This article is based on a poster originally authored by Ce Zhong, Lu Bai, Tongtong Wang, Yiying Shao, Lingyun Zhang, Lu Zhang, and Jingqi Huang.
CRISPR screens have successfully identified targetable genes for cancer research and drug development. While most CRISPR screens are performed in vitro using cell lines, these models do not adequately represent the impact of the in vivo environment.
An alternative, powerful method is in vivo CRISPR screening with xenograft models. This technique can be used to examine the complexities of cellular interactions, such as communication between organs, systemic signaling, and immune responses.
It allows for the study of biological processes in which cells are influenced by their natural environment and physiological factors.
Methods
The computational tool MAGeCK (Model-based Analysis of Genome-wide CRISPR-Cas9 Knockout) was used to calculate the gene enrichment score. Image Credit: Pharmaron
Clonal expansion of A549 cells in vivo under Trametinib treatment
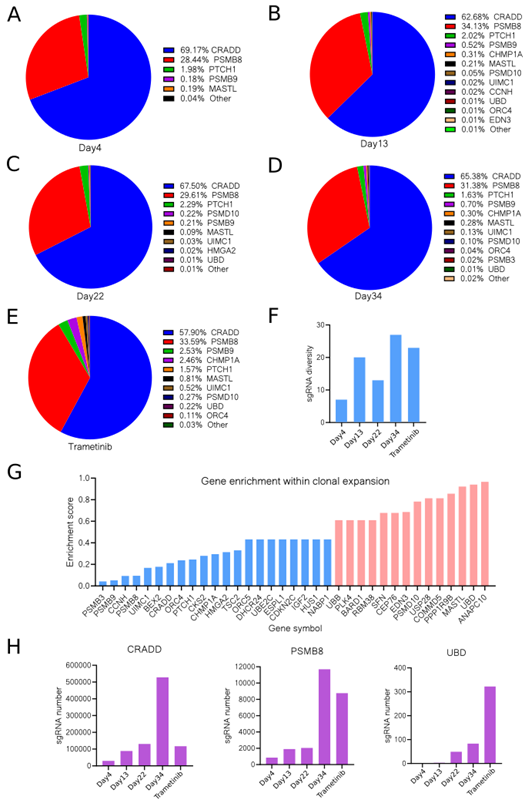
Summary of in vivo CRISPR screening in A549 cells in mice with and without Trametinib treatment. Image Credit: Pharmaron
sgRNAs were detected at several time points (A-E) following transfection, including Day 4, Day 13, Day 22, and Day 34. (F) SgRNA diversity at various time points.
(G) Gene enrichment score for each gene in comparison to vehicle and Trametinib therapy. Blue and red bars reflect genes that were negatively and favorably chosen, respectively. (H) The number of sgRNA targeting genes, such as CRADD, PSMB8, and UBD, varied depending on the time point.
Targets and compounds
Potential targets identified through the CRISPR screen. Source: Pharmaron
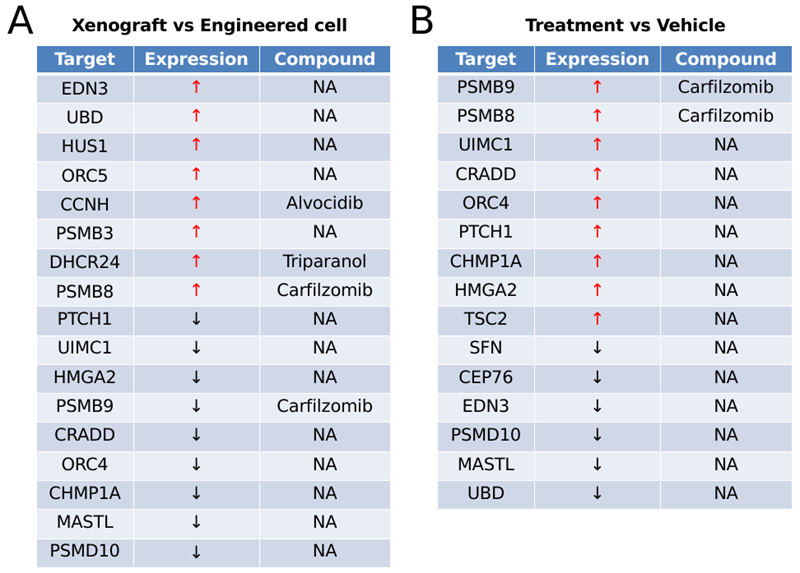
The table indicates potentially druggable sites implicated in clonal growth in two scenarios: without (A) and with (B) Trametinib treatment.
