Fibromyalgia patients suffer from a variety of symptoms, including widespread body pain and myofascial tenderness. They are ‘hyperalgesic’, which means that if you give them a stimulus that in healthy subjects is just mildly painful (or even completely innocuous), they may perceive it as excruciatingly painful.
Despite the fact that the pain these patients describe feels very ‘real’ to them, if you take a look at the sites where they report pain (e.g., the muscles) often nothing appears to be wrong (although some recent studies do suggest the presence of some alterations in some nerve fibers in the periphery).
This has historically led some to doubt that the pain these people report is real. Fortunately, in the last few years, scientists have started looking at potential changes in the brain, and have indeed produced few interesting observations which –among other things- corroborate the experiences that these patients report.
What were the main aims of this research and what did the study involve?
The main aim of this study was to assess potential dysfunctions in the neural mechanisms underlying anticipation of pain and anticipation of pain relief in fibromyalgia patients.
We focused on this, because expectancy has a role in shaping how we perceive pain as well relief from pain. For instance, if you expect that the pain you are about to receive will be stronger or milder, you will actually perceive more or less pain.
Expectancy also affects how well you respond to therapy: if you don't think a treatment is going to work, it will probably work less than if you had complete faith in it.
So, it is possible the heightened sensitivity and the reduced responsiveness to pain medications that these patients demonstrate may be at least in part explained by alterations in the brain activity associated with expectancy-related brain mechanisms.
In order to address this question, we used a brain imaging technique called ‘functional Magnetic Resonance Imaging’, which allows us to examine how brain activity changes in association with certain states or tasks.
In this study brain activity was investigated when the participants received painful stimuli, as well as when they received visual cues informing the subjects of impending pain onset (pain anticipation) and pain offset (relief anticipation).
In terms of pain stimulation, we use a vascular cuff, similar to the one used to measure blood pressure, placed on the leg. Of note, this type of stimulation is different from what is typically used in brain imaging studies (e.g., contact heat probes placed on the skin), because the ‘pain receptors’ it targets are located more in the deep tissue, rather than on the skin, and thus we think it may generate a pain sensation that is closer to the clinical pain of fibromyalgia.
The intensity of the cuff pressure was individually tailored, in order to generate a pain rating of 40-50 on a 0-100 intensity scale.
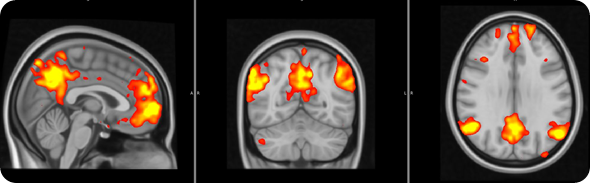
You showed the participants visual cues to forewarn them of forthcoming pain onset or offset. How would you expect the brain to respond to these cues in people without fibromyalgia?
We saw that the healthy subjects showed large brain responses, in many regions, when they were anticipating pain or relief. Some of these changes are likely to be due to the detection of ‘salient’ event. In other words, some of these responses may be non pain-specific and may simply reflect the fact that the subjects were detecting an ‘interesting event’.
However, other responses would seem to be even more interesting, and possibly pain specific: for instance the healthy volunteers activated a region called the ‘periaqueductal gray region’, which we know exerts a modulatory effect on the pain signals coming to the periphery (this is a region that if stimulated electrically can alleviate pain).
The healthy volunteers also showed that a region involved in the processing of ‘reward’/’punishment’, the so called ventral tegmental area (VTA), was activated during pain anticipation, and deactivated during relief anticipation.
How did fibromyalgia patients in your study respond to pain anticipation and relief and how did they compare to control participants?
FM patients exhibited reduced brain responses in response to visual cues for both pain anticipation as well as relief anticipation.
This reduced responsiveness was very widespread, and included the periaqueductal gray and the VTA.
Did the results of your study match your expectations?
There were certainly some surprises. While I did expect to observe reduced responses to the anticipatory cues in some regions (e.g., the regions involved in the descending pain modulatory system, which is known to be altered in these patients), I did not expect to see so many regions exhibit this behavior.
