The Prospect T2 is a cutting-edge high-frequency ultrasound system tailored for in vivo preclinical imaging of small animals, such as mice and rats.
This compact and economically viable tablet-based system delivers high-resolution images (up to 30 µm) and features advanced functionalities to track hemodynamic changes and visualize anatomical structures in real-time.
Equipped with standard ultrasound features such as B-mode, M-mode, Color/Power Doppler, Pulsed Wave Doppler, Tissue Doppler, and Contrast imaging, the Prospect T2 provides a diverse range of imaging options suitable for various applications.
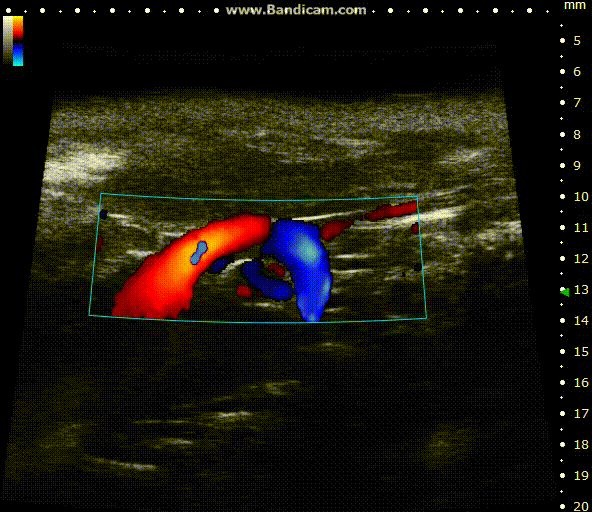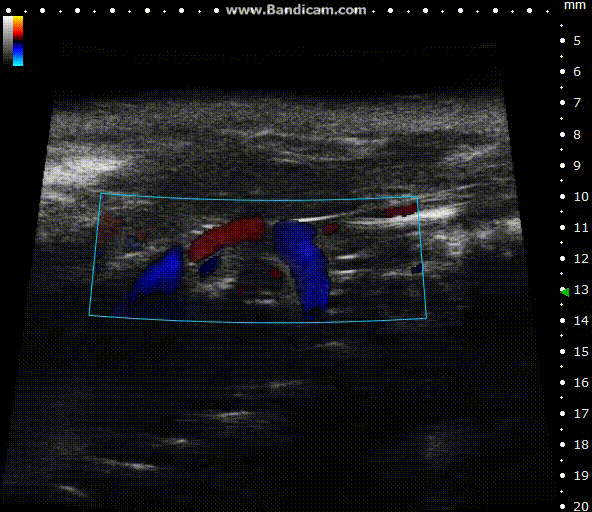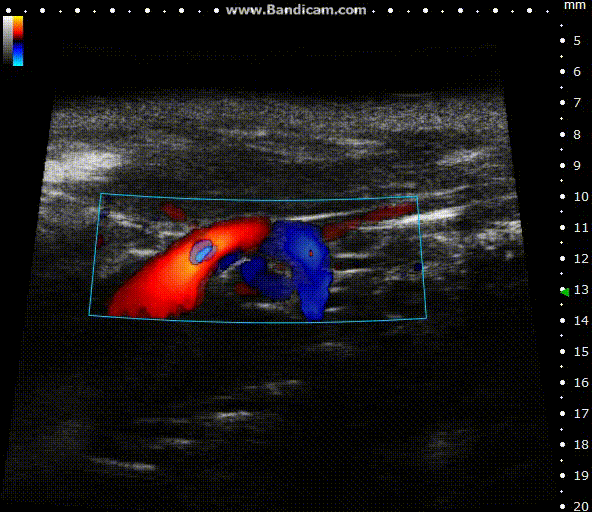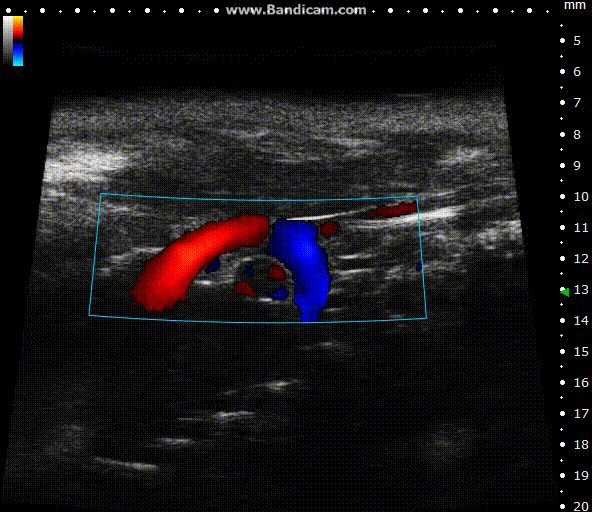
Image Credit: Scintica Instrumentation Inc
The system grants access to the RAW digital RF data, enabling the generation of quantitative results and facilitating offline data analysis.
The scanning platform included is designed with a heated surface suitable for mice or rats and features integrated physiological monitoring, which simplifies the positioning of the animal and enhances the quality of the images obtained.
There are three distinct single-element probes available for selection, tailored to the specific animal being imaged and the anatomical targets of interest. The ergonomic design of the animal handling apparatus promotes easy positioning and efficient cleaning between imaging sessions, while the analytical tools and measurement packages expedite the process of result generation.
Features and benefits
The Prospect T2 is an economical high-frequency preclinical ultrasound system that can be utilized in a laboratory setting. There is no longer a requirement for a central imaging facility.
Standard imaging modes
- Contrast Mode (Linear and non-linear/harmonic)
- Pulsed Wave, Tissue, Color, Power Doppler
- B-Mode
- M-Mode
RAW data and import scripts
Researchers seeking to examine digital RF data from any mode can readily access it and import it into third-party software for additional analysis.
Add-on hardware/software options
- Shear Wave Elastography/Acoustic Radiation Force push probe along with software integration
- Image-Guided Injection Mount, accompanied by software for image guidance
- 3D Motor, encompassing software for volumetric measurements
Probes and platforms
Probe options:
Animal handling platforms:
Compact and small footprint
A tablet-based acquisition computer and a compact scanning platform.
The system is designed for easy mobility and does not require placement in a central facility.
If required, the system can be placed in a biosafety cabinet, providing assistance to researchers working with immunocompromised animals or those facing other concerns.
Intuitive workflow with touch screen
Preclinical researchers have the ability to promptly initiate their studies, obtain and archive images from the different available modalities, analyze their data, and conduct the necessary measurements.
(Keyboard and mouse are also included)
Comprehensive measurement packages
Users are able to swiftly conduct measurements, compile data reports, and produce quantifiable results from images and spectrograms.
