In chromosomes, DNA is wrapped around proteins called histones. These histones are crucial for efficient packaging of DNA, as well as the replication and segregation of chromosomes themselves. Due to their affiliation with DNA, histones are important for successful cell replication, which takes place via the cell cycle.
Cell Cycle Timing
The cell cycle consists of four phases (G1, S, G2 and M), all of which are characterized by different factors. The S phase is characterized by DNA synthesis, and in the 1960s it was discovered that histone proteins are also synthesized during this phase.
The amount of histone mRNA increases approximately 25 to 30 times during the S phase. This occurs in part due to increased transcription, which constitutes a 3-10 fold increase in mRNA, as well as due to pre-mRNA processing and mRNA stability. Upregulation of histone gene expression is one of the hallmarks of entry into the cell cycle’s S phase.
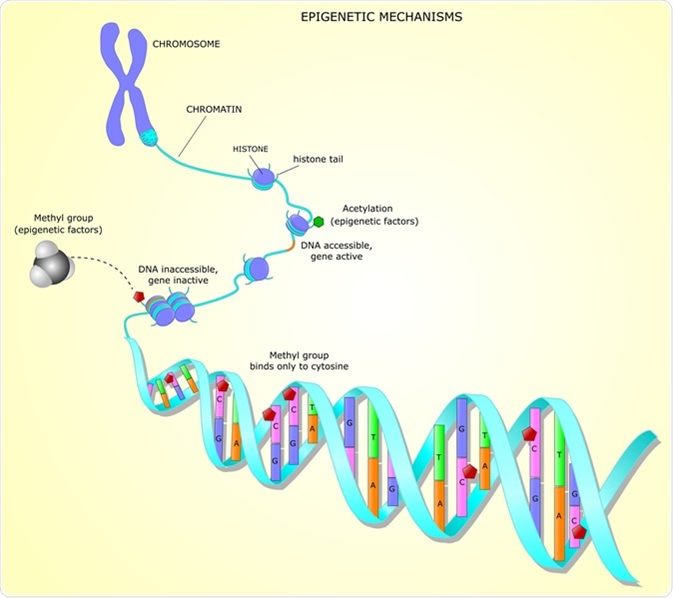
Epigenetic mechanisms. Image Credit: Ellepigrafica / Shutterstock
Regulation of Expression
The main mammalian histone subtypes (H2A, H2B, H3, and H4) and the linker H1 are located in different gene clusters. Of these, H4 and H2B are the most extensively studied. Their promoter activity during the cell cycle is governed by a subtype-specific consensus element (SSCE).
The regulation of transcription of histone genes differs between eukaryotes. In yeast, transcription is regulated by the same elements, whereas in mammals, there are different cis-acting DNA elements and other factors regulating transcription.
Each histone subtype has several copies of the same gene to allow for the large demand placed during DNA replication. Histones can be classified as replication dependent or replication independent, which is decided by their expression pattern during the cell cycle. The replication independent histone genes are transcribed at a relatively constant low rate, regardless of cell cycle stage. However, most of the vertebrate histone genes are replication dependent and are therefore more highly expressed during the cell cycle’s S phase.
How DNA is Packaged (Advanced)
Replication Dependent Histones
Given that the replication dependent histone genes all activate upon entry into the S phase, it is speculated that there exists some further upstream regulatory element. This mechanism is hypothesized to transduce a shared signal that activates the transcription of histone genes when the G1/S phase boundary is reached. This process is assumed to be under the control of the cell cycle, because of how closely linked to the S phase histone synthesis is.
Recent research has made some headway with regards to this question. Entry into the S phase in animal cells is regulated to a large extent by the cyclin E-CDK2 kinase complex. Cyclin E-CDK2 is also needed for the DNA replication and centrosome duplication events in S phase. A cyclin E-CDK2 substrate called NPAT has been found to be associated with histone gene clusters, and cyclin E-CDK2 bolsters activation of histone gene transcription by NPAT. Therefore, NPAT could be the link between cell cycle machinery and shared heightened transcription of histone genes during S phase.
Post Translation
Histones are modified after translation by processes, such as by phosphorylation, methylation, acetylation, and ubiquitination, which directly affect biophysical properties. Several of these processes change during the cell cycle, but unlike transcriptional changes, the post-translational changes occur throughout the cell cycle.
The most typical kind of cell cycle phosphorylation is on H3’s serine. It is seen first in late G2, and increases throughout the M phase. However, this is not the only, or most important, phosphorylation occurrence. Other H3 residues, as well as H2, are phosphorylated as well, often in G2 or M phase.
Methylation of histones does not usually change as much as phosphorylation does. Some methylation alterations of H3 histones occur in the M phase, whereas many other H3s do not change noticeably. However, a type of H4 histone mark, H4K20me1, is cell cycle dependent. It starts increasing in the late S phase, is at its highest point in mid to late M phase, and then decreases.
Acetylation of histones is correlated to areas where transcription is occurring. It generally decreases during M phase, however, some with active promoters can maintain acetylation. Ubiquitination is less studied in terms of its behavior during the cell cycle. Exceptions are H2A and H2B, whose ubiquitination appears to decrease in the M phase.
Further Reading
Last Updated: Oct 28, 2018