib sensitivity.
In vitro combination tests indicated that PIK3CG inhibitor substantially reduced Dabrafenib resistance.
Conclusion
This unbiased, bioinformatics-driven research revealed the underlying mechanisms behind high PIK3CG expression and Dabrafenib resistance. These findings carve out a route to the development of novel approaches to treating BRAF inhibitor-resistant malignancies.
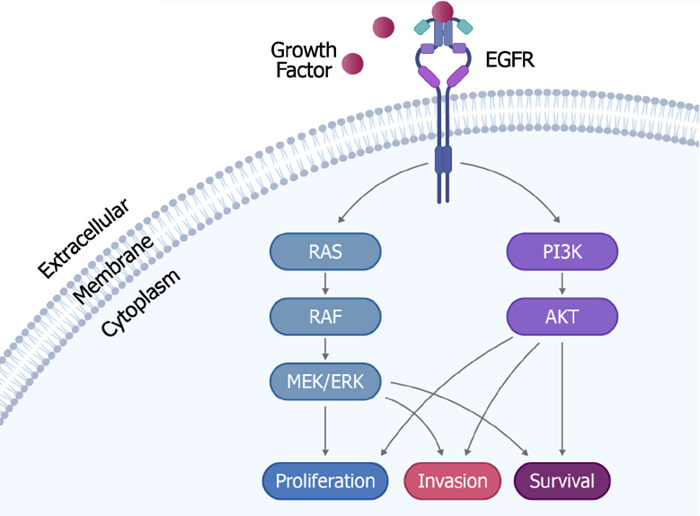
Fig 1. The Signaling Pathway of RAF/MEK/ERK and PI3K/AKT. Image Credit: DOI:10.3389/fonc.2022.1074726
The RAF/MEK/ERK signaling system regulates proliferation, invasion, and survival. The PI3K/AKT pathway acts as another survival route whose overactivation has been linked to BRAF inhibitor resistance.

Fig 2. Dabrafenib Sensitivity Comparison in Skin and Myeloid Cancer Cells. Image Credit: Pharmaron
Skin cancer cells include a considerable number of BRAF V600E mutations and are more sensitive to Dabrafenib than myeloid-derived cell lines. The CTG assay was used to determine 7-day IC50 values. The mutation status was checked using the DepMap database.
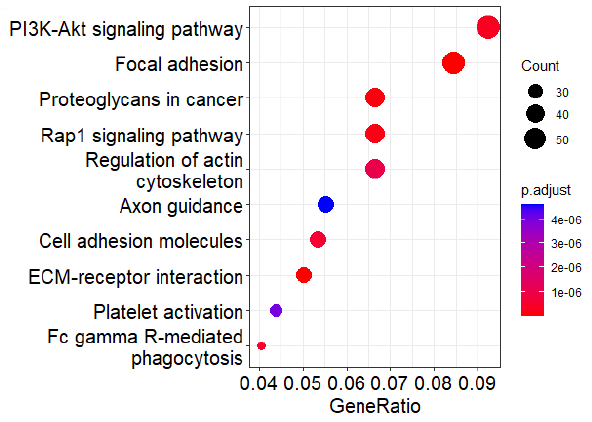
Fig 3. KEGG Enrichment Analysis Reveals PI3K-Related Pathway Ranks at the Top. Image Credit: Pharmaron
When differentially expressed genes between skin and myeloid cell lines were examined, the PI3K-Akt signaling pathway was found to be the most highly enriched.