Analysis can be carried out on the system, or alternatively, on an offline analysis workstation.
Simplified image guided injection mount
Once the probe and injection mount are connected, the user can adjust the probe/needle as necessary to carry out the injection, provided that the needle and imaging plane are properly aligned.
Non-linear contrast agent imaging (First harmonic)
This enhances the sensitivity to microbubble contrast agents without the need for reference subtraction in linear contrast agent imaging.
It also improves signal sensitivity, reducing the likelihood of motion artifacts in the images. This capability is possible with both the 20 MHz and 40 MHz probes.
ECG gated kilohertz visualization
The ECG gated kilohertz mode enables users to obtain B-mode images of the heart at rates of up to 400 frames per second.These images offer exceptional detail of cardiac motion and can be used with any of the conventional measurement tools.
Imaging modes
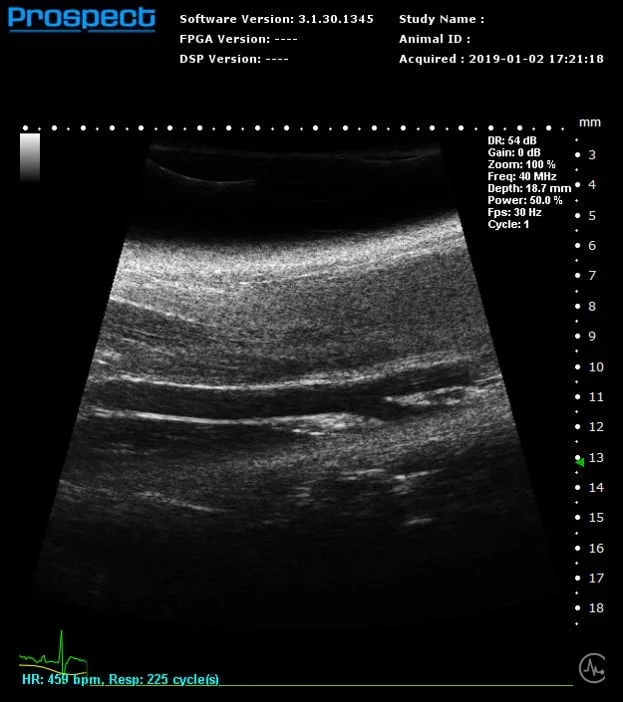
B-Mode
Mouse carotid artery – B-Mode
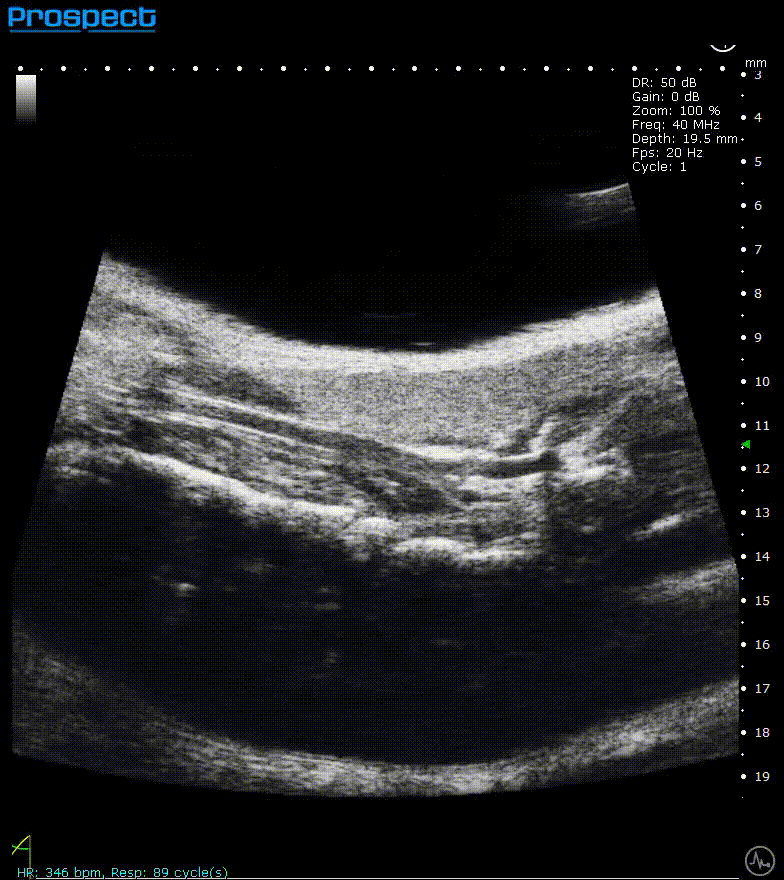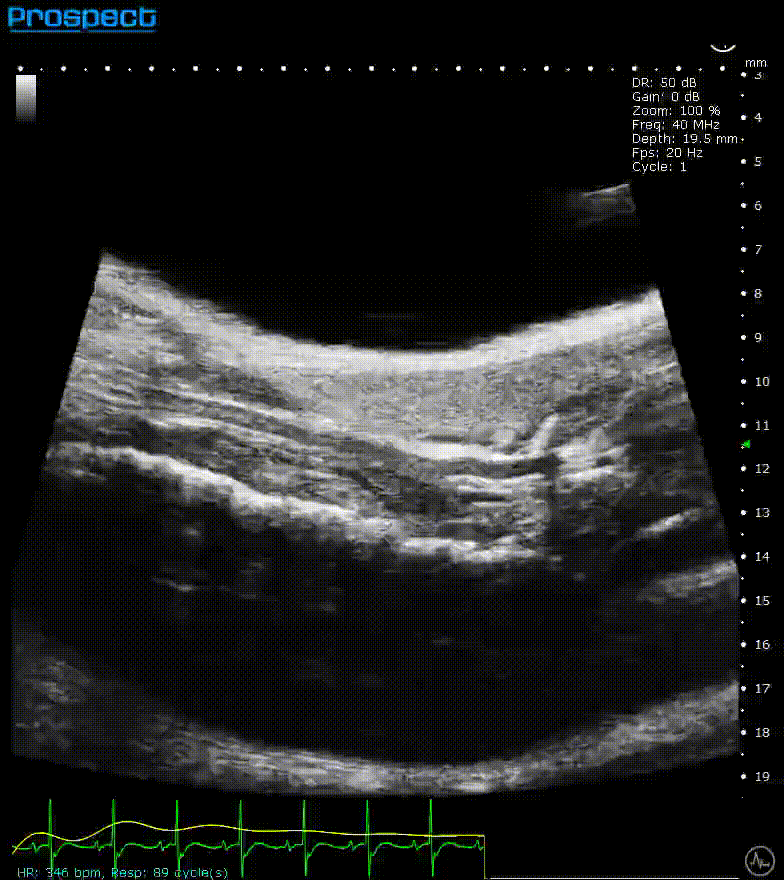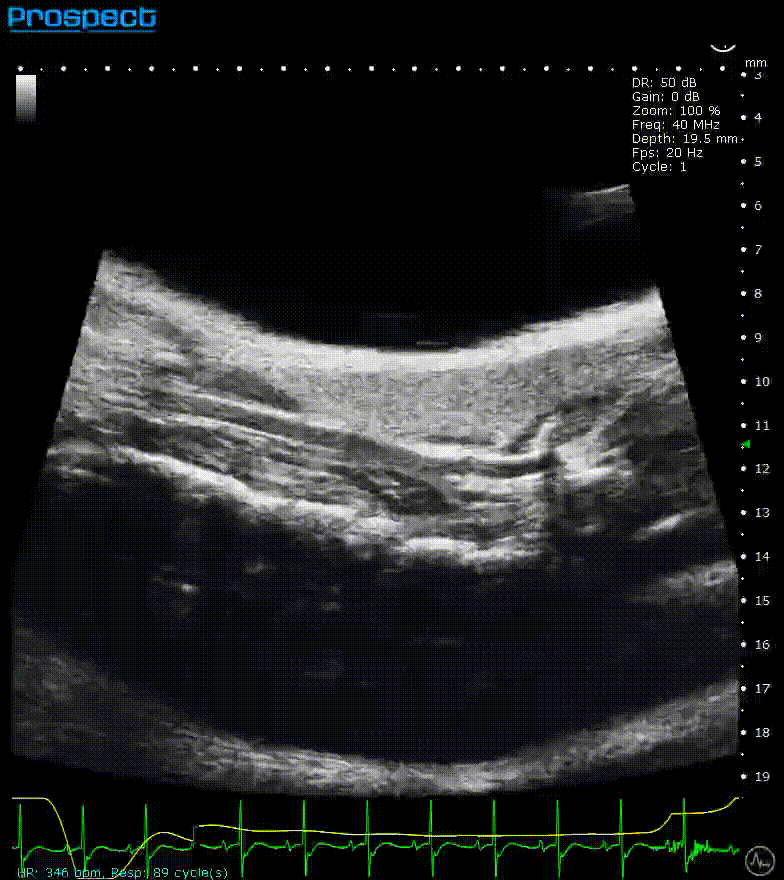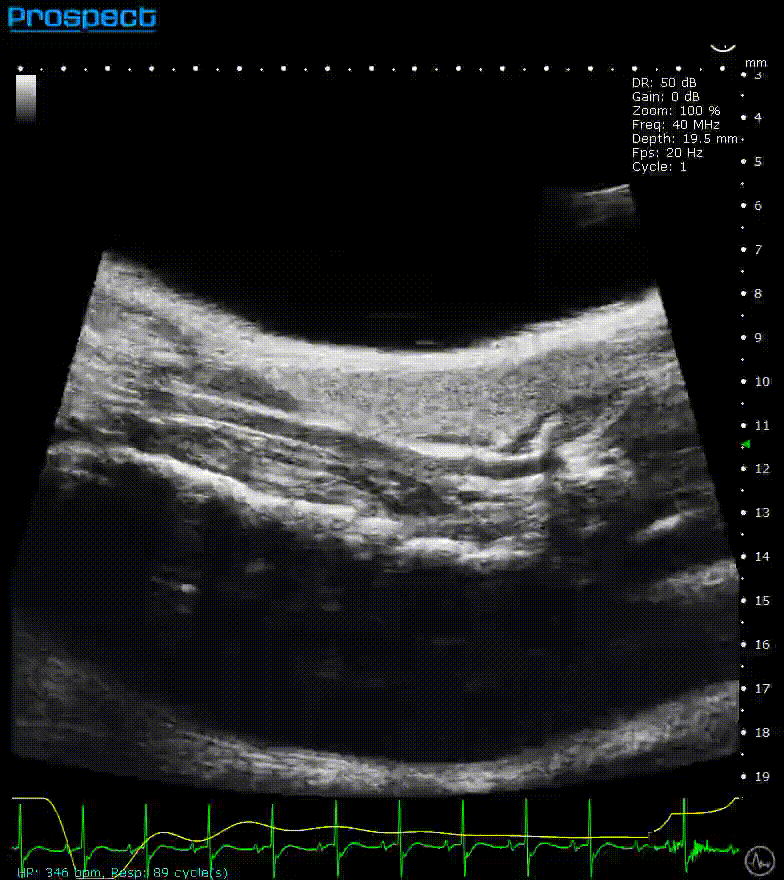
Image Credit: Scintica Instrumentation Inc
B-mode, also known as brightness mode, refers to a two-dimensional grayscale image that is presented in real time when the probe makes contact with the ultrasound gel applied to the imaging target. This mode allows for the visualization and measurement of anatomical structures, including the beating heart, abdominal organs, tumors, muscles, developing embryos, and more.
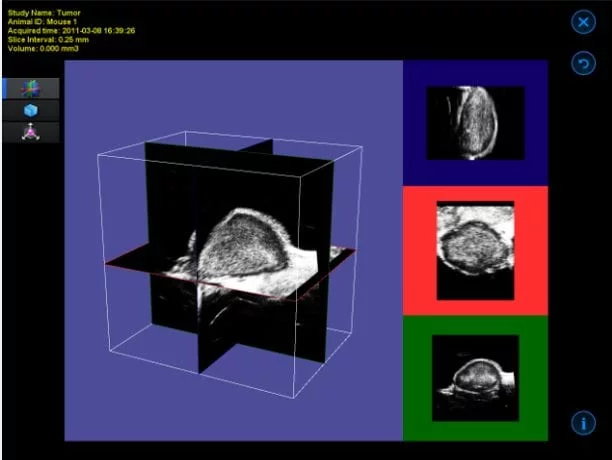
3D B-Mode
Mouse mammary fat pad tumor – 3D B-Mode
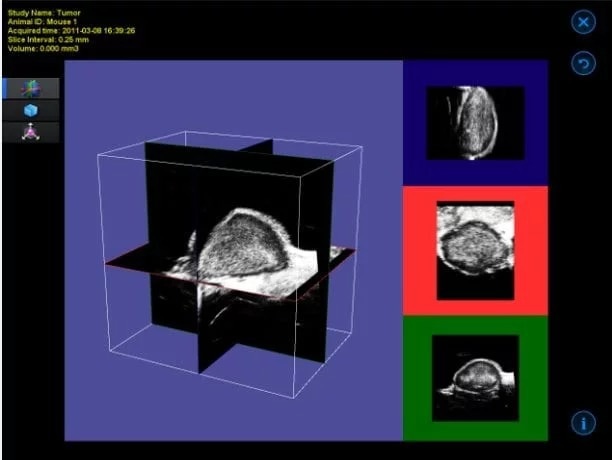
Image Credit: Scintica Instrumentation Inc
The 3D motor connects with the imaging platform to accurately position the animal bed along with the imaging subject beneath the probe. This specific form of 3D imaging is not gated by ECG, making it appropriate for abdominal organs, tumors, and similar structures.
The acquisition software provides the user with options to determine the distance the motor will traverse and the size of each step. Subsequently, the obtained imaging slices are reconstructed into a three-dimensional image.
The visualization and analysis software enables the user to navigate through the acquired image, observe it as a 3D surface, and perform volume measurements.
ECG-gated kilohertz visualization
Mouse left ventricle in long axis – B-Mode with 30 fps
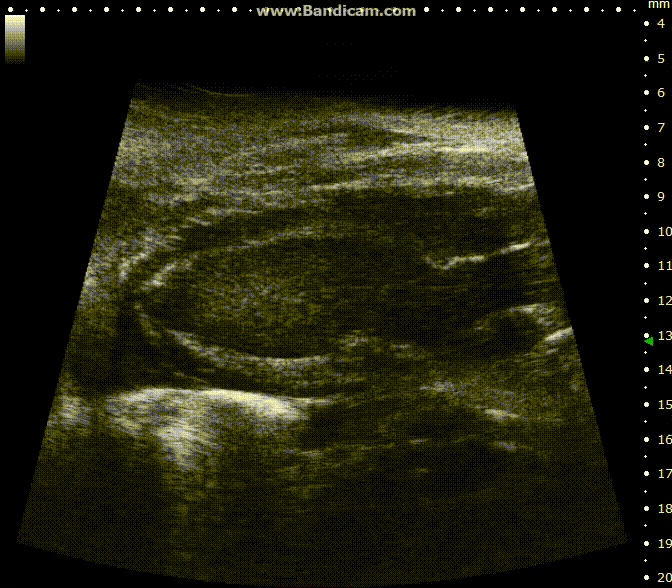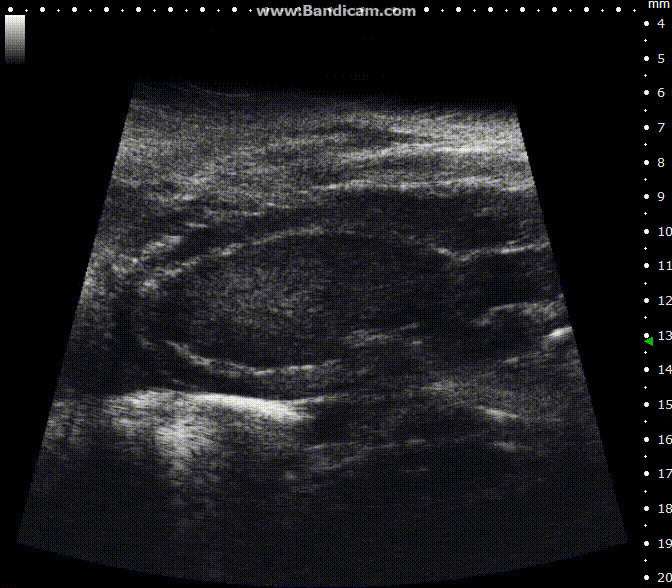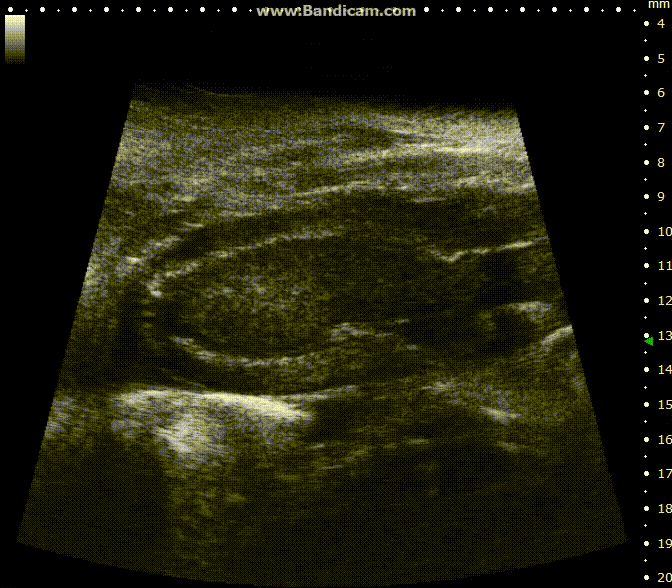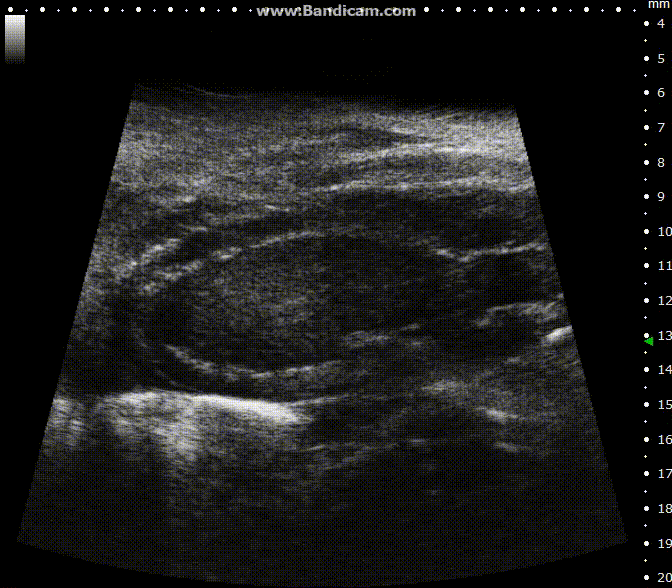
Image Credit: Scintica Instrumentation Inc
The ECG gated kilohertz visualization mode is a gated method designed to enhance frame rates during heart imaging using a single element transducer. This technique achieves a maximum frame rate of 400 fps.
There are settings available to regulate the extent of respiratory movement artifacts permitted in the image. In the image above, all respiratory artifacts are visible. Note that obtaining these images decreased acquisition time.
