Thymine, which is often abbreviated as T or Thy, can also be referred to as 5-methyluracil. Thymine is one of the pyrimidine bases found in the nucleic acid of deoxyribonucleic acid (DNA), along with adenine, guanine, and cytosine (A, G, and C, respectively). These bases are the building blocks of DNA and all life forms on earth.
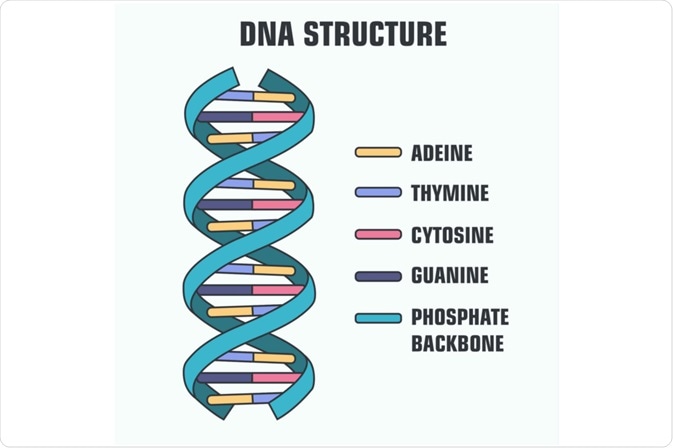
Image Credit: ShadeDesign / Shutterstock.com
Albrecht Kossel and Albert Neumann discovered thymine in 1893 when they successfully isolated thymine from the thymus glands of calves for the first time in history.
Chemical structure
The chemical structure of thymine includes the ring-shaped pyrimidine molecule, which is a similarity shared by each of the nucleobases.
During the formation of DNA, thymine and adenine are always paired together by the force of two hydrogen bonds, which creates a stable nucleic acid structure. In a comparable fashion, guanine and cytosine bind together during the generation of DNA. Under specific conditions, such as exposure to ultraviolet light, thymine dimers may also occur, although this is much less common than the thymine-adenine pairing. In most cases, thymine is not present in ribonucleic acid (RNA) structures, as it is replaced by uracil.
The scientific name of thymine, 5-methyluracil, implies that it can be derived through the methylation of uracil at the position of the 5th carbon. Specifically, in the chemical structure, this means that a methyl (-CH3) branch is added to the pyrimidine ring.
Phosphorylation
When combined with deoxyribose, thymine forms the nucleoside deoxythymidine, which is more commonly known as thymidine.
Thymidine can undergo phosphorylation, during which phosphoric acid groups are added to this substance to form thymidine monophosphate, thymidine diphosphate, or thymidine triphosphate, depending on the number of phosphoric acid groups added.
Mutations and cancer
If the natural chemical structure of thymine is defective, the DNA becomes mutated, which can lead to the formation of melanoma.
A common cause of DNA mutation involving thymine is the response to ultraviolet light. Light waves at this frequency can cause thymine bases that are located adjacent to each other to form a dimer. As this is not the natural structure of the DNA, this can alter the functionality of the DNA and may result in deregulation of cell growth, which can lead to the production of potentially cancerous cells.
5-fluorouracil is a common cancer treatment that targets thymine by acting as a metabolic analog of thymine in the synthesis of DNA. If 5-fluoruracil is incorporated into the DNA of actively dividing cancer cells, the synthesis of new DNA and cancer cells is inhibited.
Thymine in space
In 2015, NASA scientists reported evidence that thymine could successfully be produced in laboratory conditions that resembled outer space.
Meteorites often contain chemical substances such as pyrimidine polycyclic aromatic hydrocarbons; therefore, this observation was used as a starting base in the laboratory. Under simulated conditions, the scientists were able to produce thymine, uracil, and cytosine. As these nucleobases play a considerable role in the building blocks of life, their successful formation under space-like conditions raised important questions as to whether some forms of life could be supported in outer space. To date, this is purely theoretical but further research is needed.
References
Further Reading
Last Updated: Mar 18, 2021