Ultraviolet light is part of the electromagnetic spectrum between 100-400 nm. As seen in Table 1, the ultraviolet spectrum is split into three categories, UVA, UVB and UVC. On Earth, UVC is blocked by molecular oxygen in the atmosphere, yet UVA and UVB readily travel to the Earth’s surface.
Someone who has experienced sunburn can testify to the dangerous power of ultraviolet light. Although ultraviolet can be harmful, from direct applications to sanitation to our fundamental understanding of how UV affects our biology, it has helped improve human health.
Table 1. Ultraviolet Wavelengths.
| UV Common Name |
Wavelength (nm) |
UVP Crosslinker, CL-3000 Wavelength (nm) |
| UVA |
315-400 |
365 |
| UVB |
280-315 |
302 |
| UVC |
200-280 |
254 |
| Vacuum UV |
100-200 |
NA* |
*Analytik Jena does not manufacture Vacuum UV products
Ultraviolet light has been utilized in applications for the destruction of microorganisms in the air, preventing the growth of microorganisms on surfaces, and sterilization of liquids for more than 120 years.
For instance, since 1877, humans have employed ultraviolet light for food preservation/sterilization1; ultraviolet light started being used in the 1930s in operating rooms to sterilize the air2; and ultraviolet light has been employed to sterilize drinking water on and off since the early 1900s3.
The same properties which give ultraviolet light its effectiveness in the applications previously mentioned, make it dangerous to biological organisms. Upon UV exposure, at the molecular level, adjacent pyrimidines in a nucleic acid strand can undergo dimerization, which happens predominantly between thymine bases4–6.
Researchers have taken advantage of this property of ultraviolet light as a tool in studying genetic mutations for almost 100 years.
In fact, UV-induced, not X-ray induced8 mutations would be tied to Herman Muller’s landmark 1928 fly experiments had earlier attempts to harness the mutagenic properties of UV light been successful.7 These later earned him the 1946 Nobel Prize—after all, later in his career ultraviolet light became a popular strategy in his lab 9–16.
Yet, researchers were late to identify the optimal wavelength of ultraviolet light which produced the mutagenic properties (i.e. 254 nm or UVC). Researchers found that ultraviolet light produced coagulates in titrated protein solutions17,18. This was done separately and about a decade before Muller’s landmark work.
Later, protein researchers would understand the underlying mechanism that generated this coagulation was the disruption of disulfide bridges and modification of tryptophan residues19. Together, these discoveries paved the way for ultraviolet-based applications in cellular and molecular biology, and beyond.
Since 1993, Analytik Jena has been manufacturing the UVP Crosslinker using UVA, UVB, and UVC light to cover a range of applications, these are summarized in Figure 1.
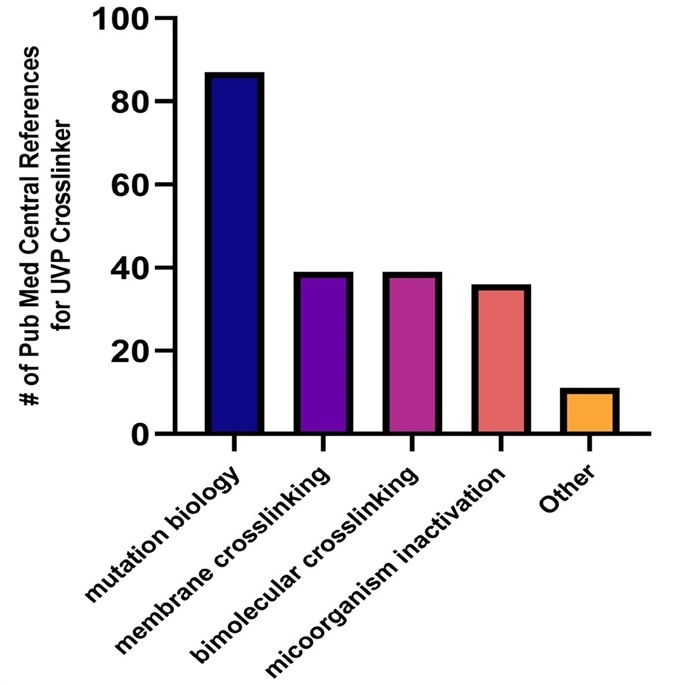
Figure 1. Applications Summary of UVP Crosslinkers from Pub Med Central Using Textspresso. We mined Pub Med Central using Textspresso20 to identify unique hits for UVP Crosslinkers. We identified 212 unique hits, which are classified into broad categories for simplicity.