ECG-gated kilohertz visualization
Mouse left ventricle in long axis – ECG- gated kilohertz visualization with 120 fps, now possible up to 400 fps
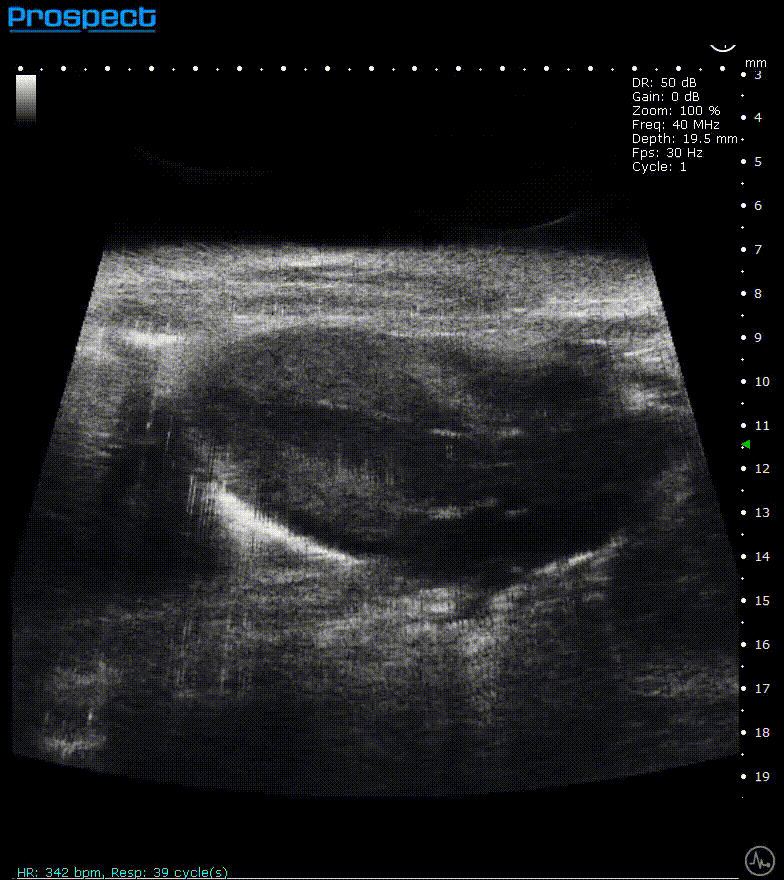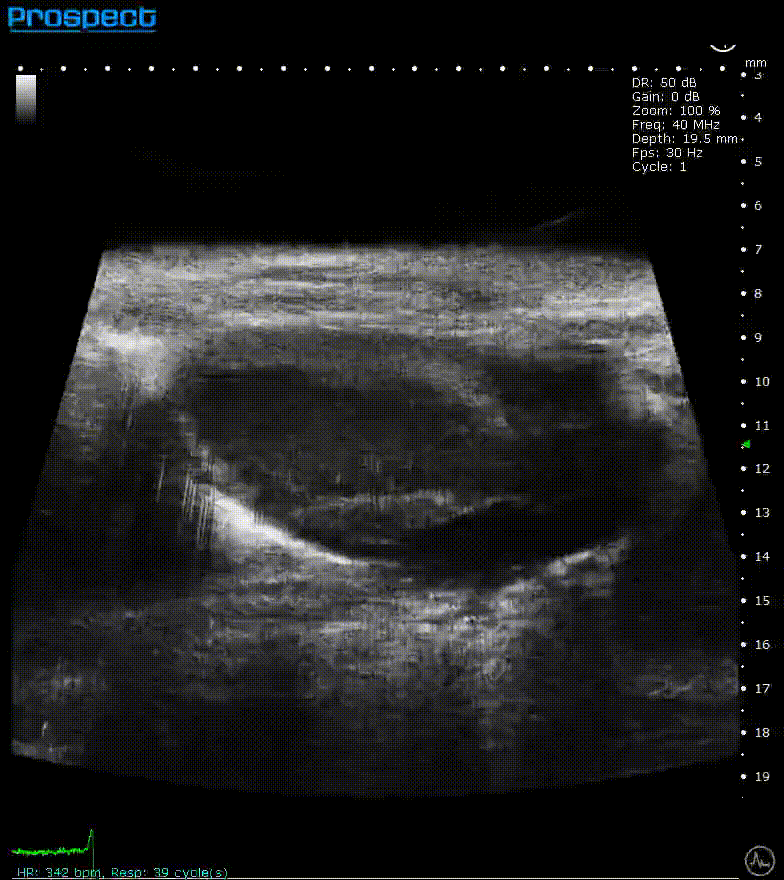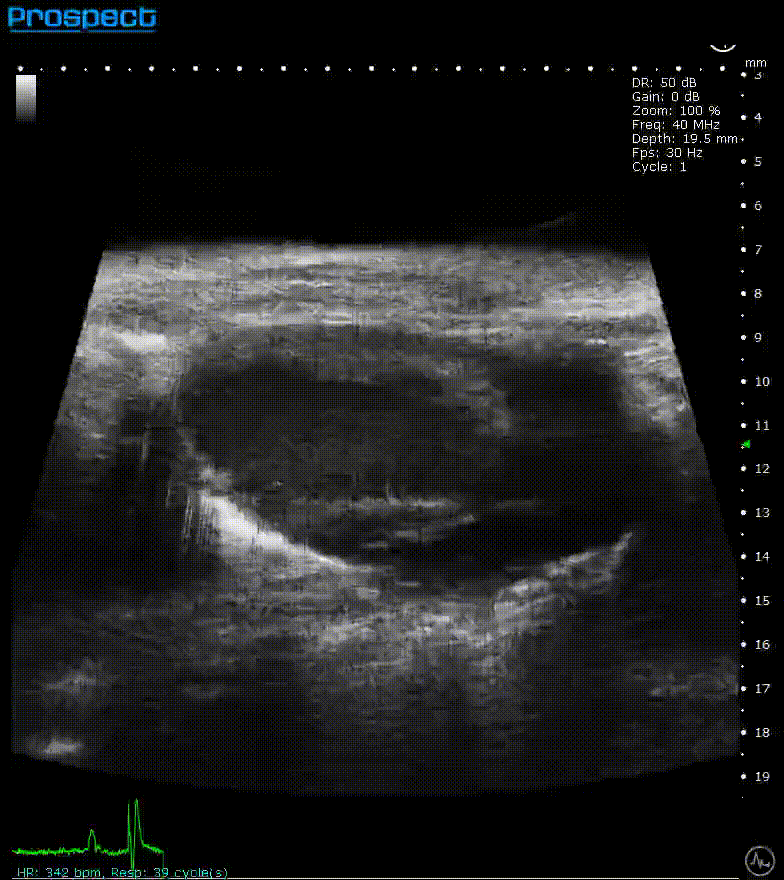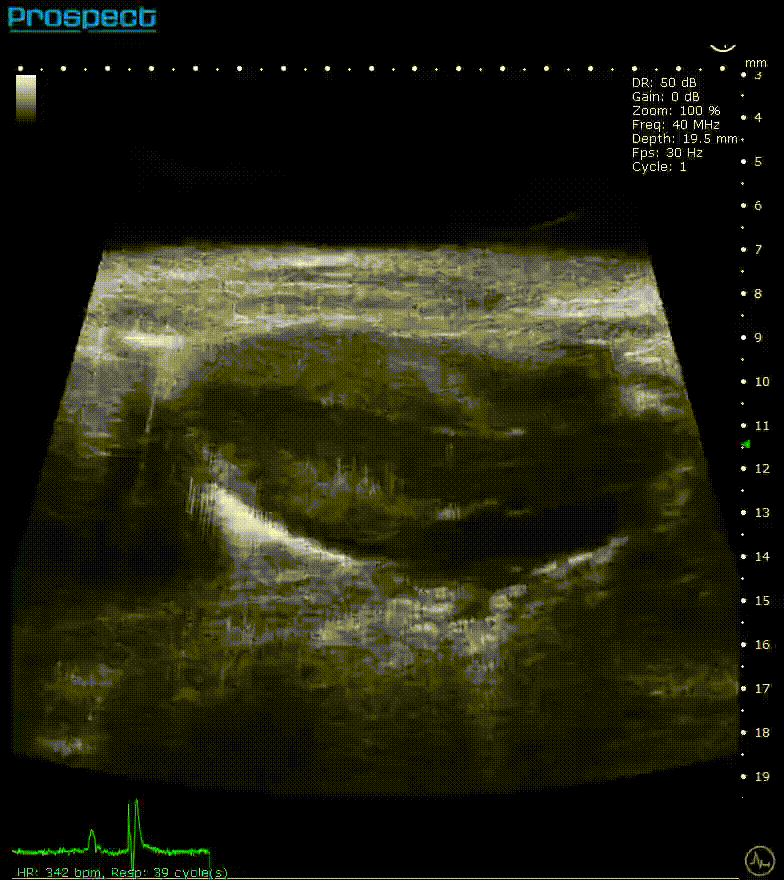
Image Credit: Scintica Instrumentation Inc
The ECG gated kilohertz visualization mode is a gated approach that allows for higher frame rates while imaging the heart with a single element transducer. The maximum framerate for this approach is 400 fps. There are choices for determining how much artifact from respiratory movement is permitted into the image. The image above shows all of the respiratory artifacts, which shortened the acquisition time.
M-Mode
Mouse left ventricle in short axis – M-Mode
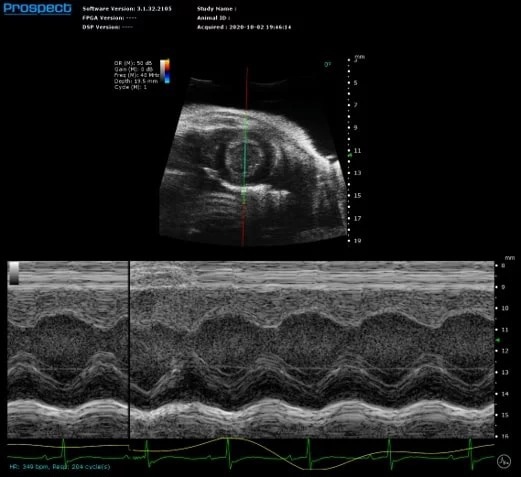
Image Credit: Scintica Instrumentation Inc
M-mode (motion mode) is a one-dimensional grey scale image used to evaluate motion of a specific object, such as the heart, along a single line. This high frame rate image allows for dimensional alterations as the myocardium moves. The sample volume is put on a 2D B-mode image that can be changed live at any time during image optimization.
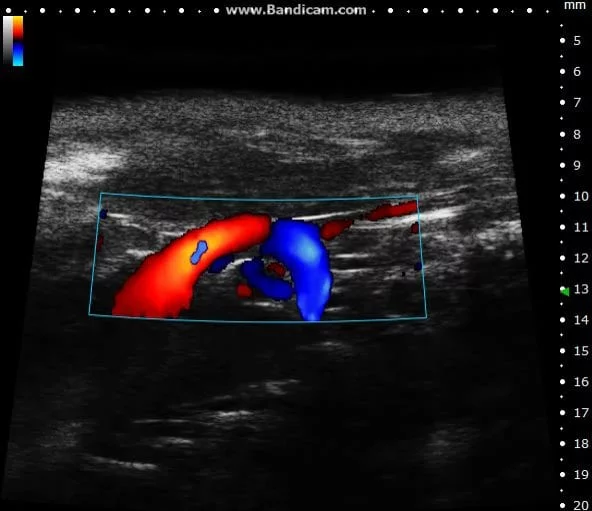
Color doppler
Mouse aortic arch – Color doppler
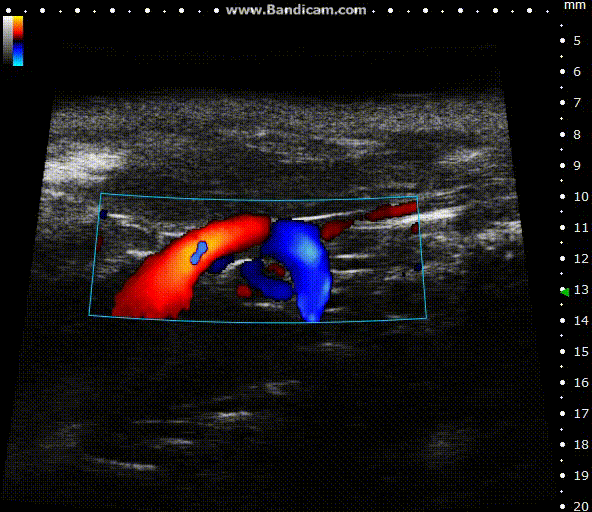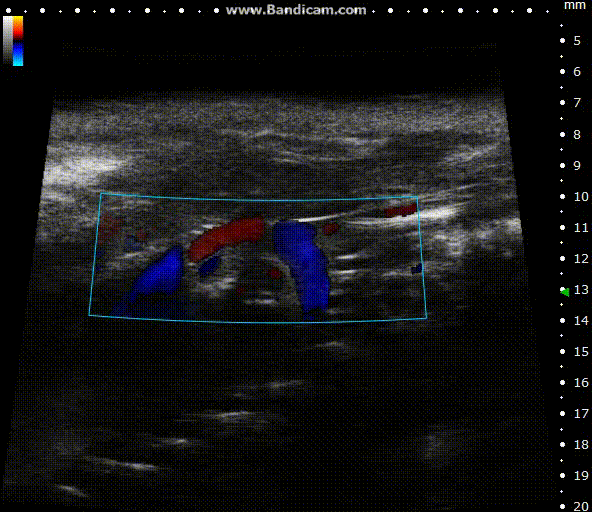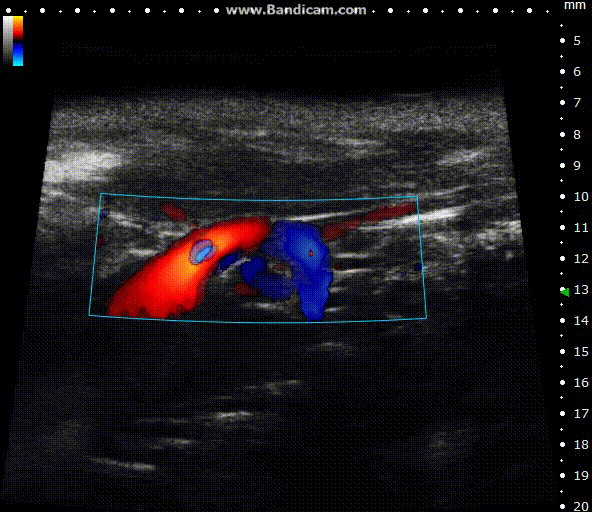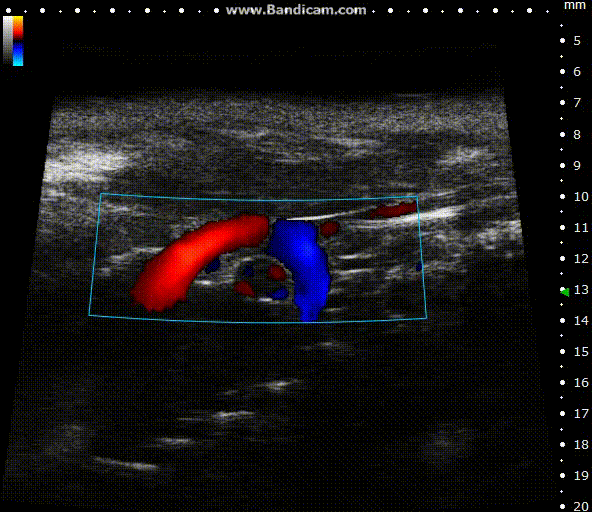
Image Credit: Scintica Instrumentation Inc
Color Doppler is a technology that overlays a heat map of blood flow over a 2D anatomical B-mode image. The directionality and velocity of the blood flow are represented by changing hues of blue and red; blood traveling away from the probe appears in blue, while blood traveling towards the probe appears red. Color Doppler pictures can be utilized to detect vasculature and guide sample volume placement for Pulsed Wave (PW) Doppler investigation.
