The immune response is a coordinated process involving diverse chemicals and signaling pathways. When these pathways are activated, sick cells and pathogens are successfully eradicated in individuals with healthy immune systems. In some cases, the immune system can become overactive, leading to autoimmune disease.
Image Credit: peterschreiber.media/Shutterstock.com
Three critical molecules have been shown to play an important role in immune response control and modulation: High Mobility Group Box 1 (HMGB-1), neopterin (Np), and the soluble Interleukin-2 receptor (sIL-2R).1-15
There are similarities and distinctions among the immune system's three musketeers. They share functions across a wide range of immune-related disorders, and researchers must consider how they should be measured and when.
All three are valuable indicators for immunological activation, and their roles are outlined in this article (Figure 1).
Inflammatory mediators
HMGB-1 is a potent inflammatory mediator produced by activated immune cells and functions as a late mediator of inflammation.1 Neopterin, a consequence of the GTP-metabolic pathway, is produced by activated macrophages/monocytes and serves as a marker of cellular immunological activation. sIL-2R, likewise released during immune activation, is a marker of T-cell activation and inflammation.2,3
Biomarkers of disease activity
Elevated levels of HMGB-1, sIL-2R, and neopterin are found in a variety of inflammatory and autoimmune illnesses, making them possible biomarkers for disease activity and severity.4-6
Prognostic indicators
High quantities of all three molecules have been linked to poor outcomes and disease progression in conditions like cancer, sepsis, and autoimmune diseases.7-9
Immune regulation
HMGB-1 can act as a chemoattractant, activating and mobilizing immune cells.10 Neopterin has been demonstrated to modulate oxidative stress and apoptosis as part of its immune regulatory role.11 Finally, sIL-2R regulates T-cell proliferation and activation, which helps maintain immunological homeostasis.12
Therapeutic targets
Strategies targeting HMGB-1, neopterin, and sIL-2R are being investigated as possible treatments for a variety of inflammatory and autoimmune disorders, including cancer, rheumatoid arthritis, and multiple sclerosis.13-15
Researchers must determine whether they need to continuously measure all three markers or whether one would suffice. This depends on the specific disease state being researched. As a rule of thumb, the three molecules are all confirmed markers of immunological activation, so the researcher or clinician could begin by comparing the disease being studied, diagnosed, or treated with previous research in the field.
The second aspect to consider is that, if tests are performed in the clinic rather than in the research environment, the 'tiebreaker' is that the clinician must use an IVD-compliant test rather than a RUO (research use only) test.
To aid the selection of biomarker(s) to monitor illness status, this article will briefly outline the research on each type before consolidating how they may be quantified.
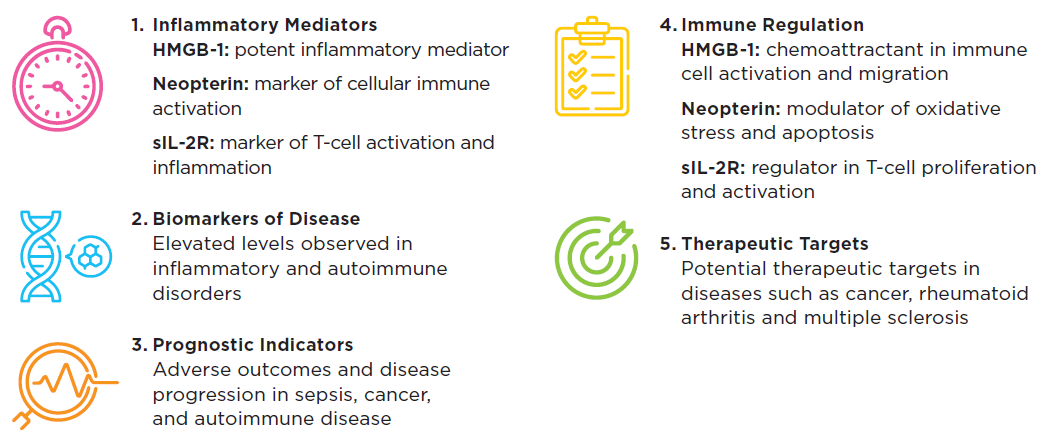
Figure 1. High Mobility Group Box 1 (HMGB-1), neopterin (Np), and soluble Interleukin-2 receptor (sIL-2R): three key molecules that play an essential role in the regulation and modulation of the immune response. Image Credit: IBL International GmbH, Part of Tecan Group
HMGB-1
Human HMGB-1 is required for cell stress response and plays a significant role in many infectious diseases, as well as ischemia, immunological disorders, neurological diseases, and cancers.16 It has received much attention in recent years for its potential as a biomarker for assessing inflammation and immune system dysfunction. The Tecan Blog series provides a more detailed explanation of HMGB-1’s mechanism of action in relation to COVID-19 infection.
