This article is based on a poster originally authored by Gillian F Watt, Simon Taylor, Marta Westwood, Li Feng, Yasong Cui, Tan Pang, Rui Yuan, Tingzhong Wang, and Kui Lin.
PARP inhibitors, such as Olaparib, have shown significant therapeutic benefit in the treatment of BRCA1/2-mutated breast and ovarian cancers. PARP enzymes are involved in numerous physiological functions, including stress response, DNA repair, and apoptosis.
As our understanding of PARP inhibition mechanisms advances, the search for PARP1-selective inhibitors has become a priority for future treatments.
This article, a collaboration between Pharmaron and NewBay Pharma, provides translational insights from multidisciplinary data integration to enhance the developability evaluation of NB-0290, a selective PARP1 inhibitor.
Identification of NB-0290, a selective PARP1 inhibitor
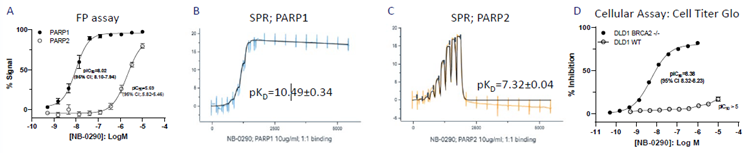
Image Credit: Pharmaron
- Prior to SPR profiling, PARP1-selective drugs were identified using a fluorescence polarization (FP) binding assay (A), confirming sub-nM binding and a delayed off-rate (B, C). An FP experiment demonstrated DNA trapping by PARP1 (pIC50=7.2; data not shown).
- NB-0290 had low nM growth-inhibitory potency in the BRCA2-deficient DLD1 cell line and no effect in the WT cell line (D). NB-0290 demonstrated activity in a BRCA1 mutant cell line (MDA-MB-436) with a pIC50 of 8.54 (95 % CI 8.39-8.17).
- A panel of 675 targets showed limited activity, indicating an off-target profile. At 30 μM, four targets (Ach M2, acetylcholinesterase, CB2, and GPR109A) exhibited activity levels more than 50 %. No hERG inhibition was observed (IC50 > 30 μM).
ADME/PK profile
Source: Pharmaron
| . |
. |
| MW / Measured log D7.4 |
407.47 / 2.16 |
Permeability
(AB/BA/ER) cm/s x10-6 |
Caco2: 3.4 / 33 / 9.7
MDCK-MDR1: 0.55 / 35 / 63
BCRP: 14 / 26 / 1.9 |
TD Solubility (uM)
SGF / FeSSIF / FaSSIF |
2125 / 1729 / 262 |
CYP inhibition IC50 (uM)
1A2, 2C9, 2C19, 2D6, 3A4 |
> 100 uM; no CYP3A4 TDI |
| PXR activation |
< 2-fold at 30 uM |
Fraction unbound, plasma
Mouse / Rat / Dog / Human |
4.8 % / 5.2 % / 36 % / 6.4 % |
Hepatocyte CLint
Suspension
Low Cl Plated format |
(uL/min/106 cells)
< 3 (M/R/Do/Hu)
0.1 (Hu) |
Source: Pharmaron
| Pharmacokinetics |
Mouse / Rat / Dog |
| IV dose |
1 /1 / 0.5 mg/kg |
| CLp (CLp,u) mL/min/kg |
0.9 / 2.2 / 17.1
(19 / 42 / 48) |
| Vss (Vss,u) L/kg |
0.3 / 0.3 / 1.1
(6.3 / 5.8 / 3.1) |
| T½ h |
4.3 / 3.0 / 1.1 |
| Brain Kp,uu |
0.09 in rat |
| Oral dose* |
1 / 3 / 2 mg/kg |
| Fpo % |
180 / 43 / 163 |
*1 % (w/v) methyl cellulose 400cp
- The in vitro ADME features aligned with an oral, once-daily drug with a promising development profile.
- The preclinical PK profile showed low/moderate clearance and volume, with unbound values consistent across species. Oral bioavailability was moderate to high and proportionate over the working dose range.
Profound tumor regression following NB-0290 oral dosing in the MDA-MB-436 xenograft model concomitant with tumor biomarker changes indicative of target engagement.
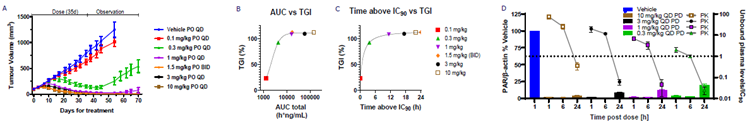
Image Credit: Pharmaron
- Oral dosing of 0.3 mg/kg once daily (PO QD) inhibited tumor growth in a dose-dependent manner. Following therapy, the tumors continued to shrink at the two highest dose levels (A).
