Silvio Aime is the Professor of General and Inorganic Chemistry in the degree course of Biotechnology at the University of Turin. He is also head of the Molecular Imaging Center.
The Molecular Imaging Center - University of Turin - Silvio Aime
Please can you outline the aims of the Centre of Molecular Biotechnologies of the University of Torino?
The Center of Molecular Biotechnology of the University of Torino was founded about 10 years ago by biologists from the Faculty of Medicine; a few chemists (our group essentially) from the Department of Chemistry and few bioinformaticians from the Physics department.
The reason for founding this interdepartmental center relied essentially on the need to join efforts in order to tackle relevant medical needs. Therefore, for us it was a unique opportunity and we are definitely happy to have done it.
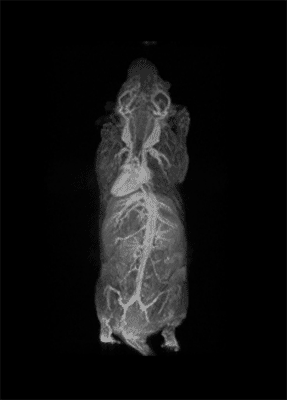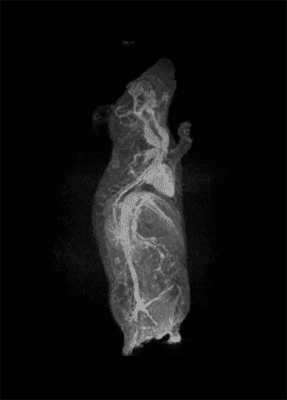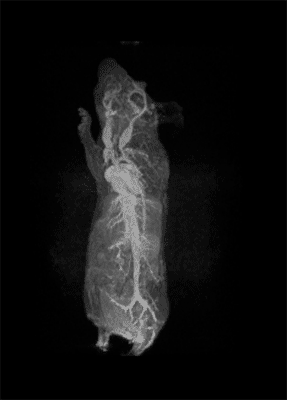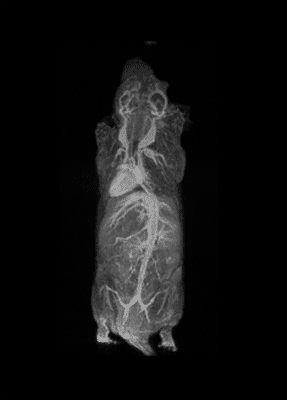
How important is molecular imaging in tackling un-met medical needs?
Molecular imaging aims at the visualization of molecules or molecular events that occur at the cellular level. Clearly it also allows the possibility of looking inside the biochemical pathway at the cellular level and therefore enables us to look at the onset of diseases well before they are resolved into structural change. This is what we are currently doing with it.
Ever more important I think is that Molecular Imaging, by allowing the visualization of the proper biomarker that are the hallmarks of diseases of interest, would allow one to assess the efficacy of a therapeutic treatment well before we can see any structural anatomical change.
So, in the year of molecular medicine, molecular imaging is really representing the level of diagnostic tool that we need to tackle it.
Molecular imaging is really a truly interdisciplinary science. It needs biologists in order to find biomarkers related to a specific disease. It needs chemists that will design and synthesize the proper probe in order to target this biomarker of interest. It needs imaging scientists in order to take maximum advantages from available imaging modalities. Of course it also requires physicists and engineers to develop the proper scanner with the sensitivity and specificity.
So, I think that molecular imaging is the way to merge all these skills in order to tackle, in the most suitable way, the issues for early diagnosis and for monitoring a therapeutic effect one given treatment that has been undertaken.
Talking Multimodality with Silvio Aime
How has multimodality impacted your workflow?
I grew up with NMR and then I passed to MRI but what I found most exciting in the last 10 years or so is really the possibility to do multimodality imaging.
So, to see how the different ways that we have to interrogate our living system and using the proper imaging modality for the advantages that it can give and then to merge the information coming from the different imaging modalities – this is the most exciting possibility we have in the field.