HMGB-1 is a non-histone nuclear protein that can be released when cells are stressed, injured, or inflamed.17 Once released into the extracellular environment, it functions as a ‘danger-associated molecular pattern’ (DAMP) and can elicit inflammatory responses by attaching to pattern recognition receptors (PRRs) on immune cells.18
Elevated HMGB-1 levels have been found in individuals with rheumatoid arthritis, systemic lupus erythematosus (SLE), multiple sclerosis, and inflammatory bowel disease (IBD), all of which are autoimmune diseases.19
In rheumatoid arthritis cases, HMGB-1 levels in synovial fluid and serum are associated with disease activity and severity, making it a possible biomarker for disease progression and therapy response.20
HMGB-1 levels are elevated in both blood and urine in cases of SLE. These levels are associated with disease activity, organ damage, and the presence of specific autoantibodies.21 HMGB-1 has also been investigated as a biomarker for sepsis, a potentially fatal illness characterized by a massive immunological response to infection.22
Elevated HMGB-1 levels have also been linked to the development and progression of several malignancies, including breast, lung, and colorectal cancer, possibly due to its involvement in promoting inflammation and tumor growth.23
There are some significant benefits of using HMGB-1 as a biomarker, including its stability in bodily fluids and its direct involvement in the inflammatory and immunological responses that underpin multiple diseases.
It has been difficult to develop standardized methods for assessing HMGB-1 levels and to demonstrate its clinical value as a diagnostic and prognostic biomarker across a variety of disorders. However, ELISA tests are now available that allow us to standardize HMGB-1 assays and, hence, compare data across labs worldwide.
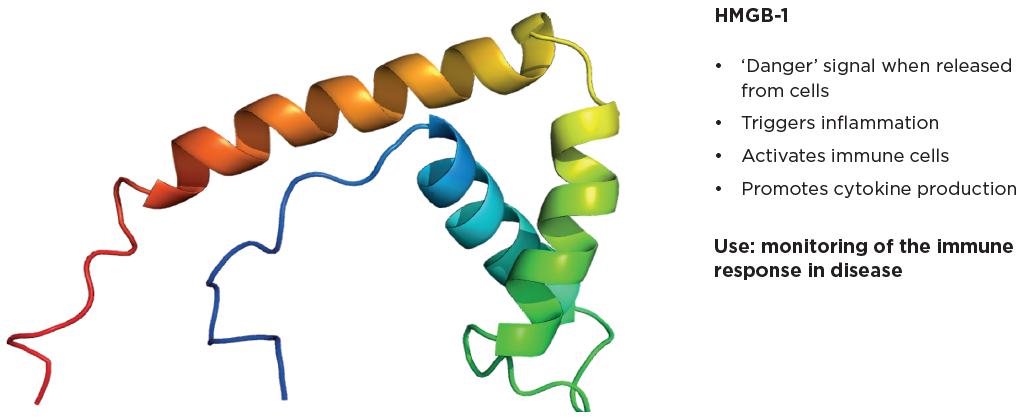
Figure 2. HMGB-1 plays a key role in the body's response to injury and infection. Image Credit: IBL International GmbH, Part of Tecan Group
Neopterin
Neopterin (Np) is produced by macrophages and dendritic cells in response to IFN-γ, a cytokine released by activated T-cells and natural killer cells during immunological reactions.24-25
Np, also known as 2-amino-4-hydroxy-6-(D-erythro-1',2',3'-trihydroxypropyl)-pteridine, is classified as a pteridine.26 This Np was first identified in 1963 from bee larvae, worker bees, and royal jelly.27 - 29 Researchers Sakurai and Goto first isolated Np in human urine in 1967.30
It has since been intensively studied across a wide range of inflammatory disorders, including viral, bacterial, and parasitic infections, cardiovascular diseases, autoimmune diseases, and malignant tumors.31-37
Neopterin levels in bodily fluids, such as serum, urine, and cerebrospinal fluid (CSF), rise in response to various inflammatory and immune-mediated diseases.38 Viral diseases such as HIV, hepatitis B and C, and CMV exhibit high Np levels, indicating that the cellular immune response to these viruses has been activated.26
Neopterin levels correlate with disease activity and severity in autoimmune illnesses such as rheumatoid arthritis, systemic lupus erythematosus (SLE), and multiple sclerosis, suggesting it could serve as a biomarker to track disease progression and therapeutic responses.39
Neopterin levels are also elevated when some cancers develop, including leukemia, lymphoma, and solid tumors, probably due to immune cell activation by tumor antigens.40
Elevated neopterin levels in allograft recipients can suggest acute rejection episodes or post-transplant infections, which help to monitor transplant outcomes.41
Neopterin has also been investigated as a biomarker for cardiovascular diseases, as it is linked to an increased risk of adverse outcomes and may reflect the underlying inflammatory processes that contribute to atherosclerosis.42
The advantages of employing neopterin as a biomarker, like those of HMGB-1, include its stability in biological fluids, its direct relationship to cellular immunological activity, and the availability of well-established analytical methods for measuring it.
Again, ELISA is a simple and reliable method for assessing neopterin because it is sensitive, robust, and accurate, even in complex matrices such as serum, plasma, and urine.