Power Doppler
Mouse subcutaneous tumor – Power Doppler
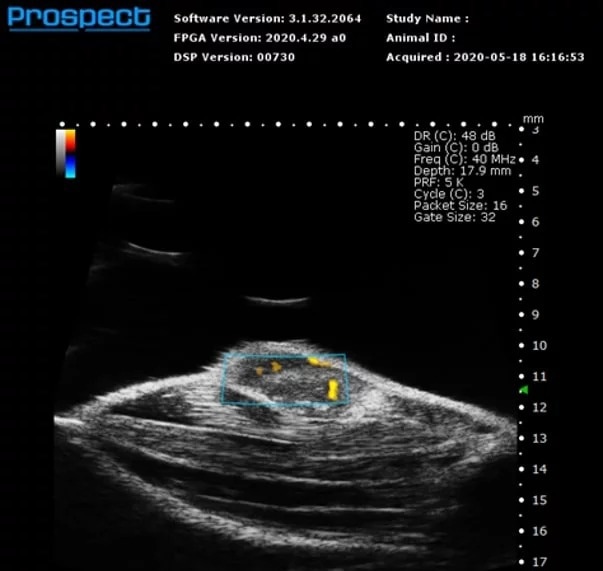
Image Credit: Scintica Instrumentation Inc
Power Doppler is a technique for superimposing a map of blood flow onto a 2D anatomical B-mode image. The Power Doppler signal does not provide directionality, but it does reveal velocity in shades of orange. This imaging modality is commonly employed to visualize general vascularity in specific anatomical targets, such as tumors.
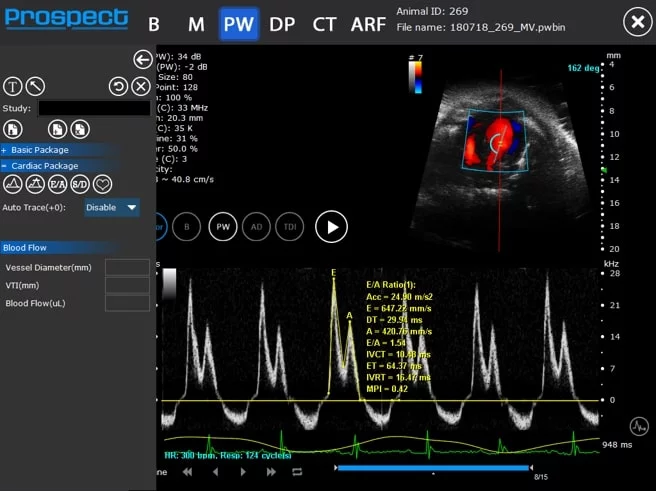
Pulsed Wave (PW) Doppler
Mouse mitral valve inflow – PW Doppler
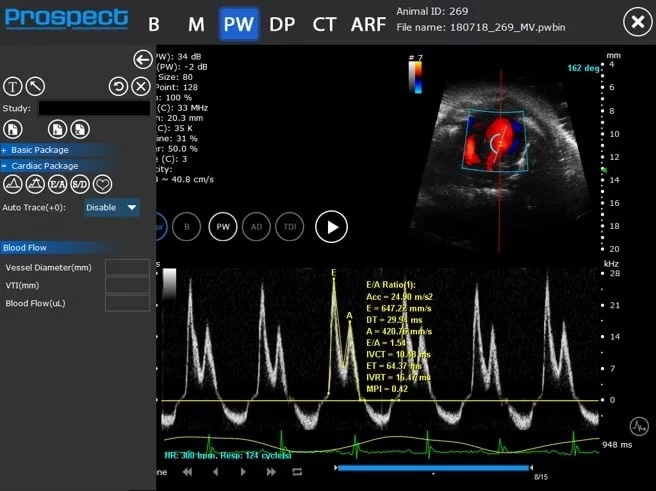
Image Credit: Scintica Instrumentation Inc
PW Doppler is utilized to assess blood flow velocity over time, represented as a spectrogram. The sample volume for this mode is generally positioned on a Color Doppler image, which can be updated in real-time for enhanced image quality and optimal sample volume placement.
In PW Doppler, users have the capability to apply an angle correction to the spectrogram data, ensuring that the displayed velocity values are precise. It is advisable to minimize this angle whenever feasible by modifying the probe's angle, to guarantee accurate and consistent measurements.
Tissue Doppler
Mouse mitral valve annulus – Tissue Doppler
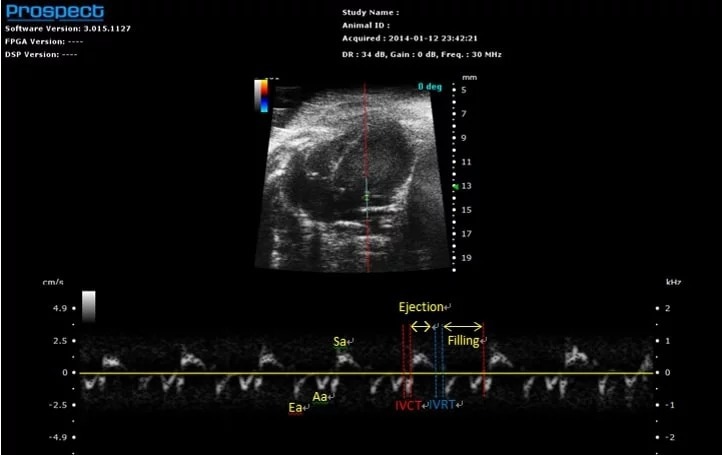
Image Credit: Scintica Instrumentation Inc
Tissue Doppler, similar to PW Doppler, presents a spectrogram over time; however, in the case of Tissue Doppler, the measurement focuses on tissue velocity rather than blood flow. The sample volume is typically positioned over the annulus of a valve leaflet, such as the mitral annulus, to assess the movement of the tissue in that region. For this mode, the sample volume is situated on a 2D B-mode image, which can be updated in real-time at any stage of image optimization.
Contrast mode
Mouse subcutaneous tumor – Non-linear contrast agent imaging with regions of interest drawn
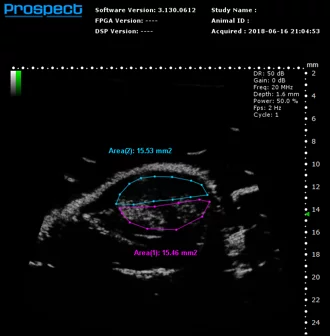
Image Credit: Scintica Instrumentation Inc
Contrast mode is employed to identify the presence of contrast agents, including USphere microbubbles (link to TrustBiosonics page), which are generally administered intravenously to the imaging subject. Two acquisition techniques are available: linear - reference subtracted, and non-linear – first harmonic imaging.
Contrast mode
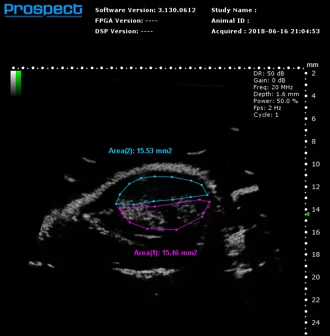
Image Credit: Scintica Instrumentation Inc
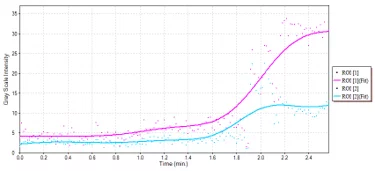
Mouse subcutaneous tumor – Non-linear contrast agent imaging Time vs. Intensity plots for drawn regions of interest. Source: Scintica Instrumentation Inc
Shear wave elastography
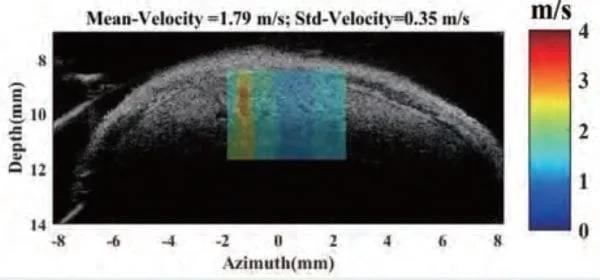
Shear Wave Elastogram showing focal lesion in mouse liver. Image Credit: Scintica Instrumentation Inc
Shear wave elastography is a method employed to examine and measure the mechanical and elastic characteristics of different tissues, such as the liver, breast, tendons, muscles, etc.
An acoustic radiation force is produced by a secondary, non-imaging component that is affixed to the side of the imaging probe. The software is utilized to configure the pulse sequence necessary for generating the shear wave; this wave propagates more rapidly through stiffer tissues and along the axes of various musculoskeletal structures.
The analysis software creates a colored elastogram that is superimposed on a B-mode image. This technique can be used to detect tissues with focal or diffuse lesions.
Models and specifications
Available probes 50 MHz, 40 MHz, 20 MHz
- PB207e – Center Frequency 20 MHz (15-25 MHz)
- PB506e – Center Frequency 50 MHz (33-50 MHz)
- PB406e – Center Frequency 40 MHz (33-50 MHz)
Available platforms mouse or rat

Image Credit: Scintica Instrumentation Inc
- Rat Platform 28.3×18.2 cm
- Mouse Platform 17.2×14.2 cm
Standard, advanced and optional imaging modes
- Contrast Mode (Linear and Non-Linear/Harmonic
- B-Mode
- Pulsed Wave / Tissue / Color / Power Doppler
- M-Mode
- RAW data available for all modes
Accessories and add-ons
3D motor and acquisition/analysis software
The 3D motor interfaces with the imaging platform to accurately position the animal bed along with the imaging subject beneath the probe. The acquisition software enables the user to specify both the distance the motor will traverse and the step size. The obtained imaging slices are reconstructed into a 3D image.
The visualization and analysis software permits the user to navigate through the acquired image, observe it as a 3D surface, and perform volume measurements.