This is what really makes molecular imaging a strong tool in the hand of the scientist to reveal new aspects of what is recorded inside living systems.
What excites you most about the current applications of molecular imaging?
MRI is based on NMR Spectroscopy, which is the best language to talk to nature. We know how it is important to exploit different field strengths in order to extract maximum information from the system that we are investigating.
Most people think that we just have to move to high-field in order to make the most of the information and of course we know that moving to high-field has its drawbacks first of all because it is quite costly to move any Tesla more in our magnetic field strength.
I think that also the low-field, let’s say the low/intermediate field to work at the 1 Tesla for instance as in the case of this beautiful Icon system, this gave us unique possibilities. Anytime we use a gadolinium-based agent or more in general a paramagnetic basic agent the best field is the 1 Tesla.
So, we can show that there is a real advantage of working at 1 Tesla in comparison to for instance 7 or 9 Tesla when one is using a relaxation enhancer to target a specific epitope in the region of interest.
In what ways has the field changed over time?
We have changed a lot in terms of the way we look at imaging proper design and testing in the last decade.
Whereas until the late 90s we were essentially attracted by the development of contrast agents for MRI application and being restricted in some domains, in particular with the vascular and the extra vascular domain, because it is always very difficult to reach enough sensitivity with MRI contrast agents entering into cells.
The possibility to use complementary imaging modality was our task in the last 10 or so years. And this changed quite drastically the structure of our group, because we have to get users quickly used to the basic principles in order to have efficient imaging reporter for the other imaging modalities.
This of course definitely enriched the capability of our center and we are very pleased to have done it.
So, our students now pass from one imaging modality to another to get used to the different classes of imaging reporter agents.
The Future of Molecular Imaging with Silvio Aime
What do you think the future holds for molecular imaging?
I think the future of molecular imaging is very bright. The possibility to look inside the body with eye spatial resolution and to look at the biochemical processes that are related to the onset of the diseases open a beautiful scenario in order to fit all the terrific achievements that have come from biology and from molecular medicine.
I think that the possibility molecular imaging is offering to look in depth inside the living system and any development in this direction that is expected for the forth coming year will make this science even more important than it is now.
Clearly we are looking for the clinical transaction of several of the achievements that have been done in the past decade in the field of clinical studies.
What barriers still need to be overcome in order to achieve these aims?
To some extent I think we are on the top of an iceberg where the potentiality can be dramatically improved by looking at small objects, such as live cells, because in principle we always aim to go down to the single cell information.
So, the increase of sensitivity of the imaging probes and the imaging technology are the barrier we have to face.
Of course we are already in a very good position and we think that some of the molecular imaging procedures are already ready to be translated to the medical world; but the improvement of sensitivity of the imaging probes as well as the improvement of sensitivity of the imaging scanners are actually the barriers we have to overcome.
What are your further research plans?
We are working in collaboration with several industries and this helps us substantially with the recognition of the mathematical need, and therefore allows us to tackle real important programs.
I think that the main direction we are putting our efforts into is one way to get more insight into metabolic imaging. To know the metabolic processes that occur at a cellular level is the way to really see the functioning of cells.
On another level, an important direction we are pursuing is deals with the development of theranostic agents to allow the clinician to simultaneously do diagnosis and therapy.
Then in the direction to improve, through the use of imaging, drug delivery and therefore to give the clinician the possibility to deposit the amount of drug at the targeting site that is really necessary and to look at the effect of the process of release and possibly the effect of the drug.
As well at another level is to support the surgeon to do his job better by using the protocol of molecular Imaging. This leads into a new emerging field of imaging-guided surgery where we use the same principle of targeting molecular events at the cellular level into the surgery theater. Therefore, the surgeon can really see residual tumor cells or a way to recognize the nerves or to recognize vessels, whereas, currently this is very difficult.
Using the ICON™ Powerful MRI, Simplified.
For more information please visit: Bruker ICON™