History
Cause and symptoms
Genetics
Epidemiology
Case reports
Diagnosis and treatment
References
Further reading
Jervell and Lange Nielsen syndrome (JLNS) is a rare cardio-auditory disorder with an autosomal recessive pattern of inheritance. JLNS was first reported in Norway in 1957. Bilateral sensorineural hearing loss, a prolonged QTc interval, and a high risk of sudden cardiac death are some characteristic features of JLNS. It's a type of long QT syndrome (LQTS) passed down through the generations.
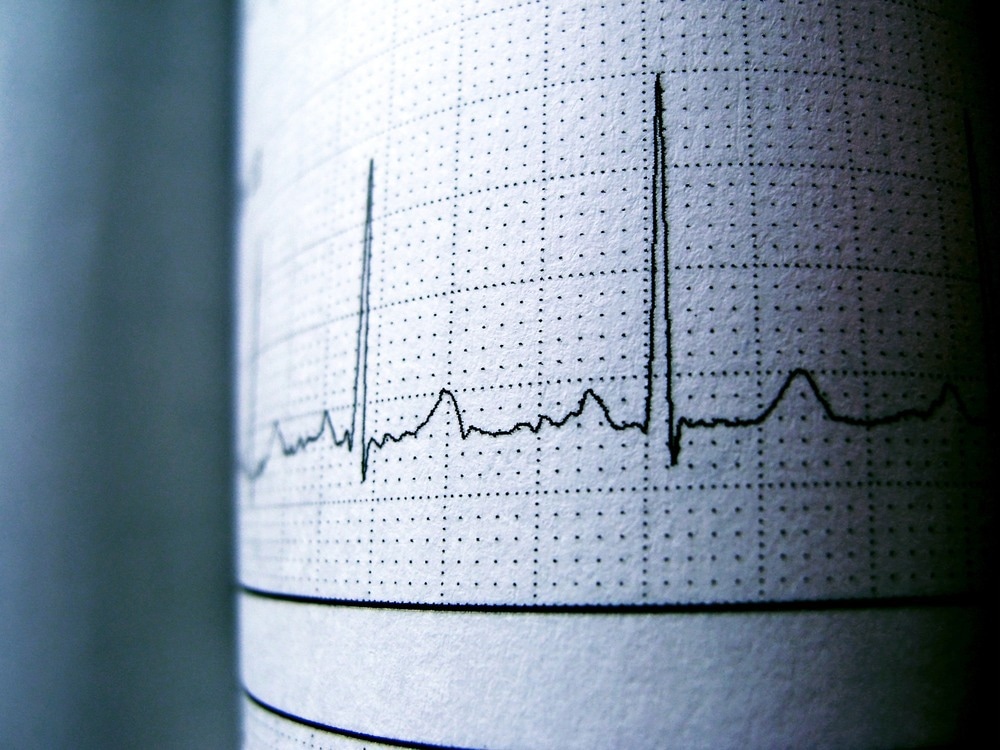
Image Credit: Luan Rezende/Shutterstock.com
JLNS is one of the most severe heart rhythm abnormalities, and it can result in sudden death in young individuals. JLNS is caused by mutations in the KCNQ1 or KCNE1 genes that are homozygous (or compound heterozygous).
History
Anton Jervell and his colleague Fred Lange-Nielsen presented the first report on a familial illness defined by a considerably prolonged QT interval, congenital deafness, and a high incidence of sudden cardiac death in childhood in 1957. This instance involved a household in which four of six children, born to unrelated parents, died unexpectedly as children.
Cause and symptoms
Mutations in the KCNE1 and KCNQ1 genes cause Jervell and Lange-Nielsen syndrome. JLNS is a congenital condition passed down through the generations in an autosomal recessive pattern. About 90% of Jervell and Lange-Nielsen syndrome cases are caused by mutations in the KCNQ1 gene.