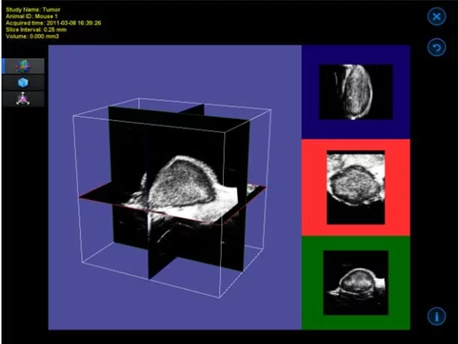
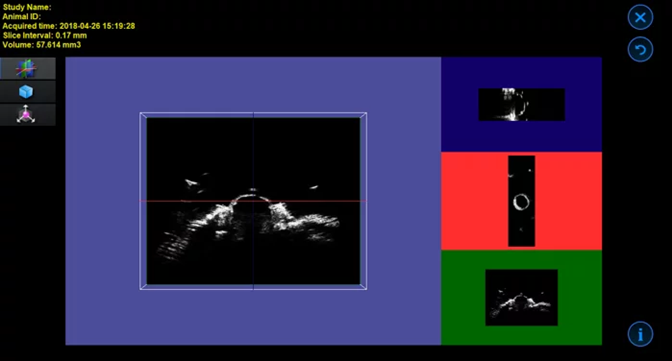
Mammary Fat Pad Tumor in Mouse – 3D Image. Image Credit: Scintica Instrumentation Inc
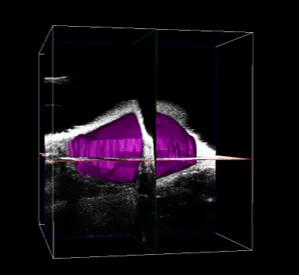
Mammary Fat Pad Tumor in Mouse – 3D Volume = 263 mm3. Image Credit: Scintica Instrumentation Inc

Mouse Hind Limb Muscle – 3D Image. Image Credit: Scintica Instrumentation Inc

Mouse Hind Limb Muscle – 3D Volume = 97.6 mm3. Image Credit: Scintica Instrumentation Inc
Image-guided needle injection mount
Image-guided needle injection is used to precisely target an injection spot, such as the myocardial, abdominal organ, vascular target, or structure within a developing embryo. The injection mount is affixed to the probe to assist in keeping the needle aligned with the imaging plane.
The image guide overlay allows users to alter the needle's trajectory and observe it. The needle is manually advanced into place while being observed on a 2D B-mode picture. The accessory can be used with either a normal syringe and needle or a pulled glass capillary auto-injector.

Injection Mount with Steel Needle and Syringe. Image Credit: Scintica Instrumentation Inc
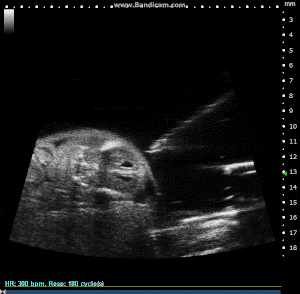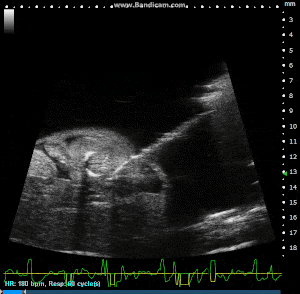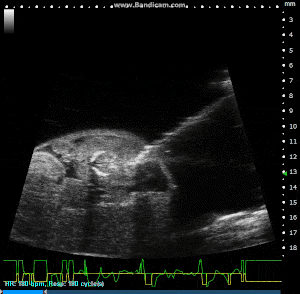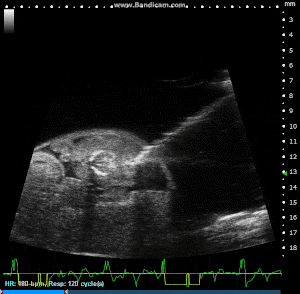
Image Guided Injection, with Pulled Glass Capillary Needle, into Externalized Mouse Embryo. Image Credit: Scintica Instrumentation Inc
Integrated shear wave elastography probe and analysis software
Shear wave elastography is a technique for analyzing and quantifying the mechanical and elastic characteristics of superficial tissues such as the liver, breast, tendons, and muscles. An acoustic radiation force is produced by a secondary, non-imaging element situated on the side of the imaging probe.
The software is used to construct the pulse sequence that generates the shear wave, which propagates more quickly through stiffer tissues and along the axis of various musculoskeletal systems. The analysis software creates a colored elastogram that is superimposed over a B-mode image. This approach is useful for identifying tissues with localized or widespread lesions.
Applications
Cardiovascular biology
The Prospect T2 is commonly used for cardiovascular imaging across various small animal models, such as mice and rats. It provides real-time images of the heart and a diverse range of blood vessels.
This system is capable of evaluating both systolic and diastolic heart function, as well as imaging and assessing blood flow through numerous vessels, including the aorta, pulmonary, carotid, renal, splenic, and femoral arteries, among others. Additionally, veins such as the vena cava and portal veins can also be evaluated.
Imaging can be conducted in utero, starting as early as day 1 post-delivery, and continuing into adulthood. The assessment of cardiovascular function may be significant as diseases progress, potentially triggered by surgical interventions, transgenic mutations, or systemic treatments involving therapies or toxic compounds.
Ultrasound imaging is a non-invasive technique, allowing for the same measurements to be taken repeatedly throughout a longitudinal study.
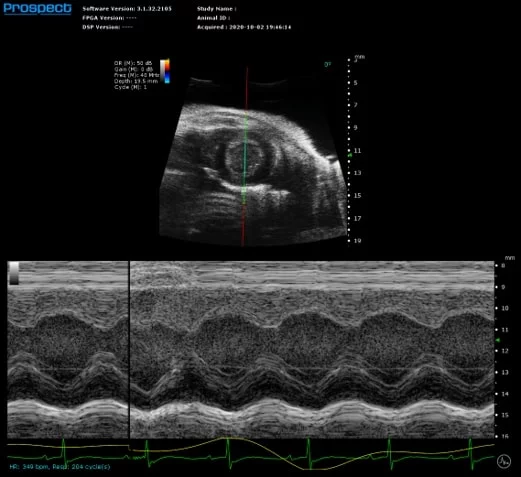
Mouse Left Ventricle in Short Axis in M-Mode with Measurements to Calculate Systolic Function. Image Credit: Scintica Instrumentation Inc
Mouse Mitral Valve Inflow in PW Doppler with Measurements to Calculate Diastolic Function. Image Credit: Scintica Instrumentation Inc
Mouse Aortic Arch in Color Doppler. Image Credit: Scintica Instrumentation Inc
Mouse Carotid Artery in B-Mode. Image Credit: Scintica Instrumentation Inc
Mouse Pulmonary Artery in PW Doppler to Assess RV Outflow. Image Credit: Scintica Instrumentation Inc
Cancer biology
The Prospect T2 is frequently utilized in a diverse range of oncology research applications. This system can be applied to xerograph, orthotopic, PDX, and transgenic tumor models across various animal models.
The system is capable of detecting tumors or metastases in numerous tissues, with the exception of the lungs, brain (due to the presence of the skull), and bones. Tumor volumes can be monitored over time, and imaging of surrounding organs and structures, such as the spleen or lymph nodes, is also possible.
Blood flow can be evaluated using Doppler techniques or microbubble contrast agents, which may additionally be employed to assess the molecular expression of specific biomarkers. Image-guided injections can facilitate the introduction of cells into a designated anatomical target or enable biopsy or aspiration procedures, thereby minimizing the necessity for invasive surgery.
Shear wave elastography can be utilized to evaluate mechanical changes within superficial organs, such as the liver, or within the tumor itself. All these techniques can be employed to monitor the progression of a lesion over time, as well as the regression or other effects resulting from therapeutic interventions. Ultrasound imaging is non-invasive, allowing for the same measurements to be taken repeatedly throughout a longitudinal study.
Mouse Live Tumor in B-Mode, Tumor Cells Orthotopically Injected. Image Credit: Scintica Instrumentation Inc
Mouse Pancreatic Tumor in B-Mode, Transgenic KPC Model. Image Credit: Scintica Instrumentation Inc
Mouse Mammary Fat Pad Tumor in 3D B-Mode, Orthotopically Injected Cells. Image Credit: Scintica Instrumentation Inc
Mouse Inguinal Lymph Node in B-Mode. Image Credit: Scintica Instrumentation Inc
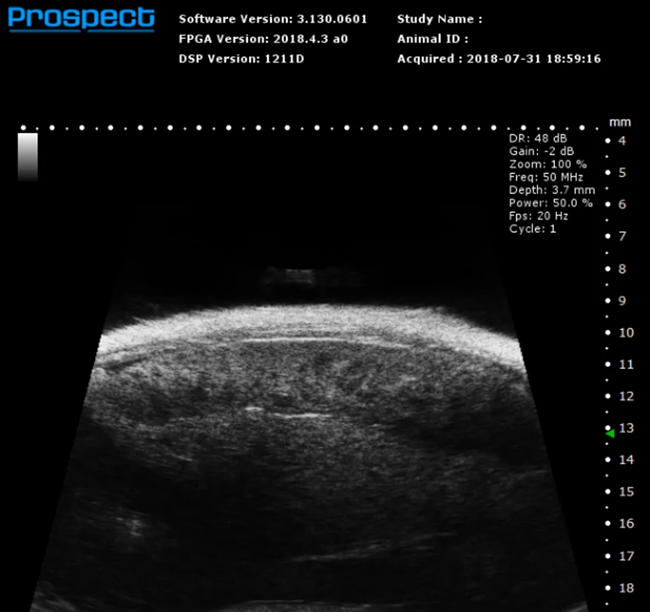
Mouse Spleen in B-Mode. Image Credit: Scintica Instrumentation Inc
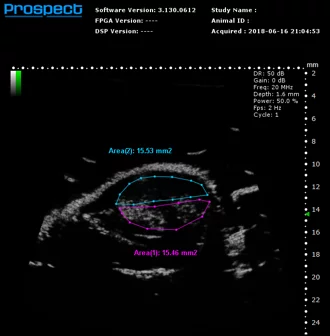
Mouse subcutaneous tumor – Non-linear contrast agent imaging with regions of interest drawn. Image Credit: Scintica Instrumentation Inc
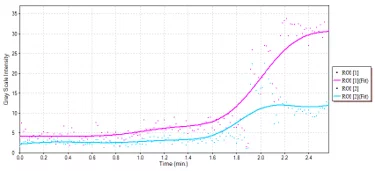
Mouse subcutaneous tumor – Non-linear contrast agent imaging Time vs. Intensity plots for drawn regions of interest. Image Credit: Scintica Instrumentation Inc
Abdominal and anatomical imaging
The Prospect T2 is capable of evaluating a range of abdominal organs and tissues, as well as various anatomical structures within the urogenital tract and musculoskeletal regions. Both organ size and position can be examined in 2D and 3D formats, while blood vessels can also be visualized and their flow evaluated.
Additionally, ultrasound can be used to image structures beyond the abdomen, including the thymus, salivary glands, and muscles, among others. Image-guided injections or biopsies can be conducted on any organ or tissue of interest, and shear wave elastography can be applied to numerous superficial structures. Microbubble contrast agents can be employed to evaluate perfusion or the expression of specific biomarkers in these regions.
All of these techniques can be used to monitor the progression of a lesion over time, as well as the regression or other effects resulting from therapeutic interventions. Ultrasound imaging is non-invasive, allowing for the same measurements to be taken repeatedly throughout the duration of a longitudinal study.
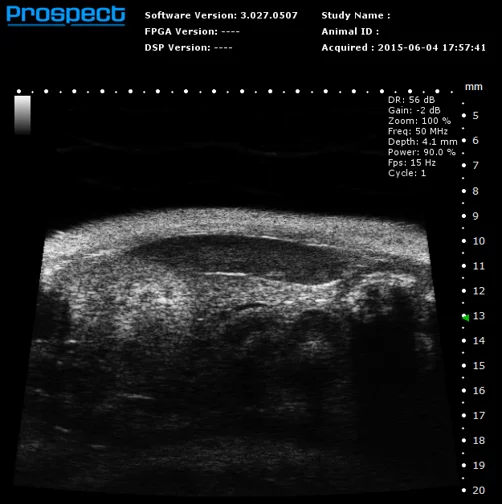
Mouse Abdominal Organs in B-Mode. Image Credit: Scintica Instrumentation Inc
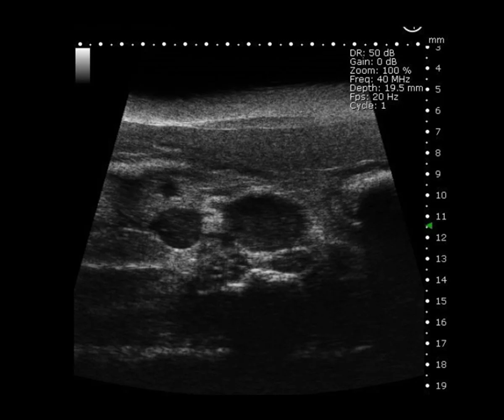
Mouse Supra-Renal Abdominal Aorta Aneurysm in B-Mode. Image Credit: Scintica Instrumentation Inc

Mouse Hind Limb Muscle in 3D B-Mode. Image Credit: Scintica Instrumentation Inc
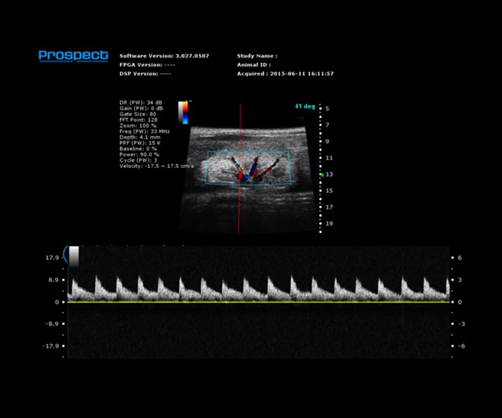
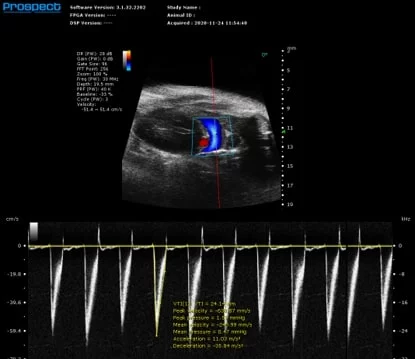
Mouse Renal Vasculature in PW Mode. Image Credit: Scintica Instrumentation Inc
Developmental biology
The Prospect T2 is capable of confirming, counting, and staging pregnancy, evaluating embryonic development, visualizing the initial signs of cardiac contraction, and monitoring embryos for viability or resorption.
