In vivo fluorescence microscopy is an imaging technique that uses fluorescence to image cells within live organisms. This type of microscopy provides highly accurate results and allows for the observation of the development of certain processes.
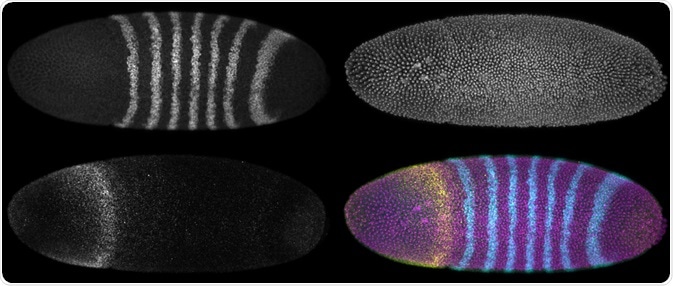 A developing Drosophila melanogaster embryo with three different fluorescence reporters. Maximum intensity projections. (Micha Weber | Shutterstock)
A developing Drosophila melanogaster embryo with three different fluorescence reporters. Maximum intensity projections. (Micha Weber | Shutterstock)
What is in vivo fluorescence microscopy?
Physiology experiments aim to understand a cellular function in the context of a living organism. This was initially achieved through histological examinations of cells in tissues. Real-time analysis was made possible using intravital microscopy, which allows for the study of physiological processes in detail and gives accurate results.
Fluorescence is very efficient and can be applied to many molecules or cells of interest. The cell events that can be studied using in vivo fluorescence microscopy include cell proliferation, migration, differentiation, or intracellular ion actions.
The two mostly used intravital imaging techniques are:
- Confocal fluorescence microscopy
- Two-photon microscopy
Two-photon microscopy provides deeper access to tissues than the confocal technique. It also has less photobleaching and photodamage, as the technique produces no out-of-focus light.
Confocal fluorescence microscopy relies on the ‘double focusing system’ which involves passing a laser beam through a pinhole in front of the light source and another in front of the detector. Both holes focus the light correctly on the sample and the detector, respectively.
When discussing advantages of using in vivo fluorescence microscopy, it has to be emphasized that ex vivo models normally cause artifacts which leads to false results. On the other hand, in vivo models have a reduced occurrence of artifacts, therefore they are better for this reason.
Additionally, in vivo experiments allow for the observation of physiological changes over a certain amount of time. This is useful for studying processes such as development, stem cell physiology, and pathogeneses.
How to prepare tissues for in vivo fluorescence microscopy
Most tissues require preparation to allow optical access. The skin-flap model involves the removal of a patch of skin which allows surgical access to the organs and tissues below. After the experiment/surgery, the patch of skin is sutured back on to its original location.
The skin-flap model has a moderate risk of necrosis of tissue, and allows for mainly the lower dermis and subcutaneous tissue to be reached. Surgical removal of skin can cause damage to the organs and tissues of interest as it is very evasive.
Micro-endoscopic probes can be used to make the optical system smaller, but this method reduces the resolution and the field of view of the acquired images. Additionally, animals moving and breathing causes blurring artifacts during imaging which compromises the results. The tissues of interest can be fixed to reduce this.
Applications of in vivo fluorescence microscopy
Intravital microscopy of the brain cortex is used in neuroscience to study the activity of neural networks. One notable study measured important checkpoints (such as extravasation, arrest found at vascular branching points, and protracted contact to micro blood vessels) towards macro-metastasis within the brain using a single metastatic cell. The effect of spinal cord injuries and the progression of regeneration have also been studied using intravital microscopy.
Furthermore, confocal ophthalmoscopy has been used to study the retina and report on the changes to microstructures over a certain amount of time. The microstructures examined include optic nerves, capillaries, and ganglion cells.
Other uses have been noted as well; for example, an abdominal imaging window was developed to track the growth of a colorectal cancer cell in the mouse liver. The formation of micro-metastasis was observed, and this experiment used 10-hour time lapse intramural imaging which revealed the cellular migration with pre-metastasis.
The same migration was not seen in micro- and macro-metastasis. Pharmacological inhibition of this migration reduced the metastatic burden of the liver cells, making it a potential target for anti-cancer therapies.
Finally, in vivo fluorescence microscopy has been used to study the homeostasis of stem cells in the intestinal stem cell niche. It has been discovered that the central stem cells (located within the crypt base) were biased towards survival and the border stem cells (located in the upper part of the niche) were biased towards loss and replacement.
Further Reading
Last Updated: Nov 21, 2018