What are the main biological questions that you have been able to answer through the use of flow cytometry?
Flow Cytometry, the measurement of various cellular characteristics as they flow through a measuring apparatus, has so many applications that it's hard to know where to begin.
I have been involved with the application of flow cytometric techniques to cervical cancer pre-screening, the purification of individual mouse chromosomes, identification of cells undergoing apoptosis, stem cell purification, the study of the T-cell receptor function and T -cell biology, HIV/AIDS, and various applications related to cancer biology.
Peter Lopez - Centrifugal Elutriation - Utility in the Flow Cytometry Laboratory
How has the use of flow cytometry changed since you began working in the field in 1977?
Flow cytometers in the 70's were relatively rare, and required a dedicated specialist to operate, maintain, and interpret the collected data.
The number of measurements that could be made from each cell was limited to less than a handful. Instruments also analysed cells at what we now consider to be slow throughput rates (<5,000 cells per second).
Flow cytometers were typically set up in shared resource facilities (cores), so that many laboratories could benefit from the instrumentation and the expertise. These instruments were also quite large and expensive, so the use of flow cytometry was limited to a few academic centers.
Today we see flow cytometers that have vastly improved throughput (analysis of 70,000 cells/second), with the capability of collecting more than 20 parameters.
The marriage of flow cytometry and mass spectrometry is an exciting new development allowing even more parameters to be analysed than traditional flow cytometry.
The replacement of the ubiquitous fluorescent labeling technique used in flow cytometry with metal isotypes to label cells promises the ability to detect cell subsets resolved with 30 or more parameters, although this CyToF technology precludes cell sorting.

Image credit: Beckman Coulter
Another development is the introduction of less complex and easy to use cytometers, with use now seen outside of the academic core setting.
Flow cytometers are now routinely seen in the pharmaceutical industry, in agriculture and plant science, in manufacturing, in food and water quality monitoring, in animal sex determination, in monitoring the marine environment, in microbiology, with a large important application in human diagnostics.
How has Beckman Coulter supported your work?
Beckman Coulter flow cytometric instrumentation has been in my laboratory since the mid 80's. More recently BC has been very supportive of our studies demonstrating the benefits of the integration of centrifugal elutriation into the flow cytometry laboratory.
MoFlo CDP Cell Sorter at NYULMC Flow Cytometry Facility. High-Speed cell sorter for bulk collection or single cell cloning of purified cell populations. Image credit: Beckman Coulter
What is centrifugal elutriation and what are the benefits compared to other cell purification techniques?
Centrifugal elutriation is a technology invented by Charles Lindberg (the famous pilot) in the 1930s, and allows separation of cells according to their sedimentation velocity in a continuous fashion using a specialized centrifuge rotor.
This rotor allows loading of cells into the spinning elutriator chamber, and then permits the introduction of a buffer opposing the direction of centrifugal force, thereby fractionating cells.
This has the important benefit of producing purified populations of cells without the need for chemically treating or labelling cells, and can produce large numbers of purified cells faster than flow cytometry. Centrifugal elutriation is also a gentle procedure.
Can elutriator systems be kept sterile? Why is this important for answering certain biological questions?
Yes, the entire elutriation system can be sterilized in situ. Cell samples can be prepared, introduced and collected from the elutriator in a sterile biosafety cabinet, maintaining cell sterility. This is important since the purification of cells is often the first step in a series of biological assays which involve cell culture.
A well-established use of elutriation is to synchronize cells into the same cell cycle phase, and these cells are then put into cell culture or other systems where drug effects may be tested, and sterility is crucial in these systems.
Where samples are being preprocessed for the elimination of small debris prior to sorting, all procedures must maintain cell sterility, including elutriation and cell sorting.
Which Beckman Coulter instruments do you use in your laboratory and why?
All of the Beckman Coulter systems in use in my lab are solid workhorse systems that we count on for daily consistent output.
We of course use Beckman Coulter centrifuges, both the Allegra X-12R benchtop unit and the Avanti J-20 floor-standing unit that drives our JE-5.0 counterflow elutriation system. All of these centrifuges are well-built, stable instruments.
For cell viability and cell size determination, we have a Beckman Coulter ViCell. Viability and cell size are crucial determinations prior to elutriation and also helpful in flow cytometry.
For cell sorting one of our workhorse systems in the Beckman Coulter MoFlo XDP high-speed cell sorter. We are in the process of placing the Beckman Coulter CytoFLEX 13-color flow cytometer into our lab's workflow.
How does flow cytometry cell sorting compare to magnetic bead separation and centrifugal elutriation?
Each technique has its strengths and weaknesses. Cell purification by flow cytometry is unmatched in its ability to purify many populations simultaneously (up to 6 populations using the Beckman Coulter MoFlo Astrios), with the ability to characterise these subpopulations for purification using 20 parameters or more.
Since each individual cell needs to be characterized for analysis and sorting, flow cytometry currently has a “speed limit”, so as a result the number of cells that can be purified is not on the same level as elutriation or magnetic bead desperation. The high purity level of sorted cells using flow cytometry-- up to 99% purity-- is also unmatched by other techniques.
Magnetic bead separation is an easy-to-use technique, and in many cases it is used prior to flow cytometric cell sorting to pre-enrich samples for a desired cell type. When used to preprocess samples before cell sorting, there is a time saving, especially when processing large numbers of cells.
The strengths of both technologies can be merged. It can also be used as a standalone purification technique, but can only separate cells based on one characteristic -– it is not a multiparametric system.
Centrifugal elutriation shares some of the same benefits and weaknesses of magnetic bead separation, but since the sedimentation velocity, which is dominated by cell size, is the discriminating factor in the purification, it can provide enrichment of cells in various stages of the cell cycle, which is not easily accomplished using magnetic separation. This type of application can be performed using flow cytometric cell sorting, although at a slower throughput.