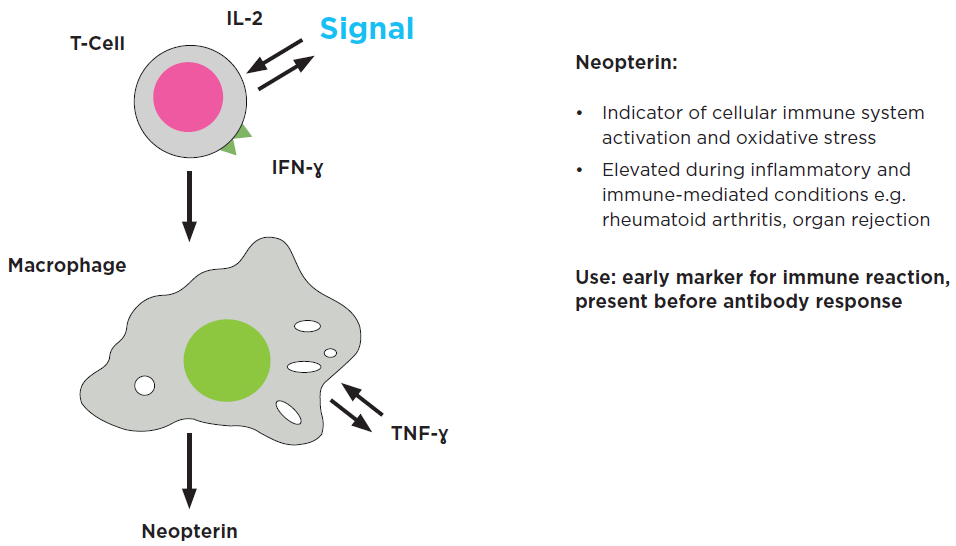
Figure 3. Neopterin is an early marker for immune activation. Image Credit: IBL International GmbH, Part of Tecan Group
sIL-2 receptor
Similar to HMGB-1 and neopterin, Interleukin-2 (IL-2) is a major signaling molecule in the human immune system. IL-2 is a cytokine, a tiny, secreted protein produced by cells that has a unique effect on cell connections and communication. This assists in regulating the body's natural response to infection, allowing it to distinguish between foreign (‘non-self’) and ‘self’.43
IL-2 binds to lymphocyte-expressed IL-2 receptors to exert its effects. The primary sources of IL-2 are activated CD4+ and CD8+ T cells.43
When T-cells are activated, the soluble form of the interleukin-2 receptor (sIL-2R) is secreted, or ‘shed’, resulting in a greater-than-background concentration of sIL-2R in patients suffering from numerous illnesses associated with an ongoing immunological response (Figure 4).43
Clinicians use quantification of soluble interleukin-2 receptor (sIL-2R) in serum or plasma in adults to assess immunological function in vivo and to explore and treat a variety of disorders.
Elevated sIL-2R levels are found in autoimmune disorders, including rheumatoid arthritis, systemic lupus erythematosus (SLE), and multiple sclerosis, and they correlate with disease activity and severity.44-45
In SLE, greater sIL-2R levels are associated with organ involvement and the presence of particular autoantibodies.46 In rheumatoid arthritis, sIL-2R levels in serum and synovial fluid are linked to joint inflammation and erosion, making it a valuable biomarker for tracking disease progression and therapy response.47
sIL-2R has also been identified as a biomarker for transplant rejection, as its levels increase during acute rejection episodes in organ transplants such as the kidney, liver, and heart.48 sIL-2R levels are directly correlated to some hematological malignancies, such as hairy cell leukemia and adult T-cell leukemia/lymphoma, indicating malignant T-cell proliferation.49
Sarcoidosis, biliary cirrhosis, chronic immune activation in common variable immunodeficiency (CVID), and hemophagocytic lymphohistiocytosis (HLH) are among the other immune-mediated disorders linked to elevated sIL-2R.50 - 54
In infectious disorders, sIL-2R levels may rise due to T-cell activation in response to infections including HIV, CMV, and tuberculosis.55
The stability of sIL-2R in bodily fluids, as well as its direct relationship with T-cell activation, a key mechanism in many immune-mediated diseases, makes it an attractive biomarker candidate.
The relative stability of sIL-2R levels throughout adulthood and its showing only small differences by gender contribute to sIL-2R's appeal as a biomarker.56 -57 However, sIL-2R levels can be altered by a number of factors, including age, renal function, and concurrent drugs, which should be taken into account during interpretation.58
Soluble IL-2 receptor levels are often measured by enzyme-linked immunosorbent assay (ELISA) or chemiluminescent immunoassay (CLIA). Commercially accessible assays should be calibrated to the international reference standard, NIBSC 97/600.