Almost half of all individuals with JLNS have symptoms before age three. A congenitally deaf kid with syncopal episodes during periods of stress, activity, or panic is the classic presentation of Jervell and Lange-Nielsen syndrome (JLNS). Congenital, bilateral, profound, and sensorineural deafness are types of deafness reported in JLNS patients. More than half of children with JLNS who are not treated die before age 15.
The QT interval is frequently quite long (>500 ms) and is linked to tachyarrhythmias (such as ventricular tachycardia, Torsades de Pointes (TdP) ventricular tachycardia, and ventricular fibrillation) that can result in syncope or sudden death. With the exception of calmodulin-related LQTS, patients become symptomatic much earlier in this variant of LQTS. Intensive or sudden emotion, competitive sports, panic, or jumping into cold water cause cardiac episodes in nearly 90% of patients. Patients with JLNS who have KCNQ1 mutations had longer QT intervals and are more likely to have arrhythmic episodes than those who have KCNE1 mutations.
Genetics
Mutations in these genes cause potassium channels to lose their normal structure and function or prevent them from forming. Hearing loss and an abnormal heart rhythm are symptoms of Jervell and Lange-Nielsen syndrome, which are caused by alterations in the movement of potassium ions in the inner ear and cardiac muscle.
The KCNE protein family consists of five members (KCNE1–KCNE5), each of which is a tiny single-pass integral membrane protein with a residue count ranging from 103 to 177. These genes direct the production of proteins that work together to generate a channel across cell membranes. Positively charged potassium atoms (ions) are transported out of cells through these channels. The flow of potassium ions via these channels is essential for maintaining proper inner ear and heart muscle activities.
Epidemiology
The prevalence of the illness varies greatly depending on the demographic investigated, but it affects between 1 and 6 people per 1,000,000 people worldwide. Denmark, Norway, and Sweden have an abnormally high incidence of 1 in 200,000 due to established genetic features. In Turkey, there have also been reports of a rise in the number of cases.
Case reports
They describe the case of a 10-year-old Moroccan boy who presented with recurrent episodes of syncope after being born with congenital hearing loss and a drastically prolonged QT interval. He is the firstborn of a healthy consanguineous couple who both hail from Morocco's northwest. There was no history of sudden death, deafness, syncope, epilepsy, or any hereditary diseases in the family.
Image Credit: Bangkoker/Shutterstock.com
The pregnancy had been medically monitored, and there had been no difficulties. His mother had no previous history of medication use or phytotherapy. His birth weight and length were normal, and he showed no indicators of dysmorphia. On auditory evoked potential testing at 6 months, he was diagnosed with severe bilateral SNHL. At the age of 24 months, he had his first syncopal episode.
An echocardiogram revealed a structurally normal heart. A -adrenergic blocker was given to the patient right away. His parents and two younger siblings, aged seven and one years, were all clinically normal. Sanger sequencing analysis was used to evaluate the complete coding region and flanking intronic regions of KCNQ1 and KCNE1 for molecular genetics. The index patient was found to have a homozygous frameshift mutation as a result of this. This mutation was found in both parents and one sibling. The frameshift mutation was not found in the family's youngest kid.
The discovery of the genetic substrate in this patient verified the clinical diagnosis of Jervell and Lange-Nielsen syndrome, allowing us to provide him with proper management and genetic counseling. Furthermore, this discovery adds to our knowledge of genetic diseases in the Moroccan community.
Diagnosis and treatment
When JLNS is suspected in a child with congenital deafness and syncopal episodes, the QTc interval is measured to see if it is longer than 500 milliseconds. The diagnosis is confirmed when pathogenic mutations in KCNQ1 or KCNE1 are found. A single-gene test, a multigene panel, or complete genomic testing can all be used to find the genes. The degree of the disease should be determined once the diagnosis has been made. A professional hearing evaluation, geneticist consultation, complete blood count to check for anemia and detailed family history should all be included in the evaluation.