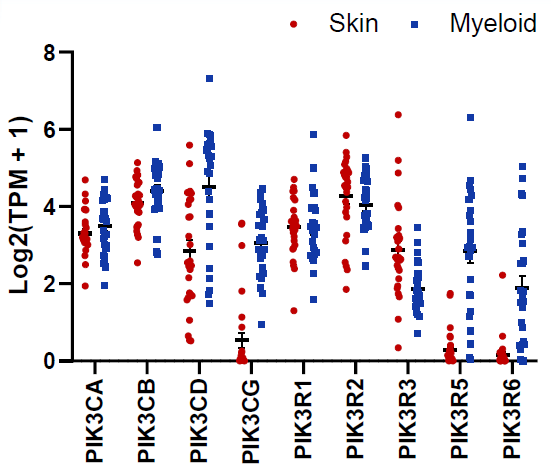
Fig 4. Gene Expression of Class I PI3K Family Members. Image Credit: Pharmaron
Skin and myeloid cell lines exhibit significantly different expression levels of PIK3CG and other PI3K family members.
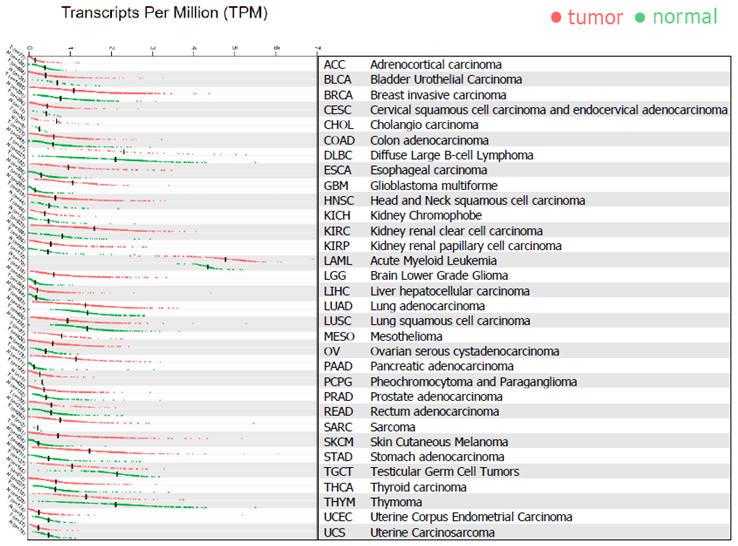
Fig 5. PIK3CG is Predominantly Expressed in Myeloid Cancer. Image Credit: Tang, Z. et al. (2019) GEPIA2: an enhanced web server for large-scale expression profiling and interactive analysis. Nucleic Acids Res. DOI: 10.1093/nar/gkz430
Clinical gene expression data from TCGA, evaluated with Gepia2, profiles PIK3CG across tumor and normal tissues from patients, suggesting that PIK3CG plays an important role in the oncogenesis of myeloid malignancies.
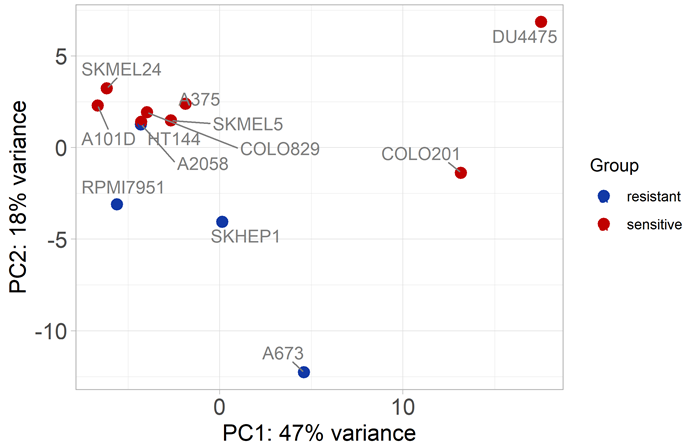
Fig 6. Principal Component Analysis (PCA) of genes involved in the PI3K-Akt Pathway in Cell Lines with BRAF V600E Mutation. Image Credit: Pharmaron
The findings revealed that cell lines that are resistant and sensitive to Dabrafenib cannot be reliably clustered.