Table 1. Soluble IL-2R is a reliable marker of an ongoing immune response. Credit: IBL International GmbH, Part of Tecan Group
| Population |
Concentration |
Level |
Reference |
| Non-diseased individuals |
| Adults > 18 yrs |
Median: 339 [282.5–440.7] U/mL |
Low |
Halacli, B., et al. (2016). Journal of critical care, 35, 185–190. |
| Adults (21-55 yrs) |
Mean: 410 (SD: 186) U/mL |
Low |
Mariotti, S., et al. (1992) Clinical endocrinology, 37(5), 415–422 |
Adults, n=20
(age 29.5 ± 1.8 yrs),
men and women |
Mean: 209 (SD: 25) U/mL |
Low |
Manoussakis, M. N., et al. (1992) Lupus, 1(2), 105–109. |
Adults, n=70
(age 37–63 yrs),
men and women |
Median: 1,028 pg/mL
(Range: 263-2210) |
Low |
Schimmelpennink, M. C., et al. (2020). Expert review of respiratory medicine, 14(7), 749–756. |
| Diseased individuals |
| Sepsis: HLH, n=10, adults |
Median: 2,259.9
[971.8-8, 180.8] U/mL |
High |
Halacli, B., et al. (2016). Journal of critical care, 35, 185–190. |
Graves’ Disease, n=61
men and women |
Mean: 1,610 (SD: 962) U/mL |
High |
Mariotti, S., et al. (1992) Clinical endocrinology, 37(5), 415–422 |
Systemic Lupus
Erythematosus,n=25,
men and women |
Mean: 682 (SD:115) U/mL |
High |
Manoussakis, M. N., et al. (1992) Lupus, 1(2), 105–109. |
Rheumatoid Arthritis,
n=41, men and women |
Mean: 734 (SD: 101) U/mL |
High |
Manoussakis, M. N., et al. (1992) Lupus, 1(2), 105–109. |
Sarcoidosis; n=104,
men and women |
Median: 5,534 pg/mL
(Range: 1,351-55,000 pg/mL) |
High |
Schimmelpennink, M. C., et al. (2020). Expert review
of respiratory medicine, 14(7), 749–756. |
Löfgren Syndrome (acute form
of sarcoidosis), n=17,
men and women |
Median: 5,682 pg/mL
(Range: 560-36,000 pg/mL) |
High |
Schimmelpennink, M. C., et al. (2020). Expert review
of respiratory medicine, 14(7), 749–756. |
sIL-2R:
- Associated with infection, autoimmune diseases, and inflammatory conditions
- Helps distinguish self from non-self
- Marker of T-cell activation
Use: measurable surrogate for T-cell activation in vivo as part of disease prognosis and management.
Measuring the ‘three musketeers’ of the immune activation pathway
All three immunological markers, HMGB-1, neopterin, and sIL-2R, have been established as indicators of ongoing immune response and, theoretically, as able to monitor a wide range of immune-mediated diseases; in essence, they can all be considered universal or generic markers of immune activation.
The researcher or clinician must next make an informed decision on which marker(s) would be most appropriate for the disease being investigated, diagnosed, or treated, based on the empirical evidence available in the literature and commercially accessible tests.
All three markers can be accurately assessed using immunoassays, most notably ELISA. The Tecan Blog articles describe the intimate relationship between ELISA tests and other accessible techniques, such as HPLC, RIA, and mass spectrometry.
ELISA is by far the simplest technique to apply. The following will outline an examination of current tests and determine how these ELISA tests may be scaled and automated to provide the most reproducible findings, therefore facilitating worldwide standardization.
Choosing and implementing ELISAs for immunology biomarkers
ELISA assays are widely available for the three outlined immune activation biomarkers (HMGB-1, neopterin, and sIL-2R).
Most of them are for research use only (RUO). This means that - even if their efficacy as a marker for a specific disease has been demonstrated through study and basic empirical evidence - their long-term usage in the clinical field cannot be guaranteed, necessitating the adoption of IVD-compliant tests.
Furthermore, most tests are intended for manual performance, but they should be easily automated and scalable to achieve optimal repeatability and inter-lab comparisons. These are only a few of the main criteria for using ELISA to assess immunological activation: a more detailed checklist may include:
- Ready-to-use assay reagents: dilution is not necessary
- Ready-to-use standard-curve reagents: no stock dilution is required
- Kit ready-calibrated against standard preparations and other methods
- Internal kit controls provided
- Easily automatable
Tecan ELISA kits can be run manually for initial testing and validation when used for IVD-compliant clinical use, or automated based on your lab’s specific standardization and throughput requirements.
Automation scripts are widely available and are ‘open system’, meaning they can be easily converted to your automation platform, and Tecan can also work directly with you to integrate them.
Many white papers and articles are offered for further study, depending on whether you want to learn more about the science behind these tests or use ELISA in your lab, with or without automation. Visit your country's website to learn more about buying ELISA testing and ongoing discounts.
Tecan's open approach to IVDR and automation is leading the way toward international standardization and the automation of ELISA, the main method for measuring immunology markers in clinical environments, opening new avenues for future research as the company continues to capitalize on the method’s enormous potential in diagnosing, treating, and monitoring disease.
References
- Andersson, U., Yang, H. and Harris, H. (2018). High-mobility group box 1 protein (HMGB1) operates as an alarmin outside as well as inside cells. Seminars in Immunology, 38, pp.40–48. DOI: 10.1016/j.smim.2018.02.011. https://linkinghub.elsevier.com/retrieve/pii/S1044532317300763.