These applications include:
- Photostability verification
- Mutation biology (survival assays, UV-induced mutations, stress assays)
- Polymer curing (e.g. methacrylate resins, hydrogels)
- Membrane crosslinking (Northern/Southern blotting, EMSA)
- Microorganisms inactivation (e.g. bacteria, fungus, virus)
- Bimolecular crosslinking (PAR-CLIP-seq, pBpa, CLIP-seq, iCLIP-seq, sulfo-SDA, psoralen)
These applications are reliant on reproducible, high-intensity, UV doses. The most recent model from Analytik Jena is the UVP Crosslinker CL-3000, seen in Figure 2A. It is designed with a built-in radiometer which is calibrated to a NIST traceable standard to ensure consistent doses irrespective of time and space.
Furthermore, the CL-3000 can generate a cumulative dose of up to 10 J/ cm2. By having the UV source and reflective housing in close proximity to the sample, highly uniform illumination can be achieved (Figure 2B and 2C).
The center of the illumination surface is 6.0 %, while the coefficient of variation across the entire illumination surface is 17.3 %. Safety of the end-user is crucial, as with all of the instruments from Analytik Jena, so the Crosslinkers have a safety-interlock to prevent accidental UV exposure.
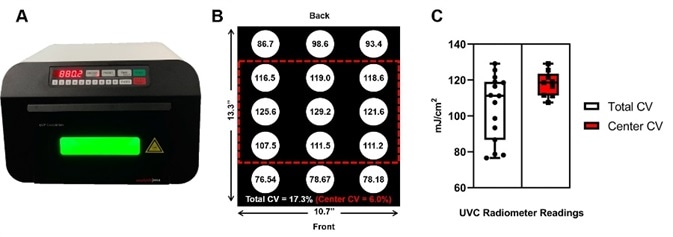
Figure 2. Uniformity of the Analytik Jena UVP Crosslinker equipped with 254 nm UVC bulbs. A) Profile image of the UVP CL-3000 Crosslinker. B) Diagram depicting the position of integrated radiometer readings in mJ/cm2 with dimensions and reported coefficient of variation (CV) calculation for total illumination surface (Total CV) and the center of the illumination surface (Center CV). C) Box-whisker plot summary of radiometer readings. Total CV: Mean=104.9, SD=18.16; Center CV: Mean=117.9, SD=7.1. We recommend users place samples in the center of the illumination surface. Samples placed in the center will receive, at a minimum, the input energy dose. If users want to place samples at the front-most and back-most positions, we recommend purchasing a UVP 254 nm radiometer (P/N 97-0015-02) and sensor (97-0016-01) to calibrate dose.
Operating the Analytik Jena UVP crosslinker CL-3000
The Analytik Jena UVP Crosslinker CL-3000 is capable of user inputs in the form of time or fluence/dose in mJ/cm2. Given that environmental variables and the age of the bulbs can affect UV output, it is recommended that users defer to entering fluence rather than time. Users will discover that both are used in the scientific literature.
Users can simply convert to mJ/cm2 if fluence is reported, this is the unit of measure used for inputs on this instrument. An example conversion can be seen in Table 2, under Fluence.
If the Fluence rate is reported, usually in µW/cm2, this value will be accompanied by exposure time. Users will have to convert Fluence Rate into mW/cm2, an example conversion can be seen in Table 2, under Fluence Rate.
Furthermore, users must convert exposure time into seconds (e.g. 10 minutes = 600 seconds). An example scenario and calculation can be seen below.
Table 2. Important Conversions for Ultraviolet Light Measurements.
| Fluence |
| µJ/cm2 |
mJ/cm2 |
J/cm2 |
J/m2 |
| 1,000,000 |
1,000 |
1 |
10,000 |
| Fluence Rate |
| µW/cm2 |
mW/cm2 |
W/cm2 |
W/m2 |
| 1,000,000 |
1,000 |
1 |
10,000 |
*Values in the literature are most commonly reported as fluence in mJ/cm2 or J/m2. In some cases users report the fluence rate (e.g. μW/cm2), in which case user should use Equation 1 to determine fluence (or dose).
