In this interview, News Medical speaks Tobias Pusterla, Ph.D from BMG Labtech about versatile automated microplate sample screening applications.
Can you give me a brief overview of the range of applications that an automated microplate sample reader could be used for? Why is it such a popular method of screening samples?
Microplate readers are divided into single-mode and multi-mode readers. A single mode reader is mainly a reader dedicated to the detection of absorbance, luminescence or fluorescence. A multi-mode microplate reader combines the aforementioned detection modes and adds as well fluorescence polarization, Time-Resolved Fluorescence (TRF) and Alpha technology.

Given the growing analytic needs of modern laboratories multi-mode readers have become quite popular. The range of applications of a multi-mode reader covers basically all non-isotopic-based detection modes.
We are talking about basic assays that are used in academic laboratories such as DNA/RNA and protein quantification via absorbance, gene reporter assays via fluorescence intensity or luminescence, to drug hits and compound screening in pharmaceutical companies.
An application that has become quite popular in recent years is detection in living cells. This has been fostered by technological improvements that nowadays allow controlling the atmosphere (CO2 and O2) and temperature in the reader as well as the ability of the detection optic head to focus exactly on the cell layer. The success of microplate readers depends on the fact that they are very flexible, allowing the measurement of several assays, they save time and produce objective data.
What screening methods are possible with a microplate reader?
In modern biological sciences, signal detection is based on light output. Reagent manufacturers continuously produce and optimize assays that allow, mainly through the quantification of a specific emission wavelength, the quantification of a sample or an interaction.
Therefore, any screening method that relies on a chemistry that is able to produce or modify light wavelengths can be detected with a microplate reader and can be used for screening purposes. To give you a few examples, the competition between a ligand and a drug compound to bind a receptor, the activation via extracellular signals of a specific signaling pathway, the ability of a compound to induce the death of cancer cells as well as the study of protein aggregation in neurodegenerative diseases, all these assays can be detected by a microplate reader.
Besides for biological and pharmaceutical screenings, microplate readers can also be used in the food industry for quality control purposes or for the detection of spirit adulteration.
What applications are there in the field of DNA and RNA quantification? Which screening methods can be used and how does this affect what can be found?
The quantification of nucleic acids (DNA or RNA) in samples is part of the daily routine in many laboratories. Basically, there are two ways to detect nucleic acids, with absorbance or fluorescence detection. Some researchers prefer the UV absorbance method and measure their samples at 260 nm as it is simple and fast.
In fact, the user doesn’t need to prepare a standard curve to relate the absorbance value to a certain DNA or RNA concentration nor to stain the samples. The nucleic acids' extinction coefficients are known and integrated into the microplate reader software so that the DNA concentration value will be immediately shown.
Moreover, the advantage to detect DNA or RNA with a spectrometer such those used on BMG LABTECH readers, is that besides 260 nm, other reference wavelengths can be measured at the same time. As well as its quantification, DNA/RNA purity is a big issue as well.
Therefore, as well as at 260 nm, researchers usually measure at 230 nm for contamination by organic compounds, at 280 nm for contamination by protein and at 340 nm for particulates contaminating the solution, causing scattering of light in the visible range.
These four wavelengths can be detected at the same time with only one measurement with a spectrometer. Filter or monochromator-based readers will have to switch filters or move mechanical components.
Today, the fluorescent quantification of DNA and RNA has become quite popular as well. With this method, an increased fluorescence signal is measured once the fluorescent dye has bound to nucleic acids. The detection of this method can be achieved with a fluorescence microplate reader and needs the purchase of a specific fluorescent dye and the measurement of a standard curve that can associate known concentrations to fluorescence values.
However, these higher efforts are rewarded with higher assay sensitivity than in absorbance detection. Moreover, besides sensitivity, fluorescent detection is also able to differentiate between double-stranded and single-stranded DNA, as well as RNA.
Can you explain NanoBRET assay technology? What can this technique tell us about cell signaling?
NanoBRET™ is a BRET (bioluminescence resonance energy transfer) based technology that uses the NanoLuc® luciferase as an energy donor. NanoLuc was developed by Promega as a genetically engineered luciferase derived from a natural luciferase isolated from deep sea shrimp.
The advantages of NanoLuc are the higher light intensity compared to classical luciferases, its relatively small molecular weight (19kDa) and its stability (up to 55°C). Unlike other luciferase-GPCR fusion constructs, it is readily expressed on the N-terminus of GPCRs and efficiently transported to the cell membrane. These properties make the NanoLuc® the perfect BRET donor.