Positively chosen genes are indicated by red arrows, whilst negatively selected genes are represented by black arrows. The list also includes FDA-approved medicines that target these genes.
Clonal expansion in A549 in vitro vs. in vivo xenograft model
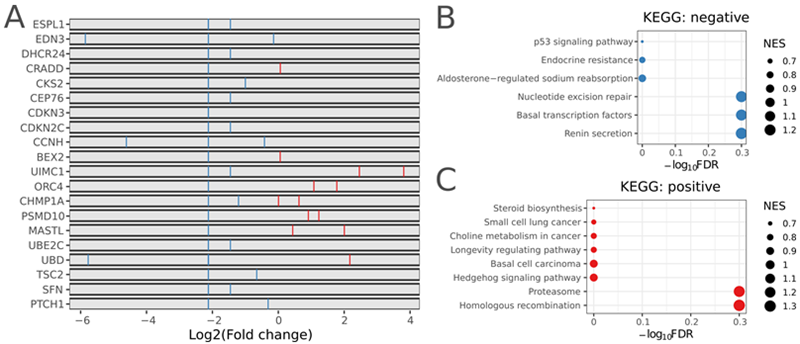
Clonal expansion and gene enrichment in the A549 xenograft model without Trametinib treatment. Image Credit: Pharmaron
(A) Comparison of sgRNA levels between xenograft and modified cell groups. The red and blue bars show increased and decreased cell proliferation, respectively, with sgRNAs targeting specific genes. (B) KEGG enrichment of negatively chosen genes. (C) KEGG enrichment of favorably selected genes.
Clonal expansion in A549 vehicle vs. Trametinib-treated xenograft model
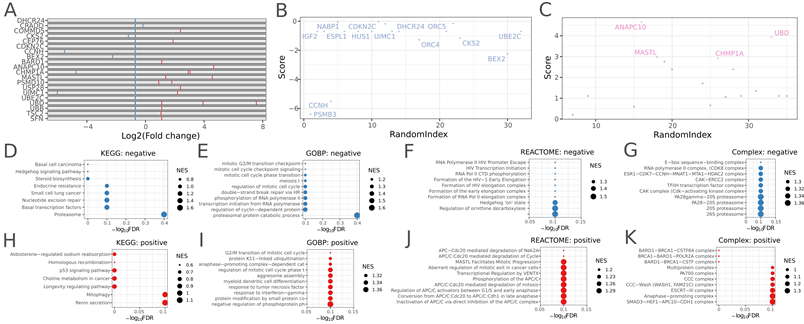
Clonal expansion and gene enrichment in A549 xenograft model with Trametinib treatment. Image Credit: Pharmaron
(A) with a fold difference in sgRNAs between the Trametinib and vehicle groups. The red and blue bars depict increased and decreased cell proliferation, respectively, induced by sgRNAs targeting specific genes. (B) Rank and enrichment score for each negatively selected gene.
(C) Rank and enrichment score for each positively selected gene. (D-G) Negatively selected gene enrichment in KEGG (D), GO (E), REACTOME (F), and complex (G). (H-K) Positively selected genes are enriched in KEGG (H), GO (I), REACTOME (J), and complex.
Conclusion
- Gene variants in the physiological environment were identified using the in vivo CRISPR screening technique
- In an A549 xenograft model, 45 sgRNAs were detected against 27 genes, with 17 overlapping with engineered cells (eight upregulated and nine downregulated)
- A comparison of model and treatment groups revealed 38 sgRNAs corresponding to 23 genes, which overlapped with 15 genes (nine upregulated and six downregulated)
- The in vivo CRISPR technique is a great tool for identifying novel cancer targets and developing new anticancer treatments.
Ce Zhong, Lu Bai, Tongtong Wang, Yiying Shao, Lingyun Zhang, Lu Zhang, Jingqi Huang*
*Corresponding author: Jingqi Huang, [email protected]
About Pharmaron
Pharmaron is a premier R&D service provider for the life sciences industry. Founded in 2004, Pharmaron has invested in its people and facilities and established a broad spectrum of research, development, and manufacturing service capabilities throughout the entire drug discovery, preclinical, and clinical development process across multiple therapeutic modalities, including small molecules, biologics, and CGT products. With over 17,000 employees and operations in China, the U.S., and the U.K., Pharmaron has an excellent track record of delivering R&D solutions to its partners in North America, Europe, Japan, and China.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.
Last Updated: Mar 19, 2026