All available imaging modalities can be utilized on the developing embryos, including M-Mode, Color Doppler, and PW Doppler to evaluate cardiac function.
Image-guided injections can be conducted on externalized embryos. After the injections are completed, the uterine horns are returned to the dam, and the embryos are delivered normally, with the effects of the injection becoming apparent.
All techniques can be employed to monitor the progression of a lesion over time, or the regression or other effects of therapeutic intervention. Ultrasound imaging is non-invasive, allowing for the same measurements to be taken repeatedly throughout a longitudinal study.
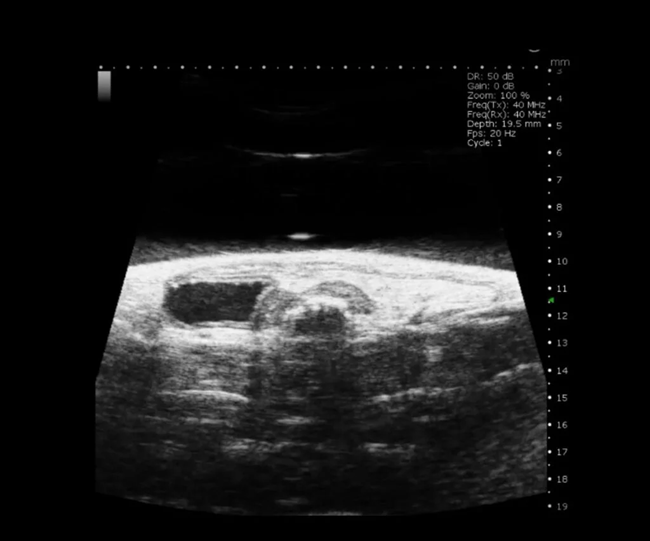
Mouse Uterine Horn in B-Mode. Image Credit: Scintica Instrumentation Inc
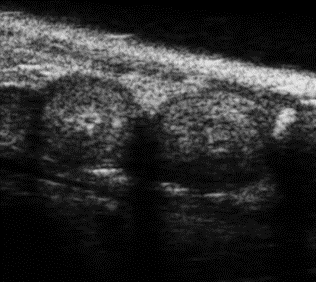
Mouse E7.5 Embryos in B-Mode. Image Credit: Scintica Instrumentation Inc
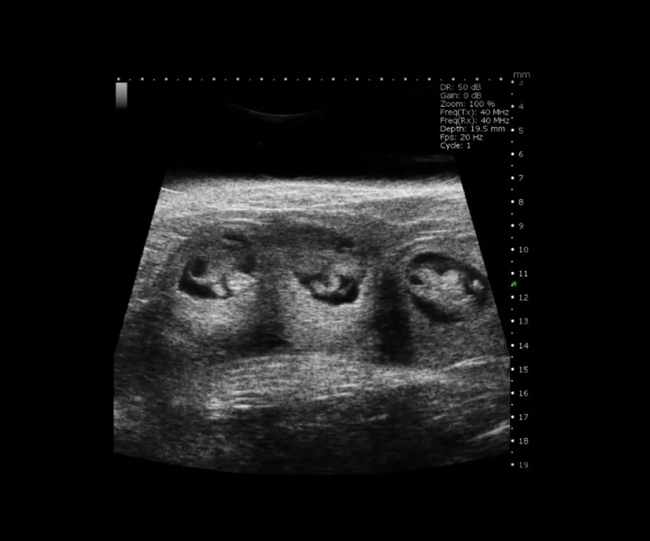
Mouse E10 Embryos in B-Mode. Image Credit: Scintica Instrumentation Inc
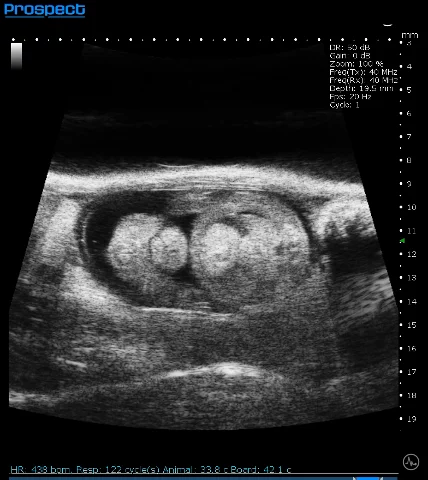
Mouse E15 Embryo in B-Mode. Image Credit: Scintica Instrumentation Inc
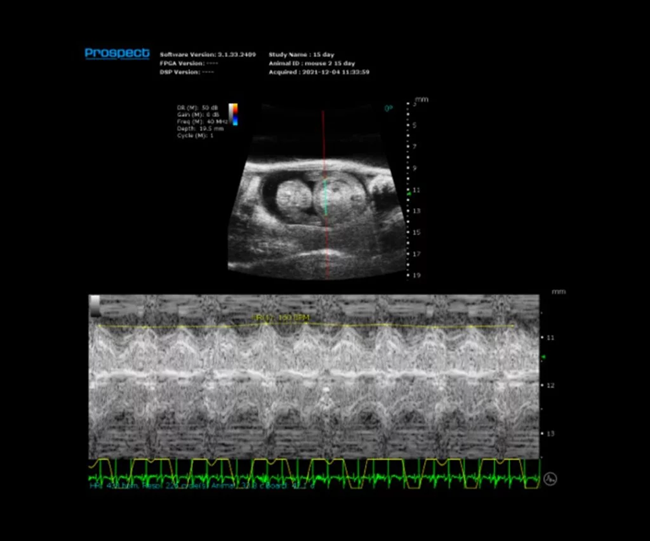
Mouse E15 Embryo in M-Mode with Heart Rate Measured. Image Credit: Scintica Instrumentation Inc
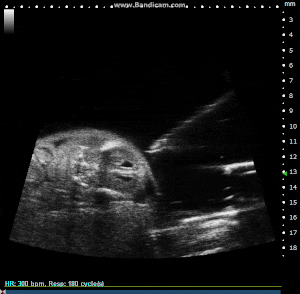
Mouse E15.5 Externalized Embryo with Image Guided Injection. Image Credit: Scintica Instrumentation Inc
Ophthalmology
The Prospect T2 is capable of visualizing both the anterior and posterior structures of the eye across various species, extending beyond just small animals. Three-dimensional imaging can also be conducted on the eye.
Doppler imaging modes are applicable for evaluating flow in larger blood vessels, such as the retinal artery, while microbubble contrast agents facilitate the assessment of perfusion within the eye. In cases where an injection or biopsy is required, the system can be utilized for image-guided injections.
All methods are effective for monitoring the progression of a lesion over time, as well as for observing regression or other effects resulting from therapeutic interventions. Ultrasound imaging is a non-invasive technique, allowing for the same measurements to be taken repeatedly throughout a longitudinal study. Image-guided injections can be administered to externalized embryos.
After the injections are completed, the uterine horns are returned to the dam, and the delivery proceeds normally, with the effects of the injection manifesting subsequently. All methods are effective for monitoring the progression of a lesion over time, as well as for observing regression or other effects resulting from therapeutic interventions.
Ultrasound imaging is a non-invasive technique, allowing for the same measurements to be taken repeatedly throughout a longitudinal study.
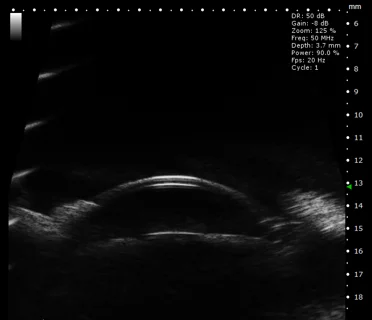
Rabbit Anterior Structure of Eye in B-Mode. Image Credit: Scintica Instrumentation Inc
Mouse Eye in 3D B-Mode. Image Credit: Scintica Instrumentation Inc
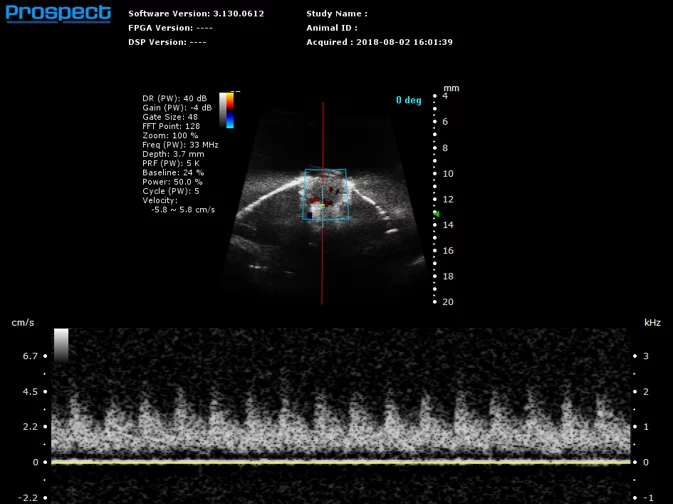
Mouse Retinal Artery in PW Doppler. Image Credit: Scintica Instrumentation Inc
Other species
The Prospect T2 is not restricted to conventional small animal models like mice and rats. It is applicable to a diverse range of other species, including Zebrafish, Chick Embryos, various amphibious species, bats, hamsters, and more. Provided there is a liquid interface between the probe and the tissue, and the sound waves can penetrate, imaging should be feasible.
When suitable, all available imaging modes can be utilized, along with the 3D and image-guided injection options. These techniques can be employed to monitor the progression of a lesion over time, or to assess the regression or other effects of therapeutic interventions. Ultrasound imaging is non-invasive, allowing for the same measurements to be taken repeatedly throughout a longitudinal study.