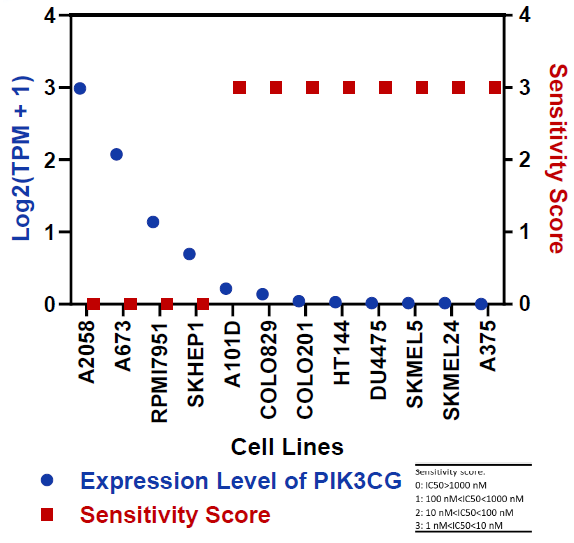
Fig 7. In Cell Lines with BRAF V600E Mutation, Sensitivity to Dabrafenib is Associated with the Expression Level of PIK3CG. Image Credit: Pharmaron
PIK3CG expression is high in Dabrafenib-resistant cell lines, but low in Dabrafenib-sensitive cell lines.
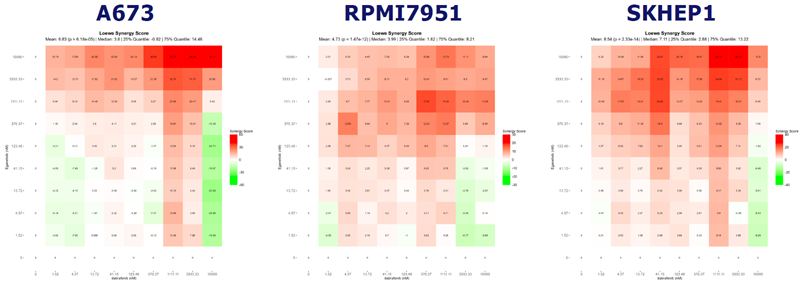
Fig 8. Combination Therapy of Dabrafenib with a PIK3CG Inhibitor Shows Synergy. Image Credit: Pharmaron
The synergistic effect of PIK3CG inhibitor Eganelisib and Dabrafenib was investigated in cell lines with high PIK3CG expression, demonstrating synergy and implying a potential combination therapy for improved outcomes.
Highlights
- Dabrafenib is more effective in treating skin cancer cell lines than myeloid cancer cells.
- The PI3K-Akt pathway, namely PIK3CG, is substantially elevated in myeloid cancer cell lines and linked to resistance to Dabrafenib.
- Clinical data indicate a considerable difference in PIK3CG expression between myeloid malignancies and normal tissues.
- In BRAF V600E-mutated cell lines, PIK3CG expression levels are associated with sensitivity to Dabrafenib.
- Combining Dabrafenib with a PIK3CG inhibitor can improve efficacy in resistant cell lines, indicating a possible treatment strategy.
Qiuyuan Yang1, Kaiqiang Hu1, Lei Liu2, Tingting Guo2, Haiyan Gu2, Pengwei Pan1, Fang He1
1in vitro Biology, Pharmaron Beijing Co., Ltd. 2in vitro Biology, Pharmaron Ningbo Co., Ltd.
About Pharmaron
Pharmaron is a premier R&D service provider for the life sciences industry. Founded in 2004, Pharmaron has invested in its people and facilities and established a broad spectrum of research, development, and manufacturing service capabilities throughout the entire drug discovery, preclinical, and clinical development process across multiple therapeutic modalities, including small molecules, biologics, and CGT products. With over 17,000 employees and operations in China, the U.S., and the U.K., Pharmaron has an excellent track record of delivering R&D solutions to its partners in North America, Europe, Japan, and China.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.
Last Updated: Mar 19, 2026