Example scenario and calculation
A researcher exposes the influenza A virus to 1000 uW/cm2 for 10 minutes. Convert the reported UV dose into mJ/cm2.
Equation 1
Fluence Rate (mW/cm2)* Time(sec) = Fluence/Does (mJ/cm2)
1 mW/cm2 * 600 sec = 600 (mJ/cm2)
Placement of samples
It is recommended that users place samples in the center of the illumination surface where the CV=6 % (Figure 2B, 2C). Samples placed in the center will receive the input energy dose at a minimum. Due to edge effects, samples placed at the back-most and front-most positions will receive less than the input dose.
A UVP 254 nm radiometer (P/N 97-0015-02) and sensor (97-0016-01) should be purchased if users want to calibrate the UV dose at the edges. This is to establish the lowest UV dose on the illumination surface of their instrument.
For instance, Figure 2B displays the front-most and right-most position as the lowest dose (78.18 mJ/cm2) when 100 mJ/cm2 is inputted. So, to account for edge effects, the user may consider inputting a higher dose (e.g. 125 mJ/cm2).
Technical data
| Technical Specifications |
CL-3000 |
| Wavelength |
254 nm, 302 nm, 365 nm |
| Bulbs |
6 x 8 Watt |
| Energy |
0000.1 - 9999.9 mJ/cm² (0 - 10 J/cm²) |
| Time |
000:01 - 999:59 mmm:ss (>300J/cm²) |
| Temperature |
15°C - 35°C |
| Humidity |
70% Non-Condensing |
| Altitude |
up to 3,000M (9,842 ft) |
| Sound Level |
≤ 50 dba |
| Housing Surface Temp |
≤ 30°C |
| Startup Time |
< 1 sec |
| External Dim (L x W x H) |
41cm x 40cm x 26.5cm |
| Internal Dim (L x W x H) |
35cm x 27cm x 16cm |
| Weight |
6.8Kg: 15 lb |
| Operating Power |
100 - 115VAC & 230VAC 50/60Hz |
| Certifications |
CE, RoHS (CSA In Process) |
Part numbers
| Part Number |
Description |
| 115V |
230V |
|
| 849-95-0615-01 |
849-95-0615-02 |
UVP Crosslinker (CL-3000), 254 nm |
| 849-95-0615-03 |
849-95-0615-04 |
UVP Crosslinker (CL-3000M), 302 nm |
| 849-95-0615-05 |
849-95-0615-06 |
UVP Crosslinker (CL-3000L), 365 nm |
| 34-0007-01 |
849-057-007 |
Replacement UV tube, 8 watt, 254 nm |
| 34-0042-01 |
- |
Replacement UV tube, 8 watt, 302 nm |
| 34-0006-01 |
- |
Replacement UV tube, 8 watt, 365 nm |
References and Further Reading
- Neckers, D. C. Photochemical reactions of natural macromolecules. Photoreactions of proteins. J. Chem. Educ. 50, 164 (1973).
- Bintsis, T., Litopoulou-Tzanetaki, E. & Robinson, R. K. Existing and potential applications of ultraviolet light in the food industry – a critical review. J. Sci. Food Agric. 80, 637–645 (2000).
- Wolfe, R. L. Ultraviolet disinfection of potable water. Environ. Sci. Technol. 24, 768–773 (1990).
- Setlow, R. B., Carrier, W. L. & Bollum, F. J. Pyrimidine dimers in UV-irradiated poly dI:dC. Proc. Natl. Acad. Sci. 53, 1111–1118 (1965).
- Wacker, A., Dellweg, H. & Jacherts, D. Thymin-dimerisierung und Überlebensrate bei bakterien. J. Mol. Biol. 4, 410–412 (1962).
- Smith, K. C. Photochemical reactions of thymine, uracil, uridine, cytosine and bromouracil in frozen solution and in dried films. Photochem. Photobiol. 2, 503–517 (1963).
- Altenburg, E. The Limit of Radiation Frequency Effective in Producing Mutations. Am. Soc. Nat. 62, 540–545 (1928).
- Muller, H. J. The Production of Mutations by X-Rays. Proc. Natl. Acad. Sci. U. S. A. 14, 714–726 (1928).
- Muller, H. J. Induced mutations in Drosophila. in Cold Spring Harbor Symposia on Quantitative Biology vol. 9 151–167 (Cold Spring Harbor Laboratory Press, 1941).