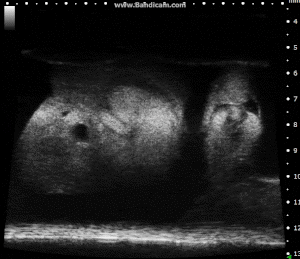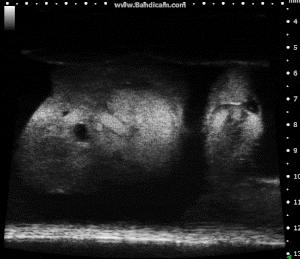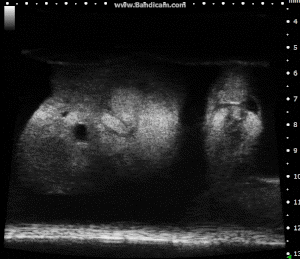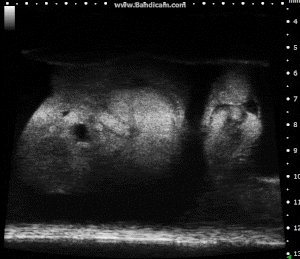
Chick Embryo Day 7 in B-Mode. Image Credit: Scintica Instrumentation Inc
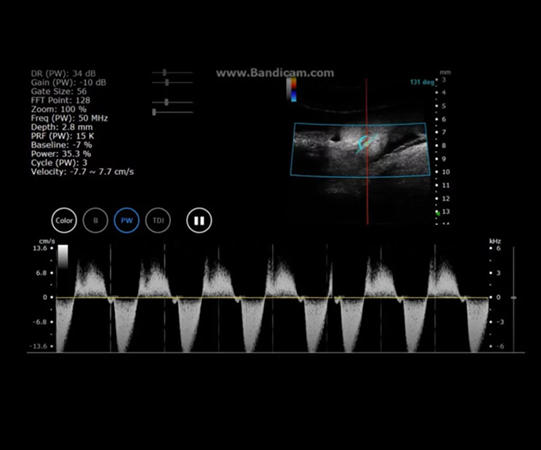
Chick Embryo Day 7 in PW Mode. Image Credit: Scintica Instrumentation Inc
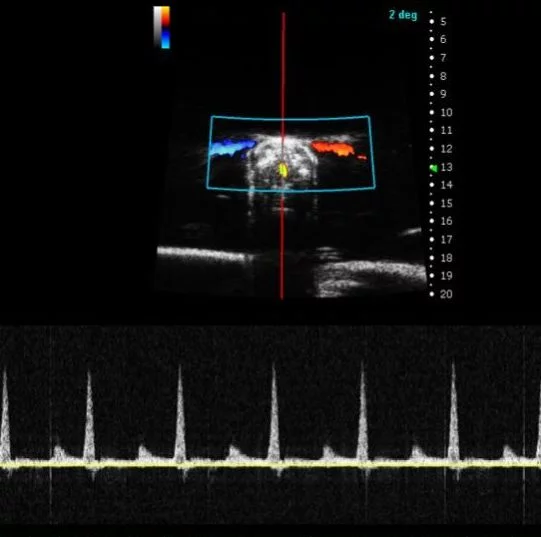
Zebrafish Heart in PW Mode. Image Credit: Scintica Instrumentation Inc
Bladder imaging
Basic bladder imaging
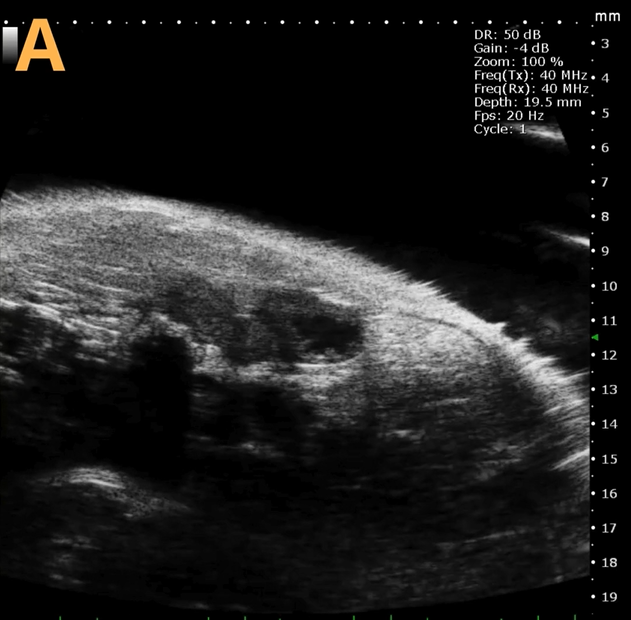
Image Credit: Scintica Instrumentation Inc
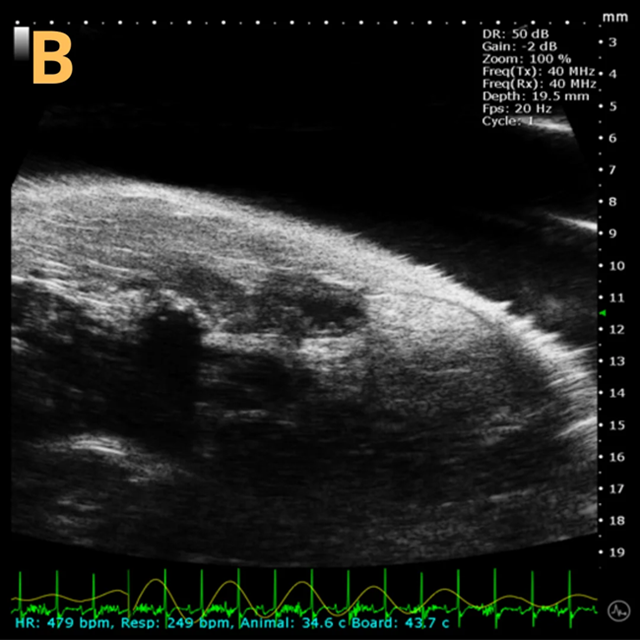
Image Credit: Scintica Instrumentation Inc
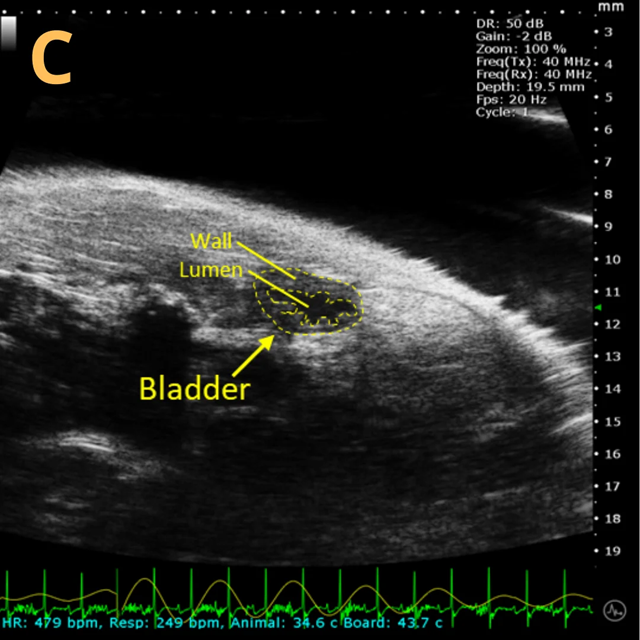
Image Credit: Scintica Instrumentation Inc
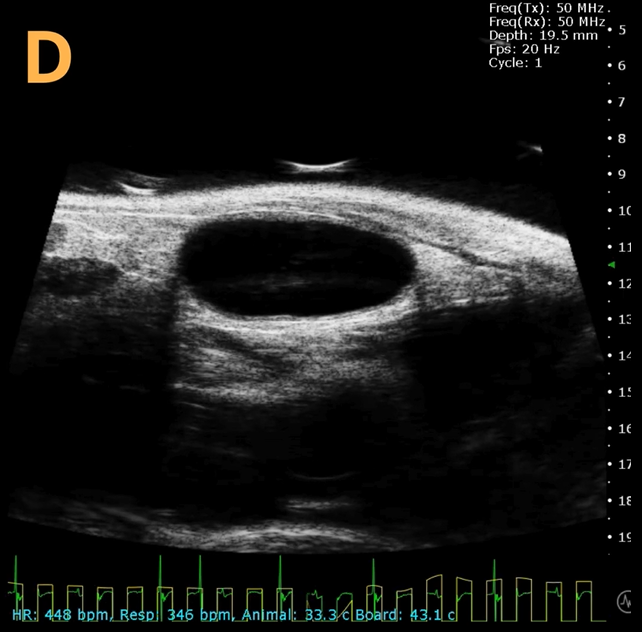
Image Credit: Scintica Instrumentation Inc
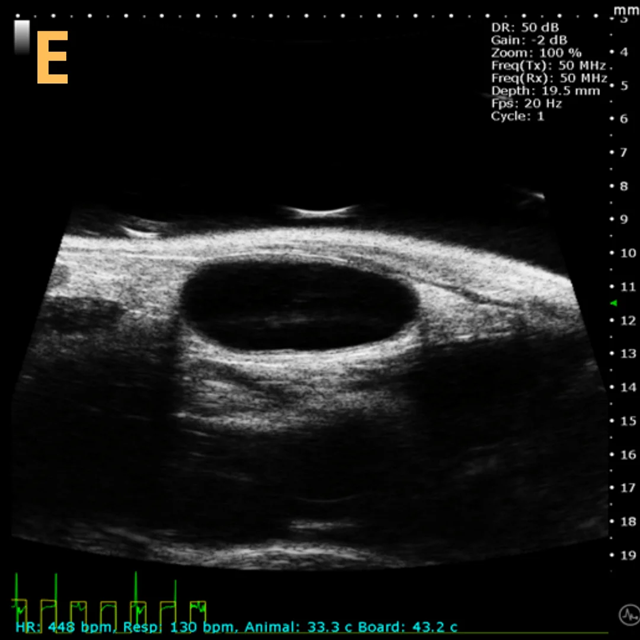
Image Credit: Scintica Instrumentation Inc
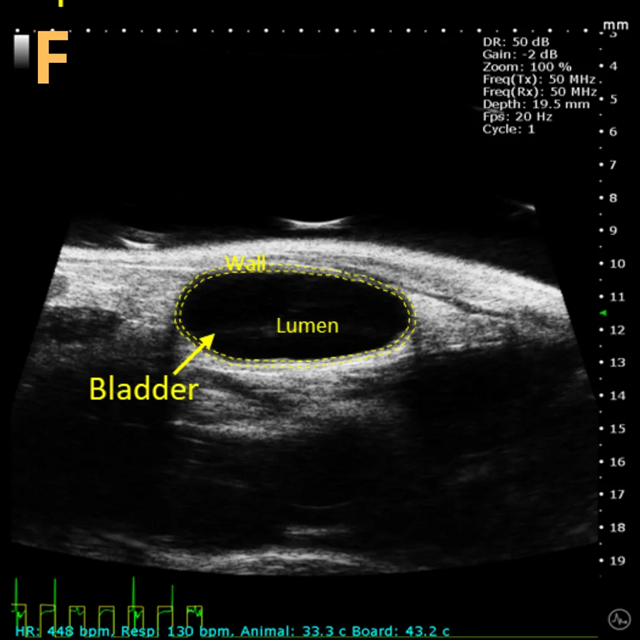
Image Credit: Scintica Instrumentation Inc
These images depict an emptied bladder (A – C) and a full bladder (D – F). Specifically, A) shows a B-mode video capturing an emptied bladder, B) presents a still frame, and C) highlights the noted landmarks. Conversely, D) illustrates a B-mode video scanning a full bladder, E) provides a still frame, and F) marks the noted landmarks.
The imaging was conducted on a wildtype C57B6J mouse model, with the acquired images sourced from Stephen Zderic’s laboratory at the Children’s Hospital of Philadelphia.
Ultrasound-guided injection
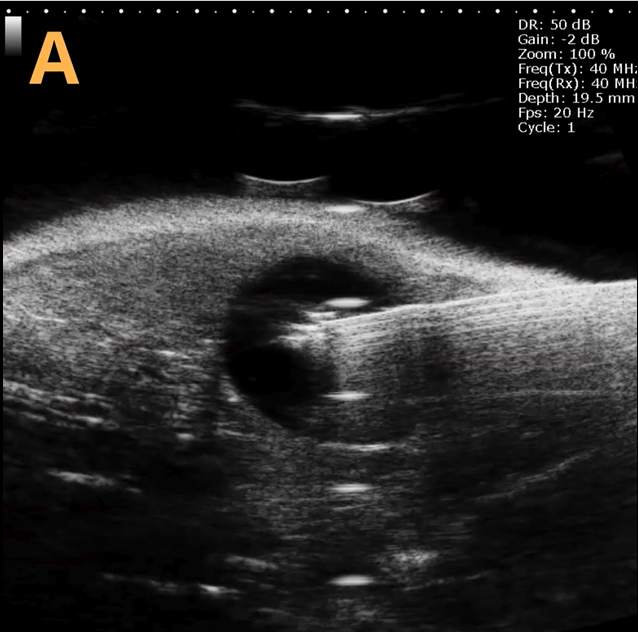
Image Credit: Scintica Instrumentation Inc
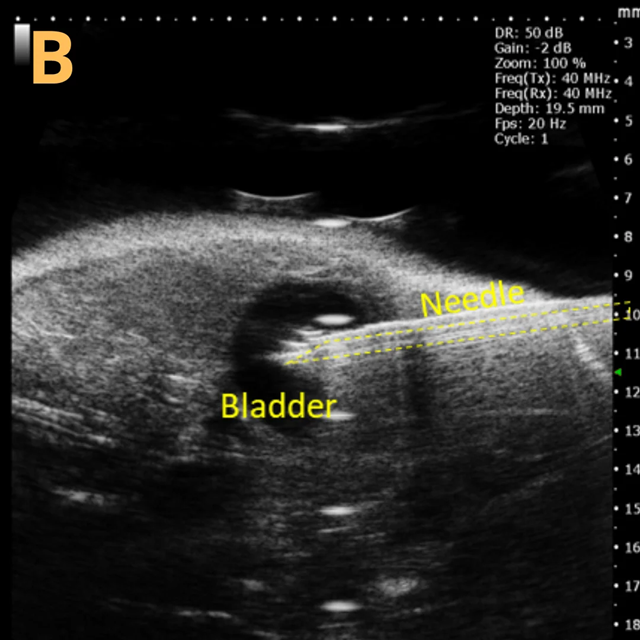
Image Credit: Scintica Instrumentation Inc
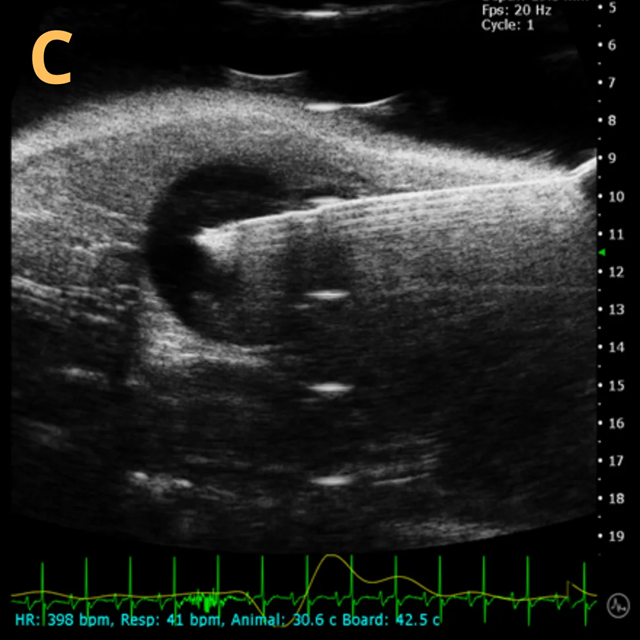
Image Credit: Scintica Instrumentation Inc
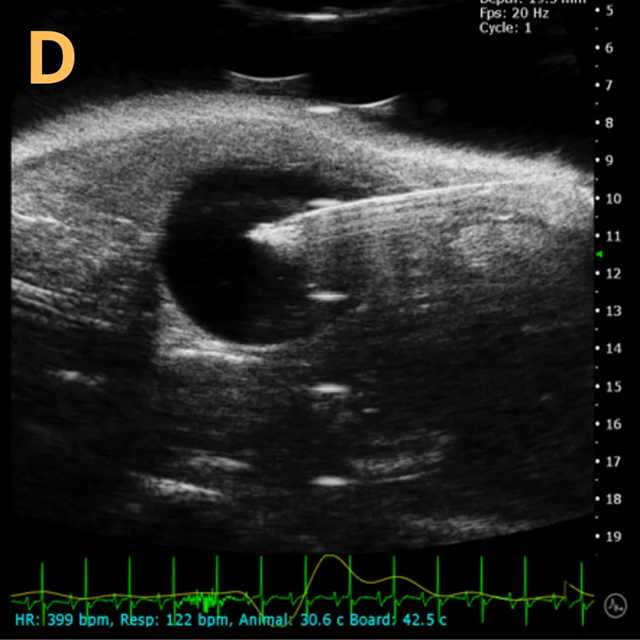
Image Credit: Scintica Instrumentation Inc
This is A) a B-mode video depicting an injection into the bladder, with B – D representing still frames of B) prior to the injection of any solution, highlighting the landmarks, C) during the injection when the bladder noticeably enlarges, and D) the conclusion of the injection just before the needle is withdrawn.
