Sponsored Content by InProcess-LSPReviewed by Olivia FrostApr 30 2026
Authors: Nick Koumakis and Rut Besseling.
Protein aggregation in downstream processing (DSP) is a product-quality concern and major process-performance issue. Aggregate populations can reduce filterability, narrow process margins, and increase operational variability.
In DSP, low-pH viral inactivation and ultrafiltration/diafiltration (UF/DF) are among the most aggregation-sensitive operations. Both can promote multiple aggregation pathways that significantly affect downstream filtration. This report summarizes current understanding, highlights analytical limitations, and describes how modern process analytical technology, specifically SR-DLS, enables efficient inline aggregate monitoring to support fouling-risk assessment, DSP development, and manufacturing efficiency.
Introduction
Protein aggregation remains a central challenge in biopharmaceutical manufacturing. Aggregation spans a continuum including reversible self-association, soluble oligomerization, insoluble aggregate formation, and subvisible particle generation, driven by conformational, colloidal, and interfacial stress.1 A substantial part of DSP complexity arises from the interplay between these aggregation pathways and their impact on both product quality and process behavior.
This process dimension is becoming increasingly important. Subvisible protein particles, including submicron species, are now recognized as important quality attributes for therapeutic proteins, and there is broad consensus that they are often monitored less closely than their significance warrants.2,3 In DSP, however, such “hidden” aggregate populations can also strongly impair processability by inducing membrane fouling, reducing throughput, increasing pressure, lowering yield, and contributing to batch-to-batch variability.
Low-pH viral inactivation (Low-pH-VI) and UF/DF are among the most aggregation-sensitive unit operations in biopharmaceutical DSP, whereas virus filtration and post-UF/DF polishing filtration are often most strongly affected by the aggregate and impurity burden generated upstream,4,5,6 as summarized in Figure 1. To optimize process performance, it is thus insufficient to monitor membrane performance alone; aggregate formation must also be monitored and quantified where it is most likely to occur.
Aggregation hotspots and filtration consequences in DSP
Low-pH-VI is a critical safety step, but it also imposes significant physicochemical stress on protein formulations. As shown in [5], aggregation during low-pH VI can arise from the acidic hold itself, but also from transient local over-acidification and from neutralization effects. In particular, a process that appears well controlled at small scale may generate substantially greater aggregate burden at larger scale because of differences in mixing and local pH exposure. Such aggregate burden may persist into later membrane operations, contributing to throughput loss, fouling, and reduced robustness.6 Besides safety purposes, viral inactivation is thus also a significant upstream source of downstream filtration risk.
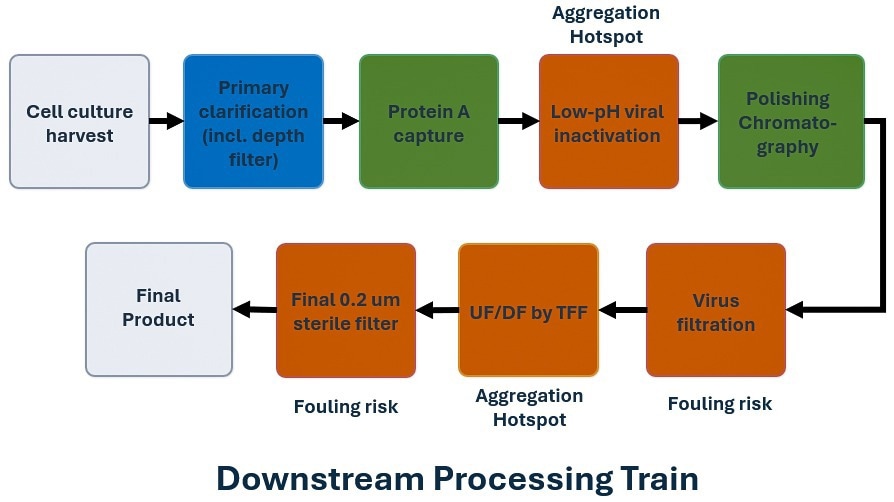
Figure 1. Protein downstream processing schematic highlighting low-pH viral inactivation and UF/DF as major aggregation-sensitive steps, and virus filtration and post-UF/DF filtration as the main fouling-sensitive steps. Image Credit: InProcess-LSP
UF/DF represents the second major hotspot. In this step, proteins are exposed to elevated concentration, long residence times, repeated pump passages, tubing contact, and membrane interfaces. UF/DF is thus one of the most aggregation-sensitive and operationally demanding operations in DSP.1 As shown in [7] and [8], peristaltic pumping alone can induce protein particle formation under process-relevant conditions and thus contribute to reduced UF/DF performance. This risk can be further amplified by membrane interactions and protein-protein interactions at high concentration.
