Please can you give a brief introduction to the Center for Advanced Biomedical Imaging (CABI) and its multidisciplinary nature?
My name is Mark Lythgoe. I'm Professor of Biomedical Imaging, as well as Director of the Center for Advanced Biomedical Imaging, here at University College London.
The Center for Advanced Biomedical Imaging or CABI is only about six years old, so we're a relatively new department. When you walk through, you're going to meet physicists, mathematicians, biologists, pharmacologists and anatomists. We even have an artist in residence.
The idea is to create a real playground where we can bring lots of disciplines together into one space and surround them with technology they can access, in the hope that we will be able to crack those problems or questions that we've been perhaps struggling to answer while working just within our own single disciplines.
I don't believe nowadays that one single discipline or one single modality can crack the really tough biological questions that we have.
For me, perhaps one of the most remarkable aspects of being at UCL is that we are right in the heart of London. Only two or three hundred meters away are all the shoppers walking up and down Tottenham Court Road, yet, here, we have one of the most advanced experimental imaging centers that goes completely unnoticed, where we're able to bring lots of different imaging modalities together to crack what I think are some of the most taxing and difficult biological and medical problems existing at this moment in time.
The Center for Advanced Biomedical Imaging at UCL
Could you please outline some of the projects you are working on?
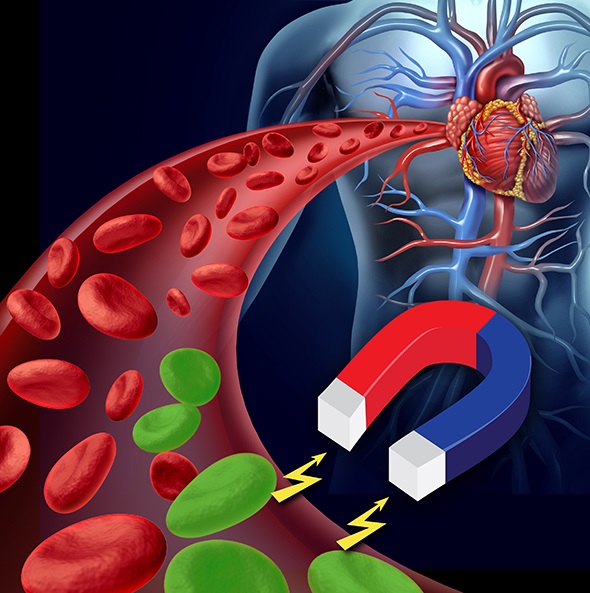
With fifty people and departments, it would take me probably a couple of hours to go through all the projects, but if I could pick some of my favorites the first one would be magnetic targeting.
One of the first things we did when we came to CABI was to try and image individual cells. We would inject cells and try to use MRI to track their route around the body.
We put little iron oxide particles in them and what occurred to us the time was that we could maybe make them magnetic and use the MRI scanner instead for imaging, to actually grab the particles and cells and steer them to sites of injury.
That’s what we've been doing - injecting cells with these tiny iron oxide particles, which we magnetize so we can use the MRI scanner to direct them where we want them to go. That gets you thinking about imaging in a completely different way, not just as an imaging tool, but now as an interventional and surgical tool.
 Shutterstock / Lightspring / Viktorija Reuta
Shutterstock / Lightspring / Viktorija Reuta
The other two projects I like are more recent. MRI isn't particularly sensitive and although it’s very good for looking at structure and function, it's not very good at looking at individual molecules.
Simon Walker Samuel came up with a technique called glucoCEST, where we're able to give patients a bottle of Lucozade, sensitize the MRI scanner to the glucose in that Lucozade and because tumors readily take up glucose, they light up on the MRI scanner. So, it allows us to see those very subtle molecular processes that we certainly weren't able to get a handle on ten or fifteen years ago.
We were also able to create a map of that across the tumor and, as we developed it here, we were able to translate it across to the hospital and within about two or three weeks of finishing the project, we were using the technique on patients. It’s lovely to see MRI techniques move to the hospital straight away and that is one of the big advantages of MRI.
The other significant challenge that we face is cell tracking. There's a lot of interest in regenerative medicine and within regenerative medicine, there is a T cell problem.