The imaging was conducted on a wildtype C57B6J mouse model, and the acquired images are sourced from Stephen Zderic’s laboratory at the Children’s Hospital of Philadelphia.
Ultrasound-guided bladder catheterization
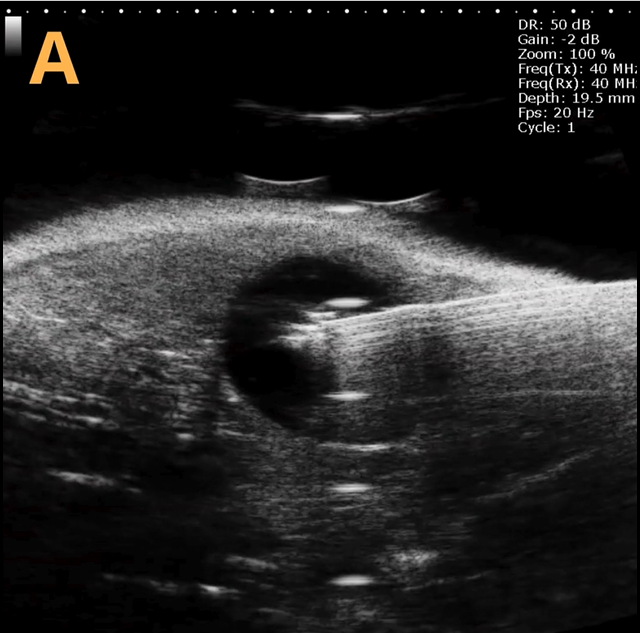
Image Credit: Scintica Instrumentation Inc
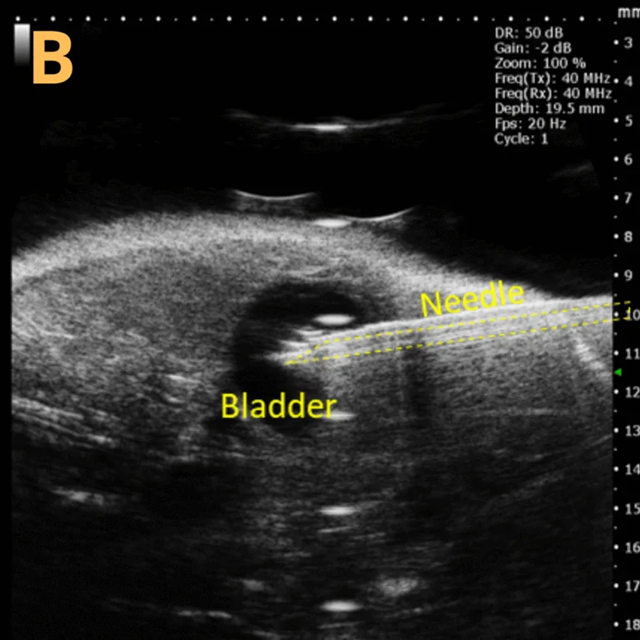
Image Credit: Scintica Instrumentation Inc
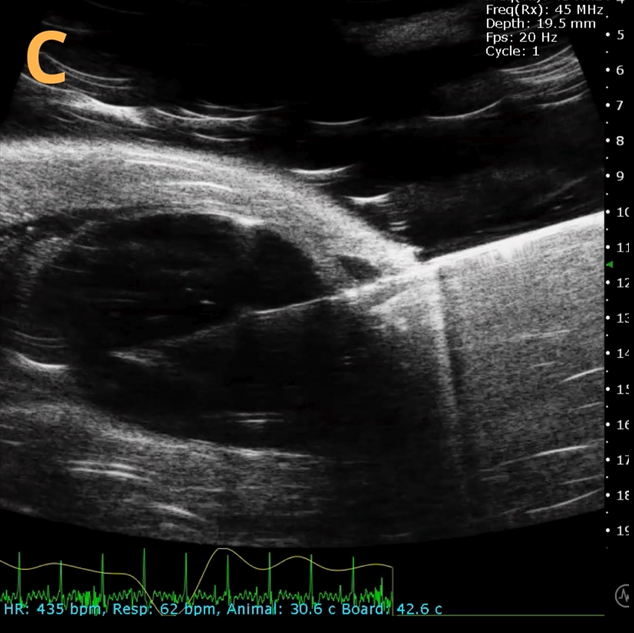
Image Credit: Scintica Instrumentation Inc
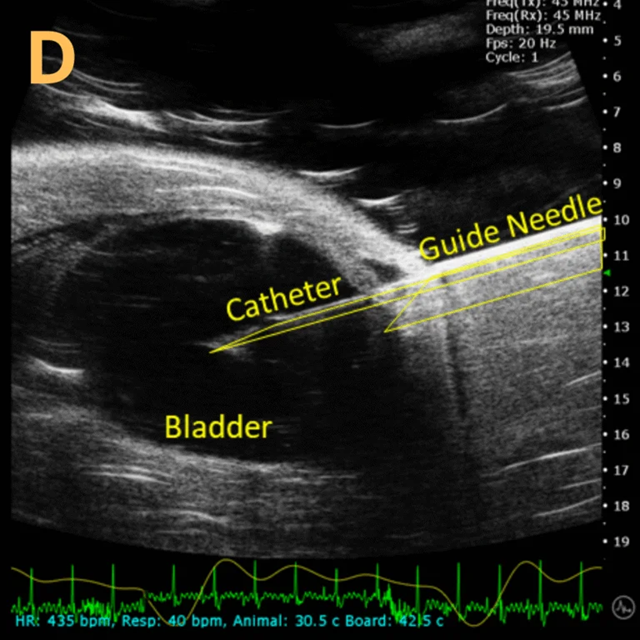
Image Credit: Scintica Instrumentation Inc
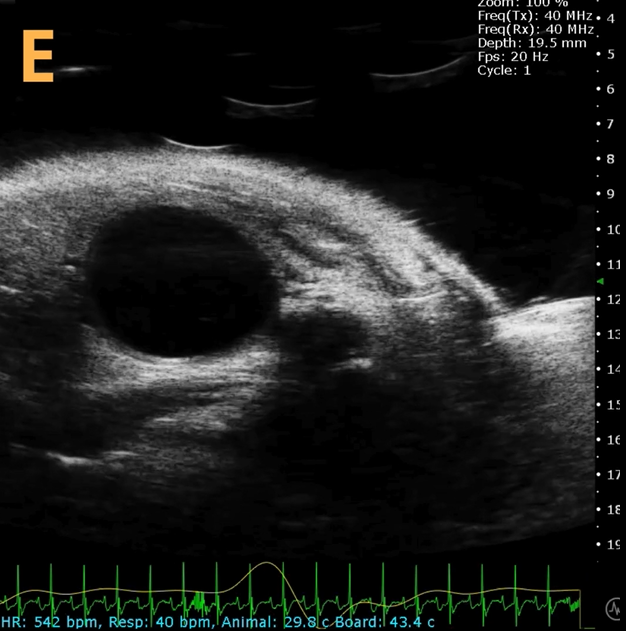
Image Credit: Scintica Instrumentation Inc
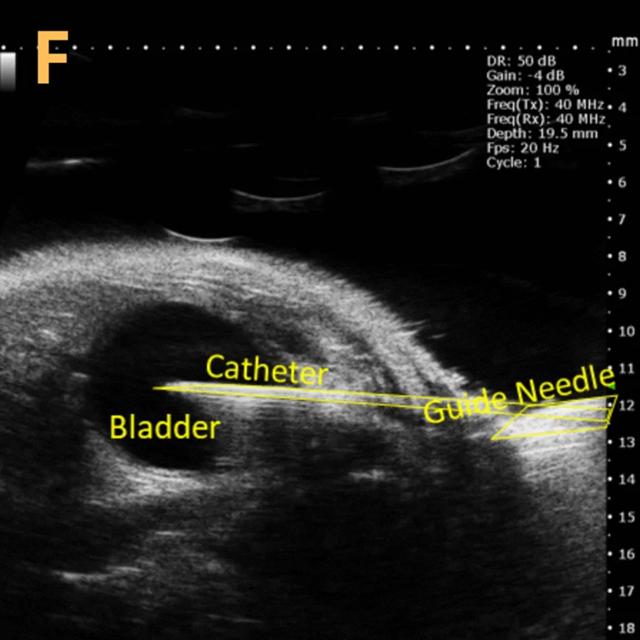
Image Credit: Scintica Instrumentation Inc
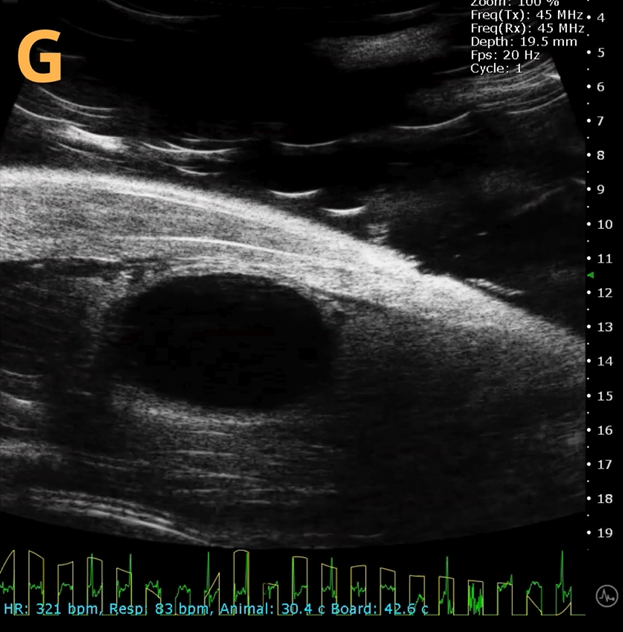
Image Credit: Scintica Instrumentation Inc
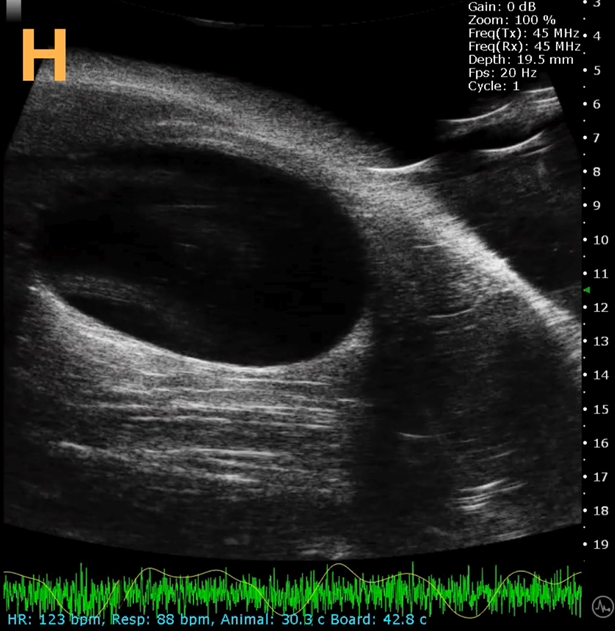
Image Credit: Scintica Instrumentation Inc
This presents A) a B-mode video that illustrates the insertion of a thin-needle catheter through a larger guide needle, with B) the landmarks indicated. C) Another instance of this technique is provided, with D) the landmarks highlighted. E) The removal of the catheter needle is depicted, along with F) the landmarks specified. The verification of successful injection through the catheter is illustrated in G – H, where G) displays the bladder prior to injection and H) shows the bladder post successful injection.
The imaging was conducted on a wildtype C57B6J mouse model, with the acquired images sourced from Stephen Zderic’s laboratory at the Children’s Hospital of Philadelphia.
An in-plane introducer (guide) needle was employed to penetrate the skin of the animal, while a thin needle catheter was advanced through the introducer needle to puncture the bladder wall. The use of the catheter mitigates the risk of leakage or total collapse of the bladder.