A beta-blocker is the first-line treatment for syncope, cardiac arrest, and sudden death. Metoprolol has been demonstrated to be inferior to propranolol and nadolol in preventing cardiac events. The most widely accepted opinion is that nadolol is the best option. An implanted cardioverter-defibrillator (ICD) should be placed in patients with a history of cardiac arrest. ICDs are also used in patients considered to be at high risk.
A QTc interval of more than 550 milliseconds, syncope before the age of five, and a guy older than 20 years with the KCNQ1 pathogenic variation are all symptoms observed in high-risk individuals.
References
- Pabba K, Chakraborty RK. Jervell and Lange Nielsen Syndrome. [Updated 2022 Apr 22]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK537300/
- Matsuda, S., Ohnuki, Y., Okami, M., Ochiai, E., Yamada, S., Takahashi, K., Osawa, M., Okami, K., Iida, M., & Mochizuki, H. (2020). Jervell and Lange-Nielsen syndrome with novel KCNQ1 and additional gene mutations. Human genome variation, 7, 34. https://doi.org/10.1038/s41439-020-00121-x
- Faridi, R., Tona, R., Brofferio, A., Hoa, M., Olszewski, R., Schrauwen, I., Assir, M., Bandesha, A. A., Khan, A. A., Rehman, A. U., Brewer, C., Ahmed, W., Leal, S. M., Riazuddin, S., Boyden, S. E., & Friedman, T. B. (2019). Mutational and phenotypic spectra of KCNE1 deficiency in Jervell and Lange-Nielsen Syndrome and Romano-Ward Syndrome. Human mutation, 40(2), 162–176. https://doi.org/10.1002/humu.23689
- Adadi, N., Lahrouchi, N., Bouhouch, R., Fellat, I., Amri, R., Alders, M., Sefiani, A., Bezzina, C., & Ratbi, I. (2017). Clinical and molecular findings in a Moroccan family with Jervell and Lange-Nielsen syndrome: a case report. Journal of medical case reports, 11(1), 88. https://doi.org/10.1186/s13256-017-1243-1
- Zhang, M., D'Aniello, C., Verkerk, A. O., Wrobel, E., Frank, S., Ward-van Oostwaard, D., Piccini, I., Freund, C., Rao, J., Seebohm, G., Atsma, D. E., Schulze-Bahr, E., Mummery, C. L., Greber, B., & Bellin, M. (2014). Recessive cardiac phenotypes in induced pluripotent stem cell models of Jervell and Lange-Nielsen syndrome: disease mechanisms and pharmacological rescue. Proceedings of the National Academy of Sciences of the United States of America, 111(50), E5383–E5392. https://doi.org/10.1073/pnas.1419553111
- Winbo, A., Stattin, E. L., Diamant, U. B., Persson, J., Jensen, S. M., & Rydberg, A. (2012). Prevalence, mutation spectrum, and cardiac phenotype of the Jervell and Lange-Nielsen syndrome in Sweden. Europace : European pacing, arrhythmias, and cardiac electrophysiology : journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology, 14(12), 1799–1806. https://doi.org/10.1093/europace/eus111
- Tranebjærg, L., Samson, R. A., & Green, G. E. (2002). Jervell and Lange-Nielsen Syndrome. In M. P. Adam (Eds.) et. al., GeneReviews®. University of Washington, Seattle.
- Jervell and Lange-Nielsen syndrome. [Online] Medline Plus. Available at: https://medlineplus.gov/genetics/condition/jervell-and-lange-nielsen-syndrome/#frequency
- JERVELL AND LANGE-NIELSEN SYNDROME 1; JLNS1. [Online] OMIM. Available at: https://www.omim.org/entry/220400#:~:text=the%20KCNQ1%20gene.-,Description,Lange%2DNielsen%2C%201957).
- Jervell and Lange-Nielsen syndrome. [Online] Orphanet. Available ta: https://www.orpha.net/consor/cgi-bin/OC_Exp.php?Expert=90647&lng=EN
Last Updated: May 3, 2023