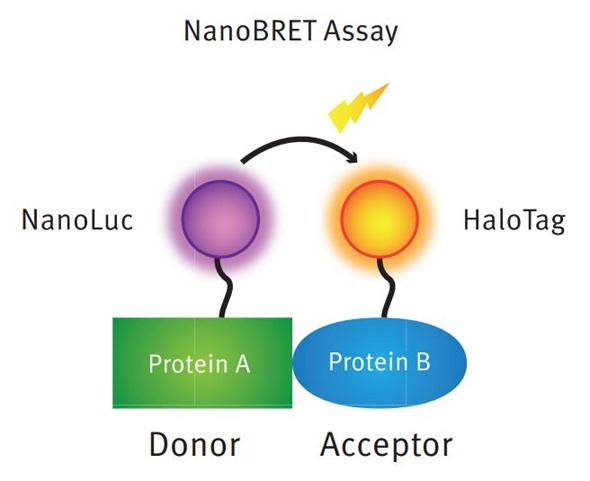
Principle of the NanoBRET assay for detecting intracellular protein:protein interactions. The NanoBRET assay is a proximity based assay measuring energy transfer between a NanoLuc donor fusion protein and a fluorescently labeled HaloTag acceptor fusion protein.
On this topic, Associate Professor Kevin Pfleger of the University of Western Australia/Harry Perkins Institute of Medical Research and Professor Stephen Hill of The University of Nottingham (UK), who are leaders in BRET, collaborated with Promega and BMG LABTECH on Australian Research Council Linkage Grant.
The grant was targeted towards developing new technological approaches enabling academics, biotech and pharmaceutical companies to discover novel treatments for a range of disorders. A major outcome of this collaboration is the recent publication in Nature Methods of the paper entitled "Application of BRET to monitor ligand binding to GPCRs" (Stoddart et al., 2015).
The paper demonstrates for the first time the principle of real time ligand binding to G Protein-Coupled Receptors (GPCRs), the single most important family of drug targets, using Bioluminescence Resonance Energy Transfer (BRET) in living cells.
In fact, NanoBRET has been shown to be an alternative to radio ligand binding experiments and extends researchers’ capability to allow assessment of real time binding kinetics in live cells. This adds to the established capability of BRET to monitor protein-protein interactions in real time in live cells, further enhancing biologists’ and pharmacologists’ insights into real time cellular processes. The NanoBRET data on this paper were generated either with the CLARIOstar or with the PHERAstar.
Microplate readers can be used with living cells to monitor cellular redox changes, how is this done and what does this tell us about the cells being studied?
The generation of reactive oxygen species is part of the normal cellular metabolism. However, reactive oxygen species are also generated from exposure to cigarette smoke, pollutants, chemicals, and other environmental toxins. Accumulation of abnormal levels of ROS in the body has been implicated in several diseases including diabetes, renal ischemia, atherosclerosis, cancer, and ageing in general.
In order to detect cellular redox changes, you firstly have to have a plate reader equipped with specific features. These are a gas control module such as our ACU (Atmospheric Control Unit) that allows the active regulation of CO2 and O2 within the reader and a temperature incubation system to keep the cells “happy” and avoid any stress factor that may bias the experiment. Moreover, it is of advantage to have the possibility to detect the microplate wells from the bottom and to automatically focus on the cell layer.
Scientists developed genetically encoded biosensors based on the redox-sensitive green fluorescent protein. These sensors are a particularly powerful tool to monitor cellular redox changes. For instance, the protein roGFP2 exhibits different redox states that directly affect its excitation spectrum. This means that the oxidized or reduced roGFP2 proteins will have different excitation peaks.
A microplate reader can therefore excite at these two different wavelengths and detect the same emission. With this double detection, scientists are then able to determine how much roGFP2 gets oxidized by living cells which have been treated with specific drugs.
Fluorescence microplate readers represent an ideal system for roGFP-based high throughput screening, for example to identify chemical compounds that modulate redox homeostasis.
%20are%20not%20evenly%20distributed%20in%20the%20well.jpg)
Well Scanning of GFP-expressing protoplasts. The well scan shows that the protoplasts (green color) are not evenly distributed in the well.
Redox processes play an important role in cellular physiology and pathology. Monitoring redox changes in cells may be quite a helpful result to gain better insight in cellular redox mechanisms and by this in the associated pathologies.
On the other hand redox changes could also be used to specifically target and kill cells. For instance, compounds that are able to disrupt reducing systems inside the cell may be considered as candidate drugs to sensitize cancer cells to chemo- or radiotherapy.
Can you give me a brief overview of homogenous time-resolved fluorescence (HTRF)? What is HTRF used for and why is it important?
HTRF is a TR-FRET-based technology from Cisbio Bioassays International. Time-Resolved Fluorescence Resonance Energy Transfer (TR-FRET) is a special form of resonance energy transfer (FRET) in which long emission fluorophores (lanthanides) are used as donors.
The ratiometric measurement of the two emitted wavelengths over time is used to calculate a TR-FRET response. HTRF is a flexible chemistry that measures biomolecular complexes of many different sizes. This includes assessment of small phosphorylated peptides, immunoassays for quantifying large glycoproteins, receptor tyrosine kinase activity and indirect detection of tagged complexes.