- NB-0290 plasma exposure increased in proportion to the dose administered. The study of concentration-effect connections revealed that efficacy is driven by unbound plasma concentrations at or above the in vitro CTG IC90 (B,C).
- PARylation, which adds poly(ADP-ribose) chains to proteins to indicate PARP enzymatic activity, is a pharmacodynamic marker for tumor response. Treatment with NB-0290 for five days inhibited PARylation in a dose- and time-dependent manner (D, left axis).
- Unbound plasma concentrations at or above the in vitro CTG IC90 significantly decrease tumor PARylation (D, right axis) and correspond with tumor growth inhibition. In vitro IC90 for at least six hours was determined as the minimal effective concentration (MEC) in plasma required for effectiveness.
Human PK and dose prediction
Human clearance, volume of distribution, and absorption, predicted independently as shown in the schematic below, were comparable to once-daily oral medication.
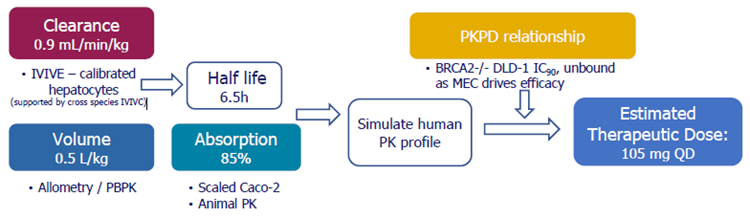
Image Credit: Pharmaron
- Clearence: The anticipated clearance rate for hepatocytes was 0.9 mL/min/kg. Metabolism was identified as the predominant mechanism of elimination.
- Volume: The tissue partition equations (Lukacova technique, Gastroplus program) projected a volume of 0.5 L/kg.
- Absorption: Caco2 and animal studies indicated a high absorption rate of 85 %.
- Predicted therapeutic dose: The predicted therapeutic dose was 105 mg once daily.
Integrated developability assessment positions NB-0290 for preclinical development
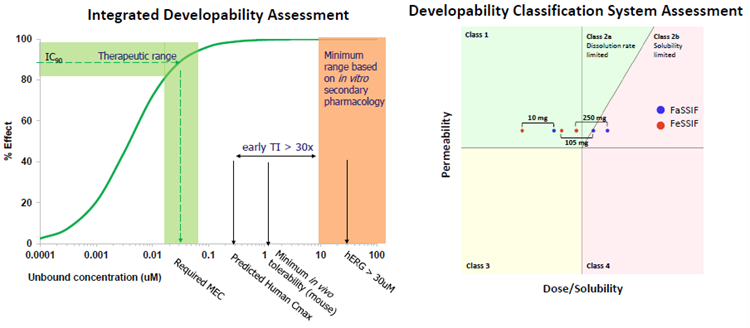
Image Credit: Pharmaron
- An integrated investigation of pharmacology, projected human PK, and early safety profiling revealed a high (∼30x) therapeutic index (TI) and developability potential.
- The DCS assessment identified possibilities for Class 1-2 formulation development.
Conclusion
- NB-0290 is an orally bioavailable, small-molecule selective PARP1 inhibitor with promising potential for treating BRCA1/2 mutant breast and ovarian cancer.
- Data was pooled across disciplines to predict whether the intended applicant profile was met.
- Cross-discipline integration resulted in a high-quality candidate for pre-clinical development within a year.
Gillian F Watt1, Simon Taylor1, Marta Westwood2, Li Feng3, Yasong Cui3, Tan Pang4, Rui Yuan4, Tingzhong Wang5, and Kui Lin5
1Drug Discovery, Pharmaron, UK | 2Biophysics, Pharmaron, UK | 3In Vitro Biology, Pharmaron, China | 4In Vivo Pharmacology, Pharmaron, China | 5NewBay Pharma, China
About Pharmaron
Pharmaron is a premier R&D service provider for the life sciences industry. Founded in 2004, Pharmaron has invested in its people and facilities and established a broad spectrum of research, development, and manufacturing service capabilities throughout the entire drug discovery, preclinical, and clinical development process across multiple therapeutic modalities, including small molecules, biologics, and CGT products. With over 17,000 employees and operations in China, the U.S., and the U.K., Pharmaron has an excellent track record of delivering R&D solutions to its partners in North America, Europe, Japan, and China.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.
Last Updated: Mar 19, 2026