Wnt signalling is an important, evolutionarily conserved signalling pathway that determines cell fate decisions and tissue patterning during embryonic development and later development.
The pathway is activated by Wnt proteins, a family of 19 highly conserved molecules that are secreted as glycoproteins that form concentration gradients across developing tissues.
Wnt signalling pathway. Image Credit: Meletios Verras / Shutterstock
Wnt signalling regulates cell fate decisions, neural patterning, cell migration, cell polarity and organ formation during embryonic development. Wnt proteins are also known to regulate the behaviour of different types of stem cells, keeping them in a state of self-renewal.
The Wnt signalling pathway plays a vital role in embryogenesis and its dysregulation has severe consequences for a developing embryo. Defective Wnt signalling is known to lead to several human pathologies, including cancer of the colon, breast and skin, skeletal effects and birth defects including spina bifida.
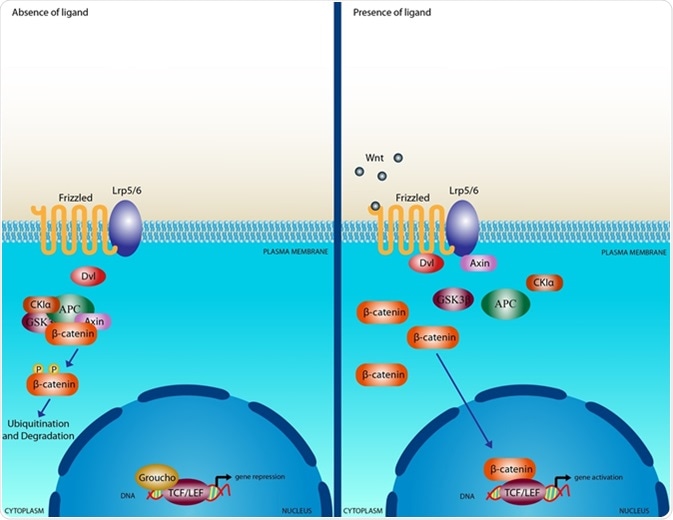
The Wnt/β-catenin signaling pathway
Wnt Proteins
Wnt proteins are globular molecules with a molecular weight of around 40 KDa. The N-terminus is made up of α-helices that have five disulfide bonds and the C-terminus is made up of two β-sheets that have six disulfide bridges.
Prior to their secretion into the extracellular environment, Wnt proteins are subjected to post-translational modification. Wnt undergoes glycosylation and palmitoylation by a protein called Porcupine and is then transported by Wntless proteins to the plasma membrane. An absence of either Porcupine or Wntless proteins stops Wnt proteins being released, which leads to the development of birth defects.
Wnt proteins induce signalling when they bind to a membrane receptor complex comprising a Frizzled (Fz) G-protein-coupled receptor and a lipopolysaccharide-like receptor protein 5/6 (LRP5/6). The Fz protein contains seven transmembrane-spanning domains and a large extracellular N-terminal domain that serves as a platform for Wnt binding.
Extracellular Wnt proteins activate intracellular signal transduction cascades downstream of the Fz receptor. These are classified as the canonical, or Wnt/β-catenin pathway, and the non-canonical, or β-catenin-independent, pathway. The non-canonical pathway is further divided into the Planar Cell Polarity and the Wnt/Ca2+ pathways.
The Canonical Pathway
Beta-catenin migrates into the nucleus, where it associates with T cell factor (TCF) and l ymphoid enhancer factor (LEF) transcription factors . This results in Wnt target genes being transcribed. When there is no Wnt signal, the TCF/LEF transcription factors interact with inhibitors of Wnt transcription, which supresses Wnt signalling.
This inhibitory effect is mediated by interactions with histone deacetylases, which prevents transcription of genes . Inhibitors of beta-catenin are ICAT and duplin, which directly interact with beta-catenin to prevent it associating with TCF/LEF.
The Non-Canonical Pathway
The non-canonical pathway or β-catenin-independent pathway is further divided into the Planar Cell Polarity pathway and the Wnt/Ca2+ pathway.
The planar cell polarity pathway
Wnt signalling via Frizzled receptors directs asymmetric cytoskeleton organization and cell polarization by causing modification of the actin cytoskeleton. A key Wnt signalling protein called Dishevelled initiates two independent pathways that induce activation of two GTPases, Rho and Rac. Rho activation leads to the activation of a Rho-associated kinase called ROCK. Rac activation induces Jun Kinase activity.
The Wnt/Ca2+ pathway
Activation of the Wnt signalling pathway via Fz receptors can also result in the release of intracellular calcium. The Frizzled co-receptors Knypek and Ror2 are activated in this pathway. Other activated intracellular messengers in the pathway are G-proteins, phospholipase C and protein kinase C. The increase in calcium can activate a calcium/calmodulin-dependent protein phosphatase called calcineurin, which results in dephosphorylation of a transcription factor called NF-AT (Nuclear factor of activated T-cells) and its accumulation in the nucleus.
Further Reading
Last Updated: Jul 21, 2023