Among filtration steps, virus filtration is particularly sensitive to feed quality. Isu et al.4 identified aggregates, host-cell proteins, endotoxin, and charge variants as major contributors to throughput loss and variability in virus filtration. Although prior polishing steps can reduce aggregate levels, the residual aggregate burden and its size distribution may still remain critical. The operational consequences of upstream aggregation thus become most evident only at the virus-filtration step.
Why aggregate monitoring remains difficult in current workflows
A central challenge is that relevant aggregates span a broad size range. Early instability may first appear as a shift in the concentration of dimers, small oligomers, and higher-order aggregates in the submicron regime. The latter (typically ≳ 30 nm) may form from small oligomers, depending on the protein, stress, and oligomer structure, and for virus filtration are often most relevant to detect.9 In addition, rarer larger particles can be more directly associated with membrane fouling, product-quality concerns, and manufacturability. Monitoring only part of this size spectrum thus risks missing the transition from early instability to filtration-relevant foulant formation.
Conventional aggregate characterization relies on multiple orthogonal methods.10 Soluble aggregate assays and particle-tracking, counting, or imaging all provide important information, but each covers only part of the full size spectrum.1,2 For example, SEC methods are often less suited for sizes ≳ 20–30 nm (a few thousand kDa), for which resolution, recovery, and reliability decrease. AF4 extends the SEC regime,11 but generally also extends method development and run times. Moreover, these methods are performed offline, requiring sample withdrawal, preparation, and significant analysis time. This makes them poorly suited for rapidly linking instabilities in low-pH VI or UF/DF to downstream process consequences.
In practice, many teams rely on hydraulic indicators such as flux decline, pressure increase, and throughput-to-endpoint when evaluating membrane performance. While indispensable, these signals are typically lagging indicators: once a clear hydraulic change is observed, foulants have already formed and membrane capacity has already been consumed. The lack of early filtration-risk signals is particularly important for continuous DSP, where substantial development effort has been invested in recent years.12,13,14
SR-DLS as an inline PAT method for aggregate monitoring
One way to address this gap is to introduce process analytical technology (PAT) that monitors aggregate formation directly inline. Spatially Resolved Dynamic Light Scattering (SR-DLS), implemented in NanoFlowSizer instruments, is specifically designed for real-time particle-size and aggregate characterization under process-relevant flow conditions. The NanoFlowSizer combines two complementary modes: PhaSR-DLS for sensitive submicron-size characterization (down to ~5 nm) and Large-Particle Detection (LPD) for monitoring rare, larger particles within a single non-invasive setup.15,16,17,8
In DSP, this combination enables the detection of some main aggregate types that can affect filtration. Submicron aggregates are particularly important because many early aggregation events in low-pH VI and UF/DF occur well before larger subvisible particles appear. SR-DLS thus can move the analytical window closer to the origin of the problem and reduce dependence on delayed offline sampling. It is not intended to replace orthogonal methods, but rather to complement them by providing earlier and more process-relevant insight. In low-pH VI, this means earlier detection of whether acidification, hold, or neutralization is pushing the protein toward instability. In UF/DF, it allows monitoring of whether pumping, concentration, and recirculation are driving the system toward a fouling-prone state before membrane performance is affected.
The applicability of the NanoFlowSizer as a monitoring tool is well illustrated by its integration into a peristaltic pumping loop, shown in Figure 2B. In this example, protein and aggregate size characteristics of a 5 mg/mL BSA solution were monitored during approximately 20 hours of circulation. At the present small scale, a flow rate of about 3.6 L/h was used, but with appropriate flow cells, flows exceeding 200 L/h – relevant to small-scale manufacturing TFF – can also be measured.