- Murr, C., et al. (1999). Increased neopterin concentrations in patients with cancer: indicator of oxidative stress? Anticancer research, (online) 19(3A), pp.1721–8. Available at: https://pubmed.ncbi.nlm.nih.gov/10470106/.
- Rubin, L.A. (1990). The Soluble Interleukin-2 Receptor: Biology, Function, and Clinical Application. Annals of Internal Medicine, 113(8), p.619. DOI: 10.7326/0003-4819-113-8-619. https://www.acpjournals.org/doi/10.7326/0003-4819-113-8-619.
- Abdulahad, D.A., et al. (2011). High mobility group box 1 (HMGB1) and anti-HMGB1 antibodies and their relation to disease characteristics in systemic lupus erythematosus. Arthritis Research & Therapy, 13(3). DOI: 10.1186/ar3332. https://link.springer.com/article/10.1186/ar3332.
- Sucher, R., et al. (2010). Neopterin, a prognostic marker in human malignancies. Cancer Letters, 287(1), pp.13–22. DOI: 10.1016/j.canlet.2009.05.008. https://linkinghub.elsevier.com/retrieve/pii/S0304383509003462.
- Witkowska, A.M. (2005). On the Role of sIL-2R Measurements in Rheumatoid Arthritis and Cancers. Mediators of Inflammation, 2005(3), pp.121–130. DOI: 10.1155/mi.2005.121. https://onlinelibrary.wiley.com/doi/10.1155/MI.2005.121.
- Sunden-Cullberg, J., Norrby-Teglund, A. and Treutiger, C.J. (2006). The role of high mobility group box-1 protein in severe sepsis. Current Opinion in Infectious Diseases, 19(3), pp.231–236. DOI: 10.1097/01.qco.0000224816.96986.67. https://journals.lww.com/co-infectiousdiseases/abstract/2006/06000/the_role_of_high_mobility_group_box_1_protein_in.3.aspx.
- Murr, C., et al. (2002). Neopterin as a Marker for Immune System Activation. Current Drug Metabolism, 3(2), pp.175–187. DOI: 10.2174/1389200024605082. https://www.eurekaselect.com/article/9114.
- Witkowska, A. M., & Sadiq, S. (2019). Soluble IL-2 receptor as a biomarker for immune monitoring in multiple sclerosis. Journal of Neuroimmunology, 329, 93-98.
- Bianchi, M.E. (2006). DAMPs, PAMPs and alarmins: all we need to know about danger. Journal of Leukocyte Biology, 81(1), pp.1–5. DOI: 10.1189/jlb.0306164. https://academic.oup.com/jleukbio/article-abstract/81/1/1/6975763?redirectedFrom=fulltext.
- Hoffmann, G., Wirleitner, B. and Fuchs, D. (2003). Potential role of immune system activation-associated production of neopterin derivatives in humans. Inflammation Research, 52(8), pp.313–321. DOI: 10.1007/s00011-003-1181-9. https://link.springer.com/article/10.1007/s00011-003-1181-9.
- Rubin, L.A. (1990). The Soluble Interleukin-2 Receptor: Biology, Function, and Clinical Application. Annals of Internal Medicine, 113(8), p.619. DOI: 10.7326/0003-4819-113-8-619. https://www.acpjournals.org/doi/10.7326/0003-4819-113-8-619.
- Mollica, L., Bours, V., & Feron, O. (2015). Anticancer activity of HMGB proteins and their derivatives. Current Medicinal Chemistry, 22(18), 2285-2293.
- Reibnegger, G., et al. (1986). Urinary neopterin reflects clinical activity in patients with rheumatoid arthritis. Arthritis & Rheumatism, 29(9), pp.1063–1070. DOI: 10.1002/art.1780290902. https://onlinelibrary.wiley.com/doi/abs/10.1002/art.1780290902.
- Bielekova, B., et al. (2006). Regulatory CD56bright natural killer cells mediate immunomodulatory effects of IL-2R -targeted therapy (daclizumab) in multiple sclerosis. Proceedings of the National Academy of Sciences, 103(15), pp.5941–5946. DOI: 10.1073/pnas.0601335103. https://www.pnas.org/doi/full/10.1073/pnas.0601335103.
- Kang, R., et al. (2014). HMGB1 in health and disease. Molecular Aspects of Medicine, (online) 40, pp.1–116. DOI: 10.1016/j.mam.2014.05.001. https://www.sciencedirect.com/science/article/abs/pii/S0098299714000375?via%3Dihub.
- Chen, R., Kang, R. and Tang, D. (2022). The mechanism of HMGB1 secretion and release. Experimental & Molecular Medicine, (online) 54(2), pp.91–102. DOI: 10.1038/s12276-022-00736-w. https://www.nature.com/articles/s12276-022-00736-w.
- Das, N., et al. (2016). HMGB1 Activates Proinflammatory Signaling via TLR5 Leading to Allodynia. Cell Reports, (online) 17. DOI: 10.1016/j.celrep.2016.09.076. https://www.cell.com/cell-reports/fulltext/S2211-1247(16)31332-8.