A very significant advantage of centrifugal elutriation when compared to both flow cytometric or magnetic purification methods is that cells for centrifugal elutriation require no chemical treatment or labelling step. This is a huge advantage for synchronizing cell populations where serum starvation or chemical cell-cycle blocking is not desirable.
What limitations still need to be overcome?
Centrifugal elutriation is a technique where experience plays an important role, which makes it similar to flow cytometry in that regard. As seen in the current trends for flow cytometer manufacturers, producing systems that can be used by less experienced individuals would be beneficial.
Another limitation is the ability to process small cell numbers of less than a million cells. Having specialized smaller elutriation systems would help resolve this limitation.
What do you think the future holds for the use of centrifugal elutriation and flow cytometry in the context of answering biological questions?
Much of the published work with centrifugal elutriation shows that it has utilization in syncronization of cells as well as purification of certain cell populations from a heterogenous starting population.
We have demonstrated the ability of elutriation to reduce the quantity of small debris present in many cell preparations destined for cell sorting. This will add a new application to the elutriation repertoire, to be used as a preprocessing step before cell sorting.
Preparations made from primary material like dissociated tumors, used in the study of tumor infiltrating lymphocytes can contain large amounts of small debris that can make it hard to identify cells of interest and lengthen time needed to cell sort. Coupling a preprocessing cleanup of debris-laden samples san improve downstream cell sorter efficiency.
Removal of small debris or other unwanted material has the potential of providing CyTOF instruments with a cleaner sample and improving efficiency in that workflow also. We will be investigating this in future collaborative studies.
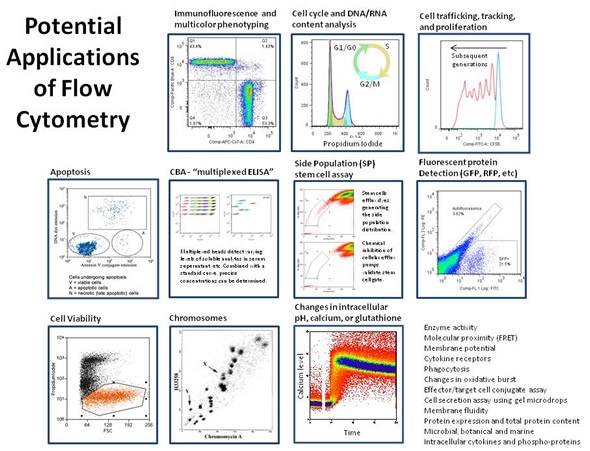
Image credit: Beckman Coulter
Where can readers find more information?
The best single source of information related to flow cytometry is “Practical Flow Cytometry”, by Dr. Howard Shapiro, now in its 4th edition. Beckman Coulter provides a link to this reference for online viewing on their website. http://www.coulterflow.com/bciflow/practical/book/index.html
About Peter Lopez
 Peter Lopez has been involved in the field of flow cytometry since 1977 and has experience in shared resource lab management, cytometer operation, instrument development, experimental design, and a wide variety of cytometric applications.
Peter Lopez has been involved in the field of flow cytometry since 1977 and has experience in shared resource lab management, cytometer operation, instrument development, experimental design, and a wide variety of cytometric applications.
Peter has managed highly-successful and respected flow cytometric and imaging core facilities at Fox Chase Cancer Institute, Dana-Farber Cancer Institute, The Aaron Diamond AIDS Research Center, and currently at the NYU Langone Medical Center.
Peter has hands-on experience with the operation and troubleshooting of instrumentation from all major cytometer manufacturers, as well as experimental prototype instrumentation.
Peter is experienced with laboratory configuration, validation and best practices for sorting infectious material. He is involved in educational program development covering cytometric theory and application.
Peter has been the president of regional professional societies such at MetroFlow ( New York/New Jersey Cytometry User group), The Northeast Regional Life Sciences Core Directors (NERLSCD) ,and is currently serving terms as an ISAC Councilor as well as being an ABRF Executive Board member. Peter serves on numerous academic and industrial advisory boards.
Peter’s current interests include redefining the role of the flow cytometry core in a distributed location model, adding new technologies such as centrifugal elutriation into the flow cytometry core service catalog, as well as methods to verify data reproducibility and strengthening the position of core laboratories as the center for best practice adherence in the academic center.
 About Beckman Coulter
About Beckman Coulter
For Beckman Coulter’s worldwide office locations and phone numbers, please visit "Contact Us" at www.beckmancoulter.com.
Introduced in the mid-1950s, the Coulter Principle became the foundation of an industry responding to the need for automated cell-counting instruments. The industry developed in three acts, as Wallace H. Coulter and his brother Joseph R. Coulter, Jr., developed the simple idea of passing cells through a sensing aperture.
In Act I, Wallace’s desire to automate the routine erythrocyte count led to a simple idea, the definition of the Coulter Principle, its patenting, its acceptance by the National Institutes of Health, and its description at a national conference.
In Act II, the Coulter brothers addressed the practicalities of a commercial instrument and of a business organization to support its manufacture and sale.
In Act III, a broad research effort developed regarding volumetric errors originating in functional characteristics of the sensing aperture, and the brothers’ growing organization found solutions permitting introduction of increasingly automated hematology analyzers. Today the industry thrives, with several participants.
Beckman Coulter offers a range of particle characterization tools including:
- Laser diffraction particle size analyzers
- Multisizer™ COULTER COUNTER®
- Zeta potential and submicron particle size analyzer
- Surface area and pore size analyzer
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.