![A) PhaSR-DLS particle size distributions at several timepoints in the pumping experiment. Inset: snapshot of an LPD image series at t = 10 h showing sparse particles >1 μm. B) Schematic of the peristaltic circulation loop with NanoFlowSizer integrated. C) Scatter intensity arising from the submicron aggregate population versus circulation time. D) Concentration of >1 μm aggregates from LPD versus circulation time. Further details are described in [8].](https://www.news-medical.net/images/appnotes/ImageForAppNote_6294_17768460826128326.jpg)
Figure 2. A) PhaSR-DLS cumulative particle size distributions at several timepoints in the pumping experiment. Inset: snapshot of an LPD image series at t = 10 h showing sparse particles > 1 μm. B) Schematic of the peristaltic circulation loop with NanoFlowSizer integrated. C) Scatter intensity arising from the submicron aggregate population versus circulation time. D) Concentration of > 1 μm aggregates from LPD versus circulation time. Image Credit: Rut Besseling, et al. (2025)
The cumulative particle size distributions in Figure 2A show both native BSA and aggregates in the 150–400 nm range. During circulation, the intensity fraction of the latter increases at the expense of native BSA. This can be attributed to mechanical stress from the peristaltic pump rollers, causing adsorption and subsequent desorption of aggregates from the tubing. A more direct measure of aggregate burden is the population-specific scatter intensity. As shown in Figure 2C, this quantity increases approximately linearly with circulation time. Although the corresponding aggregate weight fraction remains small (< 0.1 %), such fractions in UF/DF may still promote cake- or gel-like deposition and significantly affect performance.
In parallel, LPD data (inset in Figure 2A) can be used to monitor the presence of larger particles. The estimated concentration of particles exceeding 1 μm, shown in Figure 2D, again increases approximately linearly. Although these larger particles are much less numerous than submicron aggregates, the associated burden may still be substantial. Overall, these data illustrate how aggregate characterization across multiple size regimes can provide quantitative indicators of filtration risk and thereby support process development.
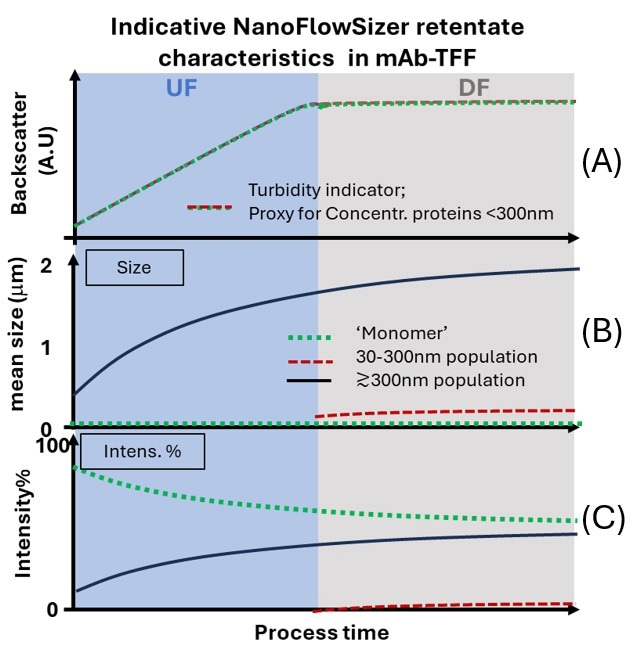
Figure 3. Schematic example of inline particle-size and concentration monitoring in a retentate stream during UF/DF. A) NanoFlowSizer backscatter signal as indicator of protein concentration. B) Mean size of different populations in the particle-size distribution, including monomers and two aggregate populations. C) Evolution of the intensity fractions of the different populations of the size distribution. Image Credit: InProcess-LSP
A more direct UF/DF-relevant example is shown schematically in Figure 3, where SR-DLS is integrated inline in the retentate stream. In Figure 3A, the backscatter intensity serves as an indicator of protein concentration, showing a typical linear increase during UF and a constant level during DF. For judging UF/DF performance and final-filtration risk, however, size information is more important. The trends shown in Figure 3B for three populations illustrate that submicron aggregates grow mainly during UF, whereas smaller aggregates form primarily during DF buffer exchange. Indicative intensity fractions are shown in Figure 3C, allowing simultaneous monitoring of the relative burden of different populations. Such information strongly complements that of spectroscopic and other monitoring tools in UF/DF,18 being more targeted towards identifying main causes of performance loss and mitigation strategies.
Implications for filtration optimization and process economics
Early aggregate monitoring in downstream processing turns filtration control from a reactive activity into a preventive one. By detecting aggregate formation before clear hydraulic symptoms appear, it can reduce failed or marginal runs, support tighter and less conservative process design, lower unnecessary membrane overdesign, and accelerate troubleshooting when performance shifts.
This is directly relevant to both virus filtration and UF/DF. In virus filtration, earlier knowledge of aggregate burden can help improve throughput and reduce fouling risk. As shown in [19], process and filter adjustments may enhance viral filtration throughput significantly. In UF/DF, it can reveal stress-driven instability early enough to reduce avoidable fouling, unnecessary recirculation, and extended processing. It can also simplify development by reducing the number of confirmation studies needed to understand scale-up sensitivity and process-induced instability.