- Harris, H.E., Andersson, U. and Pisetsky, D.S. (2012). HMGB1: A multifunctional alarmin driving autoimmune and inflammatory disease. Nature Reviews Rheumatology, 8(4), pp.195–202. DOI: 10.1038/nrrheum.2011.222. https://www.nature.com/articles/nrrheum.2011.222.
- Goldstein, R. S., et al. (2007). Cholinergic anti-inflammatory pathway activity and High Mobility Group Box-1 (HMGB1) serum levels in patients with rheumatoid arthritis. Molecular medicine (Cambridge, Mass.), 13(3-4), 210–215. DOI: 10.2119/2006–00108.Goldstein. https://link.springer.com/article/10.2119/2006-00108.Goldstein.
- Abdulahad, W.H., Lamprecht, P. and Kallenberg, C.G. (2011). T-helper cells as new players in ANCA-associated vasculitides. Arthritis Research & Therapy, (online) 13(4), p.236. DOI: 10.1186/ar3362. https://link.springer.com/article/10.1186/ar3362.
- Angus, D.C. and van der Poll, T. (2013). Severe Sepsis and Septic Shock. New England Journal of Medicine, (online) 369(9), pp.840–851. DOI: 10.1056/nejmra1208623. https://www.nejm.org/doi/10.1056/NEJMra1208623.
- Ellerman, J.E., et al. (2007). Masquerader: high mobility group box-1 and cancer. Clinical Cancer Research: An Official Journal of the American Association for Cancer Research, (online) 13(10), pp.2836–2848. DOI: 10.1158/1078-0432.CCR-06-1953. https://aacrjournals.org/clincancerres/article/13/10/2836/194155/Masquerader-High-Mobility-Group-Box-1-and-Cancer.
- Müller, M.M., et al. (1991). Neopterin in clinical practice. Clinica Chimica Acta, 201(1-2), pp.1–16. DOI: 10.1016/0009-8981(91)90019-9. https://www.sciencedirect.com/science/article/abs/pii/0009898191900199?via%3Dihub.
- Fuchs, D., et al. (1988). Neopterin as a marker for activated cell-mediated immunity: Application in HIV infection. Immunology Today, (online) 9(5), pp.150–155. DOI: 10.1016/0167-5699(88)91203-0. https://www.sciencedirect.com/science/article/abs/pii/0167569988912030?via%3Dihub.
- Murr, C., et al. (2002). Neopterin as a Marker for Immune System Activation. Current Drug Metabolism, 3(2), pp.175–187. DOI: 10.2174/1389200024605082. https://www.eurekaselect.com/article/9114.
- Rembold, H. and Buschmann, L. (1963). Struktur und Synthese des Neopterins. Chemische Berichte, 96(5), pp.1406–1410. DOI: 10.1002/cber.19630960532. https://chemistry-europe.onlinelibrary.wiley.com/doi/abs/10.1002/cber.19630960532.
- Hamerlinck, F. (1999). Neopterin: a review. Experimental Dermatology, 8(3), pp.167–176. DOI: 10.1111/j.1600-0625.1999.tb00367.x. https://onlinelibrary.wiley.com/doi/10.1111/j.1600-0625.1999.tb00367.x.
- Rembold, H. and Buschmann, L. (1963). Untersuchungen Über die Pteridine der Bienenpuppe (Apis Mellifica). Justus Liebigs Annalen der Chemie, 662(1), pp.72–82. DOI: 10.1002/jlac.19636620108. https://chemistry-europe.onlinelibrary.wiley.com/doi/10.1002/jlac.19636620108.
- SAKURAI, A. and GOTO, M. (1967). Neopterin: Isolation from Human Urine. The Journal of Biochemistry, 61(1), pp.142–145. DOI: 10.1093/oxfordjournals.jbchem.a128513. https://academic.oup.com/jb/article-abstract/61/1/142/824071?redirectedFrom=fulltext.
- Schneider-Crease, I.A., et al. (2022). Urinary neopterin reflects immunological variation associated with age, helminth parasitism, and the microbiome in a wild primate. Scientific Reports, (online) 12(1), p.21307. DOI: 10.1038/s41598-022-25298-9. https://www.nature.com/articles/s41598-022-25298-9.
- Rasmi, Y., et al. (2022). The importance of neopterin in COVID-19: The prognostic value and relation with the disease severity. Clinical Biochemistry, 104, pp.1–12. DOI: 10.1016/j.clinbiochem.2022.03.002. https://www.sciencedirect.com/science/article/pii/S0009912022000765?via%3Dihub.
- Ondrej Heneberk., et al (2022). Neopterin Levels in Periodontitis and after Nonsurgical Periodontal Therapy: Evaluation of Gingival Crevicular Fluid, Oral Fluid, Serum and Urinary Samples - A Case-Control Study. Biomedicines, 10(12), pp.3200–3200. DOI: 10.3390/biomedicines10123200. https://www.mdpi.com/2227-9059/10/12/3200.