Now the question to investigate is: what causes these alterations? We are now trying to investigate whether psychological factors can explain these results.
How do you think these results can be explained?
In healthy volunteers, the activation of regions that process signals of punishment during pain anticipation, and the deactivation during relief anticipation simply tells us that the brain of healthy volunteers registered the incoming pain stimulation as a punishing event, and the interruption of it as a rewarding event, as should be expected.
Detecting pain as a punishing event may be what cause the healthy subjects to activate strongly the PAG (as previously mentioned, one of the brain regions that modulates pain signal coming from the periphery). This may reflect the fact that healthy volunteers are preparing and ‘bracing’ for the incoming pain stimulation, by accessing the body own analgesic mechanisms.
In FM patients, the reduced activation of the PAG in preparation of pain, may reflect that the patients are less capable to engage the body own endogenous analgesic mechanisms, as if they had more difficulty getting ready to cope with the incoming painful stimulation.
The reduced responsiveness to all cues shown by the VTA (less activation in response to anticipation and experience of pain, and less deactivation in response to anticipation of relief), may suggest that the FM brain does not register the incoming pain stimulation as a punishing event, and the interruption of it as a rewarding event.
We haven’t directly tested this hypothesis, but perhaps this dampened response to warning signals might explain (at least in part) why the FM patients do not engage the descending pain modulatory systems as well as healthy subjects, when they know they are about to receive pain.
Moreover, one possible explanation for the brain activity differences observed in other brain regions across groups during the pain anticipation/relief periods involves the concept of salience, i.e., the ability of a given stimulus to ‘stand out’ from its background.
As most patients reported experiencing some amount of ongoing pain (i.e., their clinical pain) even in the absence of cuff stimulation, the cues may have only signalled the transition from a lower level of pain to a higher level of pain (or vice versa), rather than the transition from a pain-free state to a moderately strong pain state (or vice versa), as was the case for the healthy controls.
It is therefore possible that the observed group differences might partly reflect a lower salience attributed by the patients to the impending onset or offset of cuff pain stimulation.
Do these results help us to understand why pain medications such as opioids are often not very effective in fibromyalgia patients?
As previously mentioned, expectancy has a role in shaping how we perceive pain as well relief from pain. Thus, the alterations in neural responses to the anticipatory cue we observed in FM patients might contribute to explain why these patients are more pain sensitive, and respond less to some pain medications, than healthy subjects.
Interestingly, in a recent study, lower responsiveness of VTA and other ‘reward regions’ to noxious stimuli was found to predict lower opioid-induced analgesia in healthy subjects. Thus, reduced VTA response to pain (as well as pain anticipation/relief) in FM matches well the lack of therapeutic efficacy of opioids in treating FM pain.
What excites you most about this research and how should future studies build upon these results?
One potential implication of these observations is that cognitive strategies aimed at changing the patient's expectations (for instance, increasing expectancy of therapy outcome) may be beneficial.
Where can readers find more information?
There are several resources on the web which provide further information on fibromyalgia. For instance, the Nation Fibromyalgia Association (http://fmaware.org)
About Dr. Marco Loggia

Dr. Marco Loggia is Instructor at Harvard Medical School and works at the A. Martinos Center for Biomedical Imaging at Massachusetts General Hospital, in Boston, MA.
He completed his graduate studies at McGill University in Montreal, under the supervision of Dr. Catherine Bushnell, a renowned pain scientist and pioneer of noninvasive imaging of pain in humans (and now Scientific Director at NIH-NCCAM).
After his graduation in 2008, Dr. Loggia was offered a joint Research Fellowship position at Massachusetts General Hospital and Brigham and Women’s Hospital, Harvard Medical School.
His scientific interests focus on the brain mechanisms of pain processing, in both health and disease.
He is particularly interested in the study of brain alterations associated with chronic pain conditions, which he investigates using a variety of neuroimaging techniques, including functional Magnetic Resonance Imaging, Arterial Spin Labeling and Positron Emission Tomography.