The result is not only greater operational efficiency, but also better protection of product yield. By limiting aggregation-related fouling and prolonged exposure to destabilizing conditions, inline aggregate monitoring can help improve recovery, consistency, and strengthen overall process robustness. Even modest gains in these areas, for example, a few percent enhanced product yield, can have meaningful economic value. Estimates and insights from leading industries support that the value of slight yield improvement resulting from inline aggregate detection may well surpass that of resulting operational savings.
Conclusion
Optimizing filtration in biopharma DSP requires visibility into where aggregates form and how they later affect membrane performance. Low-pH viral inactivation and UF/DF are two of the most aggregation-sensitive operations in DSP, while virus filtration and final polishing filtration are among the steps most vulnerable to the aggregate burden they generate.
This is where inline SR-DLS can make a meaningful difference. By monitoring aggregate formation directly in flow, SR-DLS brings visibility to destabilization at the point where fouling risk begins to develop, rather than only after pressure rise, flux loss, or throughput decline becomes apparent. Its ability to follow submicron aggregate growth in real time, and in combination with LPD to track larger particles, provides process teams with earlier and more actionable insight into the effects of acidification, neutralization, pumping, concentration, and recirculation.
Used as an inline PAT tool complementary to orthogonal off-line methods, SR-DLS can therefore strengthen fouling diagnostics, improve process understanding, and support more robust filtration strategies during development, scale-up, and manufacturing. It can also help reduce failed or marginal filtration runs, limit membrane overdesign, shorten troubleshooting cycles, and improve yield, creating measurable economic benefit alongside technical risk reduction.
References
- Mahler, H.-C., et al. (2009). Protein aggregation: Pathways, induction factors and analysis. Journal of Pharmaceutical Sciences, 98(9), pp.2909–2934. DOI: 10.1002/jps.21566. https://linkinghub.elsevier.com/retrieve/pii/S0022354916330775.
- Singh, S.K., et al. (2010). An Industry Perspective on the Monitoring of Subvisible Particles as a Quality Attribute for Protein Therapeutics. Journal of Pharmaceutical Sciences, 99(8), pp.3302–3321. DOI: 10.1002/jps.22097. https://jpharmsci.org/article/S0022-3549(15)32519-3/abstract.
- Carpenter, J. F., et al. (2009). Overlooking Subvisible Particles in Therapeutic Protein Products: Gaps That May Compromise Product Quality. Journal of Pharmaceutical Sciences, 98(4), 1201–1205. DOI: 10.1002/jps.21530. https://jpharmsci.org/article/S0022-3549(16)32936-7/abstract.
- Isu, S., et al. (2022). Process- and Product-Related Foulants in Virus Filtration. Bioengineering, 9(4), 155. DOI: 10.3390/bioengineering9040155. https://www.mdpi.com/2306-5354/9/4/155.
- Jin, W., et al. (2019). Protein aggregation and mitigation strategy in low pH viral inactivation for monoclonal antibody purification. mAbs, 11(8), pp.1479–1491. DOI: 10.1080/19420862.2019.1658493. https://www.tandfonline.com/doi/full/10.1080/19420862.2019.1658493.
- Tanudjaja, H. J., et al. (2022). A review of membrane fouling by proteins in ultrafiltration and microfiltration. Journal of Water Process Engineering, 50, 103294. DOI: 10.1016/j.jwpe.2022.103294. https://www.sciencedirect.com/science/article/abs/pii/S2214714422007383?via%3Dihub.
- Deiringer, N. and Friess, W. (2022). Proteins on the Rack: Mechanistic Studies on Protein Particle Formation During Peristaltic Pumping. Journal of Pharmaceutical Sciences, (online) 111(5), pp.1370–1378. DOI: 10.1016/j.xphs.2022.01.035. https://jpharmsci.org/article/S0022-3549(22)00045-4/fulltext.
- Rut Besseling, et al. (2025). Protein aggregation by peristaltic pumping: Submicron and subvisible particles monitored with a single instrument. (online) doi:https://doi.org/10.13140/RG.2.2.29621.18403. https://www.researchgate.net/publication/390565063_PROTEIN_AGGREGATION_BY_PERISTALTIC_PUMPING_SUBMICRON_AND_SUBVISIBLE_PARTICLES_MONITORED_WITH_A_SINGLE_INSTRUMENT?channel=doi&linkId=67f44f5449e91c0feae8451b&showFulltext=true.