HTRF is a quite important technique because it allows for large assay windows with low background noise and high stability. As a consequence, it is one of the methods of choice for high-throughput screening. In particular, it is used in the pharmaceutical industry for compound and competition screenings.
What does BMG Labtech offer as the latest innovation in microplate readers? What features separate these readers from the rest? What are their limitations?
BMG LABTECH has always been on the cutting edge of measurement science. We can rely on more than 25 years of experience in the field and on the knowledge of our German engineers both for the development and for the construction of high-end readers.
At the moment, BMG LABTECH has in its portfolio the two most-sensitive microplate readers on the market. For the CLARIOstar, one of those two readers, BMG LABTECH reinvented the monochromator technology.
Instead of using a conventional grating-based technology with its limitations, our engineers developed a monochromator based on linear variable filters (LVF monochromator). The LVF Monochromator uses neither a concave grating nor does it mechanically break up the light.
Instead, LVF monochromators use special adjustable filters, so called linear variable filters (LVF), to select wavelengths. LVFs are defined as filters whose spectral properties vary linearly from one end of the filter to the other.
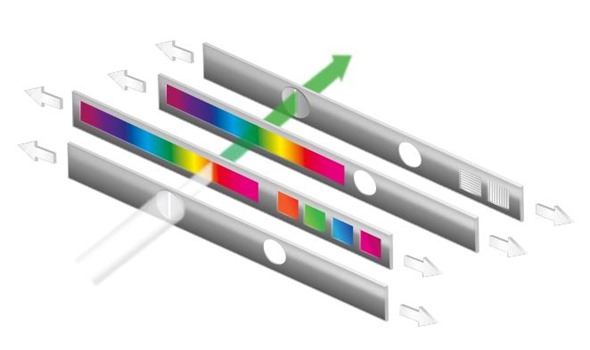
Simplified schematic of the CLARIOstar’s LVF Monochromator.
The LVF Monochromators™ consist of two LVFs that form the rising and falling edge of a filter. By moving the LVF slides relative to each other, the system separates light into distinct wavelengths and continuously adjustable bandwidths.
The CLARIOstar reader consists of two LVF Monochromators, one for excitation and one for emission. In addition, a Linear Variable Dichroic Mirror slide is used to separate the excitation from the emission light and further reduce undesired wavelengths.
The CLARIOstar is the most-sensitive monochromator-based plate reader on the market and has so far been a success story.
At the start of this year, we launched our new high-end plate reader specifically dedicated to high-throughput screening, the PHERAstar FSX. The PHERAstar FSX is the evolution of the PHERAstar FS, another success story, especially in the screening community.
Introducing the PHERAstar FSX at SLAS 2016
The PHERAstar FSX comes with technical innovations such as an ultra-fast excitation laser for TR-FRET that allows for the measurement of a 1536-well plate in 27 seconds. Moreover, it has the ability to detect simultaneously multiplex AlphaLISA-AlphaPlex assays thanks to Simultaneous Dual Emission, and has the best sensitivities on the market for fluorescence intensity and polarization.
The limitations are the ones that every reader manufacturer has to fight against. The tendency is towards upgradeable multi-mode readers as they are a better return of investment compared to a single mode reader, have a smaller footprint, and allow multiplex detection e.g: combined absorbance and fluorescence detection of one sample.
The down side, at least for the manufacturer, is that to combine multiple detection technologies you have to agree upon a compromise between them. A dedicated single mode reader has always the capability to be a more sensitive reader than a multi-mode one because it is fully dedicated and optimized for only one purpose.
Nevertheless, thanks to the continuous technological development, multi-mode readers are closing this gap.
What does the future hold for the PHERAstar FSX reader and other BMG Labtech microplate readers?
We are very positive! The beta-testers or customers that were allowed to test a PHERAstar FSX prototype were quite enthusiastic! The first readers will be delivered in April and we are looking forward to hearing the feedback of our customers.
As for our other instruments, we are always thinking at ways how to improve, make readers more intuitive and easy to use, and fulfill the requirements of the scientists. At the moment we are further developing the Atmospheric Control Unit of the CLARIOstar that will be upgraded with further capabilities.
Where can readers find more information?
Readers can find more information about our instruments and a quite extensive collection of application notes on our website www.bmglabtech.com.
Through social media (Facebook, Linkedin, Twitter) you can always be in touch with us and get updated information about events, new application notes and product releases.
About Tobias Pusterla

Tobias Pusterla is the international marketing manager with BMG LABTECH. He studied biotechnology at the University of Milan, Italy. After obtaining a Ph.D. in Molecular and Cellular Biology, he worked as a post-doc researcher at the German Cancer Research Center (DKFZ) in Heidelberg, Germany.
He started his career at the microplate reader manufacturer BMG LABTECH as a product specialist. In 2014 he took over the management of the International Marketing division.