- Pink, R., et al. (2016). Salivary neopterin concentrations in patients with cancer of the oral cavity. Pteridines, 27(3-4), pp.53–58. DOI: 10.1515/pterid-2015-0017. https://www.degruyterbrill.com/document/doi/10.1515/pterid-2015-0017/html?srsltid=AfmBOoqnNyY9jAICnlnCHV0vltnPvkrAi83PVRKV30c9obmq-XwJXQCy.
- Dogheim, G.M., Amralla, M.T. and Werida, R.H. (2022). Role of neopterin as an inflammatory biomarker in congestive heart failure with insights on effect of drug therapies on its level. Inflammopharmacology, 30(5), pp.1617–1622. DOI: 10.1007/s10787-022-01028-5. https://link.springer.com/article/10.1007/s10787-022-01028-5.
- Labouret, M., et al. (2024). Juvenile neuropsychiatric systemic lupus erythematosus: A specific clinical phenotype and proposal of a probability score. Lupus, 33(4), pp.328–339. DOI: 10.1177/09612033241229022. https://journals.sagepub.com/doi/10.1177/09612033241229022.
- Huber, C.G., et al. (1984). Immune response-associated production of neopterin. Release from macrophages primarily under control of interferon-gamma. 160(1), pp.310–316. DOI: 10.1084/jem.160.1.310. https://rupress.org/jem/article-abstract/160/1/310/23335/Immune-response-associated-production-of-neopterin?redirectedFrom=fulltext.
- Hailemichael, W., et al. (2021). Neopterin: A Promising Candidate Biomarker for Severe COVID-19. Journal of Inflammation Research, Volume 14, pp.245–251. DOI: 10.2147/jir.s290264. https://www.dovepress.com/neopterin-a-promising-candidate-biomarker-for-severe-covid-19-peer-reviewed-fulltext-article-JIR.
- Mangoni, A.A. and Zinellu, A. (2023). A systematic review and meta-analysis of neopterin in rheumatic diseases. Frontiers in Immunology, 14, pp.1271383–1271383. DOI: 10.3389/fimmu.2023.1271383. https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2023.1271383/full.
- Sucher, R., et al. (2010). Neopterin, a prognostic marker in human malignancies. Cancer Letters, 287(1), pp.13–22. DOI: 10.1016/j.canlet.2009.05.008. https://www.sciencedirect.com/science/article/abs/pii/S0304383509003462?via%3Dihub.
- Reibnegger, G., et al. (1991). Posttransplant neopterin excretion in renal allograft recipients--a reliable diagnostic aid for acute rejection and a predictive marker of long-term graft survival. Transplantation, 52(1), pp.58–63. DOI: 10.1097/00007890-199107000-00012. https://journals.lww.com/transplantjournal/abstract/1991/07000/posttransplant_neopterin_excretion_in_renal.12.aspx.
- Sugioka, K., et al. (2010). Neopterin and Atherosclerotic Plaque Instability in Coronary and Carotid Arteries. Journal of Atherosclerosis and Thrombosis, 17(11), pp.1115–1121. DOI: 10.5551/jat.4606. https://www.jstage.jst.go.jp/article/jat/17/11/17_4606/_article.
- Liao, W., Lin, J.-X. and Leonard, W.J. (2011). IL-2 family cytokines: new insights into the complex roles of IL-2 as a broad regulator of T helper cell differentiation. Current Opinion in Immunology, 23(5), pp.598–604. DOI: 10.1016/j.coi.2011.08.003. https://www.sciencedirect.com/science/article/abs/pii/S0952791511001063?via%3Dihub.
- Rafaqat, S. and Rafaqat, S. (2023). Role of IL-2/IL-2 receptor in pathogenesis of autoimmune disorders: Genetic and therapeutic aspects. World Journal of Medical Genetics, (online) 11(3), pp.28–38. DOI: 10.5496/wjmg.v11.i3.28. https://www.wjgnet.com/2220-3184/full/v11/i3/28.htm.
- Neish, C., et al. (1993). Elevated serum soluble interleukin-2 receptor levels in subacute cutaneous lupus erythematosus. Journal of Dermatological Science, 5(3), pp.143–149. DOI: 10.1016/0923-1811(93)90761-d. https://www.jdsjournal.com/article/0923-1811(93)90761-D/abstract.
- Illei, G. G., et al. (2004). Biomarkers in systemic lupus erythematosus: II. Markers of disease activity. Arthritis & Rheumatism, 50(7), 2048–2065. DOI: 10.1002/art.20345. https://onlinelibrary.wiley.com/doi/10.1002/art.20345.
- Symons, J.A., et al. (1988). Soluble IL-2 receptor in rheumatoid arthritis. Correlation with disease activity, IL-1 and IL-2 inhibition. Journal of immunology (Baltimore, Md. : 1950), (online) 141(8), pp.2612–8. Available at: https://pubmed.ncbi.nlm.nih.gov/3262665/.
- Zucchelli, G.C., et al. (1990). Increased circulating concentrations of interleukin 2 receptor during rejection episodes in heart- or kidney-transplant recipients. Clinical chemistry, (online) 36(12), pp.2106–9. Available at: https://pubmed.ncbi.nlm.nih.gov/2253354/.