- Knight, M.J., et al. (2022). The impact of forced degradation conditions on mAb dimer formation and subsequent influence on aggregation propensity. mAbs, 14(1). DOI: 10.1080/19420862.2022.2127172. https://www.tandfonline.com/doi/full/10.1080/19420862.2022.2127172.
- Bansal, R., Gupta, S. and Rathore, A.S. (2019). Analytical Platform for Monitoring Aggregation of Monoclonal Antibody Therapeutics. Pharmaceutical Research, 36(11). DOI: 10.1007/s11095-019-2690-8. https://link.springer.com/article/10.1007/s11095-019-2690-8.
- Marioli, M. and Kok, W.T. (2019). Recovery, overloading, and protein interactions in asymmetrical flow field-flow fractionation. Analytical and Bioanalytical Chemistry, 411(11), pp.2327–2338. DOI: 10.1007/s00216-019-01673-w. https://link.springer.com/article/10.1007/s00216-019-01673-w.
- São Pedro, M.N., et al. (2021). White paper on high‐throughput process development for integrated continuous biomanufacturing. Biotechnology and Bioengineering. DOI: 10.1002/bit.27757. https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/abs/10.1002/bit.27757.
- Rathore, A.S., et al. (2015). Continuous Processing for Production of Biopharmaceuticals. Preparative Biochemistry and Biotechnology, 45(8), pp.836–849. DOI: 10.1080/10826068.2014.985834. https://www.tandfonline.com/doi/abs/10.1080/10826068.2014.985834.
- Rathore, A.S., et al. (2021). Challenges in process control for continuous processing for production of monoclonal antibody products. Current Opinion in Chemical Engineering, 31, p.100671. DOI: 10.1016/j.coche.2021.100671. https://www.sciencedirect.com/science/article/abs/pii/S2211339821000034?via%3Dihub.
- Besseling, R., et al. (2019). New unique PAT method and instrument for real-time inline size characterization of concentrated, flowing nanosuspensions. European Journal of Pharmaceutical Sciences: Official Journal of the European Federation for Pharmaceutical Sciences, (online) 133, pp.205–213. DOI: 10.1016/j.ejps.2019.03.024. https://www.sciencedirect.com/science/article/abs/pii/S0928098719301332?via%3Dihub.
- Hermes, M., et al. (2024). A new method for non-invasive or inline detection of aggregates and oversized particles in nanosuspensions. AZo News-Medical. Available at: https://www.news-medical.net/whitepaper/20241106/A-new-method-for-non-invasive-or-inline-detection-of-aggregates-and-oversized-particles-in-nanosuspensions.aspx.
- Koumakis, N., et al. (2024). PhaSR-DLS’: A new advancement in Spatially Resolved DLS for enhanced inline and off-line nanoparticle sizing. AZo Nano. Available at: https://www.azonano.com/article.aspx?ArticleID=6749.
- Rolinger, L., et al. (2020). Multi-attribute PAT for UF/DF of Proteins - Monitoring Concentration, particle sizes, and Buffer Exchange. Analytical and Bioanalytical Chemistry, 412(9), pp.2123–2136. DOI: 10.1007/s00216-019-02318-8. https://link.springer.com/article/10.1007/s00216-019-02318-8.
- J. De Souza, K. Scott, and P. Genest, “Virus-filtration process development optimization: The key to a more efficient and cost-effective step,” Bioprocess Int., vol. 14, no. 4, 2016.
About InProcess-LSP
InProcess-LSP, headquartered in Oss at Pivot Park, is a rapidly growing, innovative company founded in 2014. Backed by a team of in-house experts - comprising physicists, chemists, and software engineers - InProcess-LSP is at the forefront of nanotechnology solutions. The company’s leading product, the NanoFlowSizer, is a cutting-edge instrument designed to deliver inline, real-time measurements of nanoparticles in solution, making it indispensable across various industries.
Utilizing Spatially Resolved Dynamic Light Scattering (SR-DLS) technology, the NanoFlowSizer enables accurate characterization of nanoparticles in flowing liquids, providing critical data such as hydrodynamic diameter, polydispersity index (PDI), and D90 within seconds.
This state-of-the-art instrument empowers both scientists and industries by offering a robust solution for analyzing nanoparticle properties, paving the way for breakthroughs in product development, improved formulations, and pioneering applications.
Innovators in process analytical technology and nanoparticle characterization.
With their strong background in process analytics as well as many years of academic and industrial experience InProcess offer a highly skilled and experienced team of scientists and process specialists addressing the needs of your PAT and nanotechnology challenges.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.