- Muller, H. J. & Mackenzie, K. Discriminatory effect of ultra-violet rays on mutation in Drosophila. Nature 143, 83–84 (1939).
- Muller, H. J., Altenburg, L. S., Meyer, H. U., Edmondson, M. & Altenburg, E. The lack of proportionality between mutation rate and ultraviolet dose in Drosophila. Heredity 8, 153–185 (1954).
- Altenburg, L., ALTENBURG, E., Meyer, H. U. & Muller, H. J. The lack of proportionality of mutations recovered to dosage of ultra-violet administered to the polar cap of Drosophila. in Genetics vol. 35 95–95 (428 EAST PRESTON ST, BALTIMORE, MD 21202, 1950).
- Ellenhorn, J., PROKOFIEVA, A. & Muller, H. J. The optical dissociation of Drosophila chromomeres by means of ultraviolet light. CR Acad Sci USSR I 238, 241 (1935).
- Mackenzie, K. & Muller, H. J. Mutation effects of ultra-violet light in Drosophila. Proc. R. Soc. Lond. Ser. B-Biol. Sci. 129, 491–517 (1940).
- Meyer, H. U. & Muller, H. J. Influence of oxygen and temperature on the rate of autosomal recessive lethals induced by ultraviolet in teh polar cap of drosophila-melaongaster. in Genetics vol. 37 604–604 (428 EAST PRESTON ST, BALTIMORE, MD 21202, 1952).
- Meyer, H. U., Edmondson, M., Altenburg, L. & Muller, H. J. Studies on mutations induced by ultraviolet in the polar cap of Drosophila. in Genetics vol. 35 123–124 (428 EAST PRESTON ST, BALTIMORE, MD 21202, 1950).
- Bovie, W. T. A Preliminary Note on the Coagulation of Proteins by Ultraviolet Light. Science 37, 24–25 (1913).
- Bovie, W. T. The Temperature Coefficient of the Coagulation Caused by Ultraviolet Light. Science 37, 373–375 (1913).
- McLaren, A. & Luse, R. Mechanism of Inactivation of Enzyme Proteins by Ultraviolet Light. Science 134, 836–837 (1961).
- Müller, H.-M., Van Auken, K. M., Li, Y. & Sternberg, P. W. Textpresso Central: a customizable platform for searching, text mining, viewing, and curating biomedical literature. BMC Bioinformatics 19, (2018).
About Analytik Jena US
Analytik Jena is a provider of instruments and products in the areas of analytical measuring technology and life science. Its portfolio includes the most modern analytical technology and complete systems for bioanalytical applications in the life science area.
Comprehensive laboratory software management and information systems (LIMS), service offerings, as well as device-specific consumables and disposables, such as reagents or plastic articles, complete the Group’s extensive range of products.
About life science
The Life Science product area demonstrates the biotechnological competence of Analytik Jena AG. We provide a wide product spectrum for automated total, as well as individual solutions for molecular diagnostics. Our products are focused to offer you a quality and the reproducibility of your laboratory results.
This will surely ease your daily work and speed up your work processes in a certain way. All together we support you through the complete process of the lab work. Besides we offer customized solutions and are able to adapt our products to your needs. Automated high-throughput screening systems for the pharmaceutical sector are also part of this segment’s extensive portfolio.
About analytical instrumentation
Analytik Jena has a long tradition in developing high-performance precision analytical systems which dates back to the inventions made by Ernst Abbe and Carl Zeiss. We have grown to become one of the most innovative manufacturers of analytical measuring technology worldwide.
Our business unit Analytical Instrumentation offers excellent competencies in the fields of optical spectroscopy, sum parameters and elemental analysis. Being proud of our core competency we grant all our customers a long-term warranty of 10 years for our high-performance optics.
About lab automation
With more than 25 years of market experience, Analytik Jena with its CyBio® Product Line is a leading provider for high quality liquid handling and automation technologies. In the pharmaceutical and life science industries, our products enjoy the highest reputation for precision, reliability, robustness and simplicity.
Moreover, the Automation Team designs, produces and installs fully automated systems tailored to our clients' application, throughput and capacity requirements. From stand-alone CyBio® Well up to fully customized robotic systems we handle your compounds, biomolecules and cells with great care.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.