Biotechnology broadly refers to the utilization of cell cultures and other biological systems to manufacture or develop products.
Biotechnology plays a key part in pharmaceutical manufacturing – in recent years, this has increased quickly at a growth rate which is double that of conventionally produced pharmaceuticals.1 Biopharmaceutical sales made up around 30% of all drug sales in 2018.2
Detailed, accurate monitoring of metabolic processes in bioreactors is vital for the optimization and development of processes in biotechnology. The development of effective Process Analytical Technology (PAT) is becoming more and more crucial as the biotechnology industry grows.
Raman spectroscopy has grown from a novelty to a proven global PAT strategy for bioprocessing in recent years. Based on the webinar delivered by Senior Application Scientist Justin Moretto, an introduction to Raman spectroscopy is provided in this article, and the quantifiable value it can produce for biotechnology organizations is demonstrated.
How Raman spectroscopy works
Raman spectroscopy is a type of vibrational spectroscopy employed to identify different compounds by monitoring the vibrational characteristics of the molecular bonds present in a sample.
The photon’s energy temporarily manifests as vibrational energy in one of the molecule’s chemical bonds when a molecule absorbs a photon in the infrared/visible/ultraviolet range. A photon with the same frequency is usually released after the bond “relaxes”.
However, occasionally, a small amount of energy is “left behind” in the molecule, and a photon with a slightly lower frequency (which corresponds to its loss of energy) is emitted. This is known as inelastic scattering, and Raman spectroscopy concerns itself specifically with these inelastically scattered photons.
The tiny alterations in frequency between the emitted and absorbed photons correspond to specific vibrational modes of the chemical bonds, and so correspond specifically to both the bond orientation and bond type.
As inelastic scattering is a “linear” effect (the amount of inelastically scattered photons scales in proportion to the amount of a bond present), even in complex solutions Raman spectroscopy can supply a quantitative analysis of the molecules present in a sample.
Raman is non-destructive and requires no sample preparation for PAT applications. This means that Raman analysis can be carried out at regular intervals to give an unprecedented insight into batch evolution in aqueous cell culture solutions.
Quantifying the value of Raman spectroscopy
Raman provides multiple significant returns on investment within the framework of PAT. Where income is generated is where the largest ROIs are seen: in manufacturing. Yet, Raman still provides significant value in development organizations.
Raman eliminates analyzer consumable costs since it does not need consumables. It decreases the contamination risk that is associated with sampling and reduces the human capital requirements for data collection.
Yet, the true value of Raman is in the quality and quantity of the data it produces, and in the utilization of this data to characterize and optimize processes precisely. The high-cost, high-value nature of the biotechnology industry means that through slight optimizations, significant value can be realized.
For instance, if an organization runs 52 batches a year, a productivity increase of around 2% would enable the elimination of one batch per year, which could save around $3 million per year. These types of optimizations can be readily achieved by utilizing the data supplied by Kaiser Raman.
There are several other advantages which are more difficult to quantify, but that are still significant, for example, the avoidance of facility expansion costs, the value of reduced duty cycle on equipment, or even the ability to form strategic partnerships by utilizing newly found excess capacity.
Source: https://www.biophorum.com/download/in-line-monitoring-real-time-release-testing-in-biopharmaceutical-processes-prioritization-and-cost-benefit-analysis/?49433
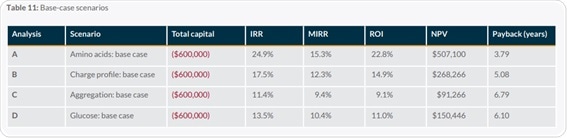
Demonstrated success
By looking at examples throughout peer-reviewed literature, the value of Kaiser Raman is made clear. Researchers could ‘reprogram’ a cell culture in a bioreactor to consume lactate preferentially instead of glucose in a study conducted by a team at Biogen. This was achieved by using an automated system controlled by Raman spectroscopy.3
Raman measurements were taken to continuously monitor glucose levels while restricting glucose supply to the cells. This was done to ensure that they did not drop so far that the cells would die. Whenever levels of glucose fell to a predefined lower limit (0.5 grams per liter) an automated glucose feed was activated.
Similarly, the glucose feeds were ended entirely when lactate levels went above 4 grams per liter. By utilizing this system, the researchers could restrict glucose levels but ensure that they did not fall low enough to destroy the batch.
The cells began to exhibit a marked change in behavior after nine days, as the cells started to preferentially consume lactate. The resulting reduction in glucose and lactate accumulation not only led to higher cell viability and cell density, but it also increased productivity by 85%.
Perhaps more crucially, the Critical Quality Attributes (CQA) of the process became more predictable. A team at Pfizer used Raman spectroscopy for inline amino acid monitoring in 2018.4
By developing a chemometric model capable of measuring concentrations of tryptophan, tyrosine, and phenylalanine with low RMSEP values, the team could show the potential of Raman spectroscopy data for quantifying amino acid levels.
The difference in performance between Mass Spectrometry (MS) and Raman analysis was shown by researchers monitoring bioreactor off-gas to calculate real-time oxygen uptake and carbon dioxide evolution rates (OUR and CER respectively).
The Kaiser Raman system used by the team monitored four simultaneous gas streams over a five month period, and the results were compared to measurements taken by a mass spectrometer.5 The Raman system recorded more than five times as much data as it was capable of making uninterrupted measurements.
Furthermore, the Raman system drifted by 0.00%, while the MS system drifted by 5% accuracy over the course of the study (in line with the manufacturer’s expectations). Over the last decade, Raman has changed from a development curiosity to one of the key drivers of PAT in biotechnology.
Kaiser Raman systems provide nearly unlimited possibilities in data collection, enabling automation of bioprocesses and fast and precise characterization.
References and further reading
- Wurm, F. M. Production of Recombinant Protein Therapeutics in Cultivated Mammalian Cells. Nature Biotechnology 2004, 22 (11), 1393–1398. https://doi.org/10.1038/nbt1026.
- Raman spectroscopic techniques for biotechnology and bioprocessing - European EvaluatePharma® - World Preview 2019, Outlook to 2024. (2019).
- Matthews, T. E. et al. Closed loop control of lactate concentration in mammalian cell culture by Raman spectroscopy leads to improved cell density, viability, and biopharmaceutical protein production. Biotechnol. Bioeng. 113, 2416–2424 (2016).
- Bhatia, H., Mehdizadeh, H., Drapeau, D. & Yoon, S. In-line monitoring of amino acids in mammalian cell cultures using Raman spectroscopy and multivariate chemometrics models. Eng. Life Sci. 18, 55–61 (2018).
- Kaiser Raman Analyzers by Kaiser Optical Systems, Inc. Available at: https://www.endress.com/en. (Accessed: 18th August 2020)
About Kaiser Optical Systems, Inc.
Kaiser Optical Systems, Inc. (Kaiser), an Endress+Hauser company, is the global leader in Raman spectroscopic instrumentation for laboratory, process, and manufacturing environments. Our solutions harness the powerful analytical information of Raman Spectroscopy to help our customers understand, measure, and control their chemistries.
As a trusted partner in Raman for over 30 years, Kaiser has a long history in production, including GMP manufacturing, with many proven successes. Our unmatched expertise, high quality solutions, and exceptional customer service sets Kaiser far above any other Raman option in the marketplace. Kaiser Raman technology is currently used throughout the chemical, food and beverage, oil and gas, pharmaceutical, and biopharmaceutical industries to optimize process efficiency and deliver quality products. Kaiser’s manufacturing and headquarters facility is in Ann Arbor, Michigan.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.