- Murakami, J., et al. (2019). Serum soluble interleukin‑2 receptor levels for screening for malignant lymphomas and differential diagnosis from other conditions. Molecular and Clinical Oncology. (online) DOI: 10.3892/mco.2019.1922. https://www.spandidos-publications.com/10.3892/mco.2019.1922.
- Hong, T., et al. (2017). Serum soluble interleukin-2 receptor level is more sensitive than angiotensin-converting enzyme or lysozyme for diagnosis of sarcoidosis and may be a marker of multiple organ involvement. Journal of Dermatology, 44(7), pp.789–797. DOI: 10.1111/1346-8138.13792. https://onlinelibrary.wiley.com/doi/10.1111/1346-8138.13792.
- Peerlings, D., Mimpen, M. and Damoiseaux, J. (2021). The IL-2 – IL-2 receptor pathway: Key to understanding multiple sclerosis. Journal of Translational Autoimmunity, 4, p.100123. DOI: 10.1016/j.jtauto.2021.100123. https://www.sciencedirect.com/science/article/pii/S2589909021000435?via%3Dihub.
- Barak, V., et al. (2009). Serum inflammatory cytokines, complement components, and soluble interleukin 2 receptor in primary biliary cirrhosis. Journal of Autoimmunity, 33(3-4), pp.178–182. DOI: 10.1016/j.jaut.2009.09.010. https://www.sciencedirect.com/science/article/abs/pii/S0896841109001243?via%3Dihub.
- Litzman, J., et al. (2012). Chronic immune activation in common variable immunodeficiency (CVID) is associated with elevated serum levels of soluble CD14 and CD25 but not endotoxaemia. Clinical & Experimental Immunology, 170(3), pp.321–332. DOI: 10.1111/j.1365-2249.2012.04655.x. https://academic.oup.com/cei/article-abstract/170/3/321/6429053?redirectedFrom=fulltext.
- Lin, M., et al. (2017). Clinical utility of soluble interleukin-2 receptor in hemophagocytic syndromes: a systematic scoping review. 96(8), pp.1241–1251. DOI: 10.1007/s00277-017-2993-y. https://link.springer.com/article/10.1007/s00277-017-2993-y.
- Carcelain, G., et al. (2001). Clinical Immunology, 99(3), 286-294.
- Rubin, L.A. (1990). The Soluble Interleukin-2 Receptor: Biology, Function, and Clinical Application. Annals of Internal Medicine, 113(8), p.619. DOI: 10.7326/0003-4819-113-8-619. https://www.acpjournals.org/doi/10.7326/0003-4819-113-8-619.
- Taniguchi, T. (1993). The IL-2/IL-2 receptor system: A current overview. Cell, 73(1), pp.5–8. DOI: 10.1016/0092-8674(93)90152-g. https://www.cell.com/cell/abstract/0092-8674(93)90152-G?_returnURL=https%3A%2F%2Flinkinghub.elsevier.com%2Fretrieve%2Fpii%2F009286749390152G%3Fshowall%3Dtrue.
- Alende-Castro, V., et al. (2023). Factors influencing serum concentrations of soluble interleukin-2 receptor: a general adult population study. All Life, 16(1). DOI: 10.1080/26895293.2023.2169958. https://www.tandfonline.com/doi/full/10.1080/26895293.2023.2169958.
About IBL International GmbH, Part of Tecan Group
Specialty diagnostics to improve people’s lives and health
With decades of experience, Tecan has built a strong legacy of innovation in in vitro diagnostic testing for endocrinology, immunology and autoimmunity at IBL International, specializing in the development, manufacture and supply of immunoassays as well as LC-MS solutions. These products are designed and produced to the highest standards, providing diagnostics labs with reliable data and improved workflow efficiency to assess various health conditions from blood, urine, saliva and cerebrospinal fluid samples.
Tecan’s reagents portfolio includes a number of specialty diagnostic assays for endocrinology, immunology, neurotransmitters and autoimmunity in clinical diagnostics, along with key assays for the research segment, including BD-Tau LUM and NF Light®* ELISA.
By combining Tecan’s proven automation capabilities and leadership in instrumentation with IBL International’s specialized immunoassay and LC-MS portfolio, Tecan offers complete solutions tailored to the needs of specialty diagnostics and research laboratories. These offerings streamline lab workflows by boosting productivity, increasing efficiency and meeting high regulatory standards.
This focus on compliance helped Tecan respond quickly to the EU’s In Vitro Diagnostic Regulation (IVDR), becoming one of the first companies to achieve product certification under the new rules.
Tecan continues to invest in innovation, advancing its portfolio to address emerging diagnostic needs in both clinical and research settings. Recent developments include specialized assays such as BD-Tau, sIL-2R, SCCA2 and Periostin ELISAs, supporting neurology and immunology applications, as well as LC-MS based assays for vitamins B1 and B6, A and E testing in endocrinology.
Disclaimer:
Products manufactured and distributed by IBL International. Availability and regulatory status may vary across regions depending on local country specific registration. The combined use of the reagents, process script and instrument has to be validated individually on site by each laboratory.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.