People use T cells as a therapy here at the cancer institute, targeting the tumors and using the T cells to eat the tumors up. However, they also wanted to track the cells as they target tumors and we've been genetically modifying the cells so that when they are injected, they take up a radioactive isotope, light up, and emit radioactivity. We can then get a three-dimensional map by looking at the distribution of the cells.
Traditionally, imaging techniques used to live in medical physics departments, where physicists worked on them, but now we're seeing biologists, cell biologists and developmental biologists looking at cellular processes and it's those advances that are really enabling imaging to move forward in a way that it hasn't previously been able to.
How important is multimodality at CABI?
Across CABI, we have eleven or twelve imaging modalities and I think it's quite unique in that respect. We have optical devices, ultrasound, magnetic resonance, nuclear, and within those, we have lots of different strands.
On one level, we're trying to go across scale - from embryos, whether they be mice or zebrafish, for example - all the way through to whole organ or trying to look at individual cells and seeing how they perform in a system or organ.
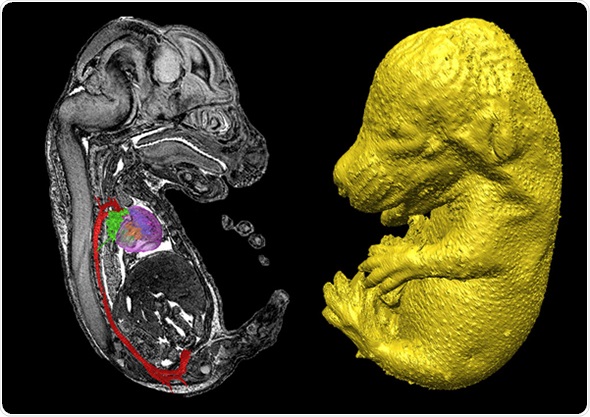
I think the difficulty is that there is no one technique you can use to completely understand a particular disease or process you are looking at. You have to be able to image across different scales and the only way you can do that is by using different modalities.
Sometimes, we'll combine things such as the information obtained about anatomy or structure from MRI, with information about function obtained from some of the optical techniques.
Part of this idea was born out of my own frustration that, within certain departments such as radiology, there was an element of competition between MRI and ultrasound over which was the best technique.
I wanted to get around that, as I thought it was creating boundaries and wasn't productive for a research environment. So, when we came here, it was genuinely about trying to mix up not only the different disciplines, but also the different techniques.
What are the advantages of this strategy?
Let me tell you about some of the disadvantages to start with, because there are always disadvantages. One, you have to know about three or four different techniques to be able to decide which one you're going to use. Then, you have to know the advantages and disadvantages of each one of those.
Also, a lot of space is needed to put lots of different equipment in and there’s usually a lot of regulation and legislation surrounding that, if you're going to move things from a radioactive lab into an MRI lab. It's definitely harder work.
So, the question is, what do we gain from that? Well, the problem is that there is no one technique that can do everything. However how hard you work at it, you are not going to get all the information you can, nor all the imaging information about a particular pathology or disease using just that one technique.
If you're going to look at genuine function, cellular processes, anatomy and structure across a disease or across a therapy, then you need to use different techniques and within that, you're going to have to use different elements of the technique.
You have to be able to not only look at the beating of the heart, which is the function, but also at the molecular processes within that, which you may need to use a nuclear medicine or an optical technique for.
I think only then can you start to get a real picture of that particular organ or that particular pathology, and only then can you really advise the clinical departments about sensitivity. If you've genuinely compared two techniques, you have a really good feel for what's going to work for you, as well as what's going to work in the clinical environment. Unless we put these two techniques side by side and genuinely look at them, we're never going to be able to give good, sound advice.
Which disease areas do you focus on at CABI?
There are no particular disease areas we focus on, we are pretty broad. I think it's about trying to ask exciting questions and it doesn't matter whether it's the liver, the kidney, the brain or the heart. There are exciting questions within each of those organs or within each of those fields or disciplines.
We have a cardiovascular group, an oncology group, a neurology group, and a cell tracking group that moves across all of those, looking at cells in the brain and in the heart. We also have a chemistry group because if you're making new genetic reporters or new contrast agents for imaging, again, those could equally apply to the brain and the heart.
What has been interesting is that you can sometimes take an idea arising from cancer research and then apply it to the brain. That has been a real eye-opener for all of us.
The disadvantages are that, at times, you feel a little bit out of your depth because you've got so many different things going on. However, as those individual groups grow and become quite strong and independent, you can get the cardiovascular people to interact with the brain people, so that information can move fluidly between the two disciplines.
Once you can do that, you can start to maximize efficiency across the imaging lab, which then yields ten times the amount of output, data and knowledge. It really is in those orders of magnitude if you get that right interdisciplinary approach across those fields.
What are your reasons for using MRI?
We use MRI, partly because I did my Ph.D. in MRI and partly because it is the cornerstone of the department and I think that is the case for most imaging departments.
Labs will have MRI as their gold standard. The technique is completely non-invasive, which makes it very easy to carry out in studies of volunteer and patients.
For us, we use MRI as our building block, and then expand outwards from that to the opticals, ultrasound and nuclear medicine.
For example, we have recently been working with gold particles in an optical environment. We then tried them out to see if they worked on the MRI scanner and then from the MRI scanner, we actually used them in CT.
Having these different devices allows us to look at particular contrast agents, particles, cells or pathology in a slightly different way. Basically, the cornerstone is the MRI, and then we build on what we find from there.
What are the advantages of working with a desktop system?
The first thing that you realize when you get a piece of kit, is that you've got to get it into the lab and installed. A big 9.4T scanner is going to take you some weeks to get set up, running, and working, whereas when the bench-top system came, we were up and running certainly within a week and if not, within a few days.
The next question is, where are you going to put it? For every piece of kit, you've got to find a space to put it in and the desktop’s got a lovely small footprint.
Probably the biggest advantage for us was being able to locate it in a different imaging environment. The MRI stands at one end of the department, but the Icon stands within the nuclear imaging capability.
There are two reasons for that. One, the biologist working within that environment can walk freely around the bench top system because they don't have the high field and therefore don't have to worry about their cards or instruments.
Secondly, because of the footprint, we can closely locate it between the MRI and the nuclear medicine systems because there are no interactions between the two.
That means we can get the anatomy information from the MRI and then quickly take the animal over and get the information about function from the nuclear medicine systems. This set-up offers us ease of access, ease of location and ease of combining two or more modalities.
Working with Compact MRI
What are the specific advantages of working at low field?
The most obvious advantage of working at low field is the practical side of it. Someone can just walk straight up to it, unlike when working with high field systems, where you have to make sure that you've got no metal on you.
Also, it's safe, easy to access and easy to use. From my point of view, those are the three things that are very attractive about having it in the nuclear imaging facility.
Of course, there are disadvantages of using low field. For 90% of the things that we do, it's never going to be as good as our 9.4T scanner. The question is whether it is good enough, because I sometimes think the 9.4T and high field systems are overkill.
If all you want to do is simply measure the volume of a tumor, spending hundreds of pounds an hour to obtain just one image is not cost effective. Since the low field systems are cheaper, you can get information that's of an acceptable quality for that study, at a lower price.
It doesn't mean to say the throughput is necessarily any faster, but it's very attractive to a biologist who just wants to measure the size of a tumor. They can simply put the mouse in, find out the tumor volume and then it's onto the next mouse.
I think it's early days in terms of trying to decide exactly where the killer application is, but when we've compared the 9.4T with the Icon, I’ve been pleasantly surprised when it comes to measuring the volume of tumors.
You are strongly involved in the Crick institute; please can you explain the vision behind this exciting project?
As the Director of the Crick, Paul Nurse, would put it, it is a place without boundaries, a place without walls.
We will not have traditional departments and the idea is to bring the best of the best into the largest biomedical institute we have and to see if breaking down those boundaries will enable us to come up with ideas that might crack the problems we haven't been able to crack so far.
I think there will be a completely different philosophy and approach to the type of science that we're used to doing.
What do you hope the future holds?
I hope the future will bring a change in technology. The low field systems are just so easy to work with and so easy to use that ideally, I'd like to be able to get all the information I get from the very expensive and difficult to use 9.4T, out of something like the Icon.
There is going to have to be a shift in technology in order to get that kind of information, whether that be in terms of electronics, changing the coil design, or refining the magnet design. I don't know if that's possible and we don't think it is possible, but if you asked me what my dream is, that would be it.