The hygiene hypothesis refers to the idea that decreased exposure to certain infectious agents (because of better hygiene) is the reason why we have seen an increase in inflammatory diseases in the developed world.
For example, maybe because we are less infected by parasitic worms, our immune system does not develop the way it is supposed to.
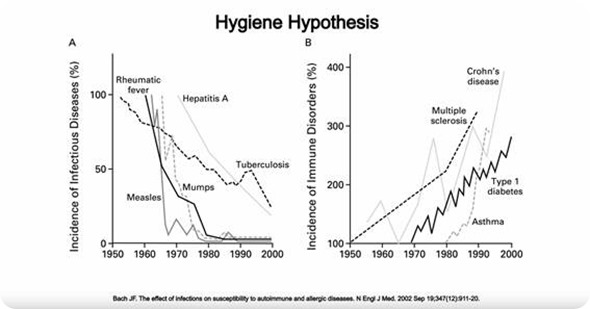
How does the global distribution and prevalence of worm infections compare to IBD?
The places in the world in which you have high incidence of IBD, like U.S. and Western Europe, are the opposite of the places where you have high incidence of worm infections, like South East Asia.
How did your recent study test the link between parasites and bacteria and the origin of IBD?
We tested whether parasite infection can treat disease in mice that have a mutation in NOD2, a gene that is linked to IBD in humans.
These NOD2 mutant mice develop disease because they have an imbalance in their intestinal bacterial populations (the gut microbiome). Therefore, we also looked at the effect of the parasite infection on intestinal bacteria.
Worm infection counters intestinal inflammation by changing gut microbiome
Additionally, we looked at indigenous people in Malaysia, who are infected with parasites and generally do not develop IBD, to examine what the effect of parasite infection on intestinal bacteria in humans.
What were the main findings of your research?
We found that parasite infection reversed IBD in NOD2 mutant mice, and that it did this by changing the content of the intestinal bacteria.
Specifically, it led to an increase in beneficial bacteria called Clostridiales, and these beneficial bacteria reduced the number of disease-causing bacteria called Bacteroides.
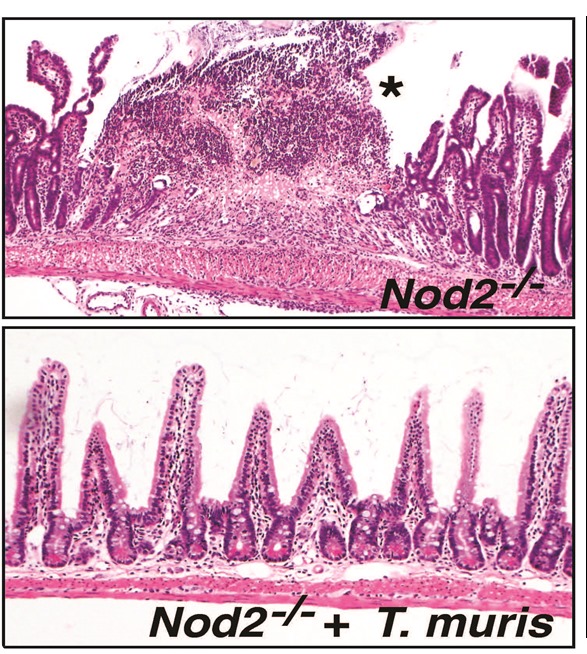
We then looked at the indigenous people in Malaysia and observed the same changes in their intestinal bacterial populations.
When the individuals were infected with parasites they had an increase in Clostridiales and a decrease in Bacteroides.
Is it the effect on the gut bacteria rather than the worms directly that aid IBD?
In our system, this seems to be the case. The worms are causing changes in the gut bacterial community, which then determines the disease outcome.
Can you outline how this research builds on Dr Loke’s previous study that found giving worm eggs to monkeys protected them from the simian version of IBD?
In our previous study on monkeys with colitis, we found that after worm infection, the monkeys bacteria were more similar to healthy monkeys. This was observational. We did not identify specific types of bacteria that could help restore the healthy communities.
In this research, we were able to identify bacteria of the Clostridia class that could restore healthy gut communities and show that the worms were helping the Clostridia to grow and expand.
Worm infection counters intestinal inflammation by changing gut microbiome
What impact could your research have for scientists and physicians looking for new ways to treat Crohn’s and ulcerative colitis?
There are efforts to develop Clostridia as probiotic bacteria for treatment of CD and UC. Our results indicate that the environment would affect the colonization of these bacteria. A mucus rich environment may help the Clostridia to colonize the gut.
Also, it is important to think about genotypes when treating CD or UC patients. Our results indicate that maybe worms and Clostridia would be most beneficial for people carrying the Nod2 polymorphism, perhaps less so for other CD and UC patients.
Alternatively, they may be more beneficial for people who have inflammatory Bacteorides bacteria.
Do you think your research could have implications for other autoimmune diseases like type 1 diabetes and multiple sclerosis?
Dysbiosis of the gut bacterial communities is a common feature of many autoimmune diseases, but all of these diseases are very complicated and we need to work out the specific details for every disease.
What do you think the future holds for IBD patients?
We’ve seen a lot of breakthroughs in “personalized medicine” for other diseases like certain types of cancer. Our research, along with many other findings in the field, are suggesting that similar approaches will be possible to treat IBD.
What further research is needed?
We are trying to understand how Clostridia can outcompete Bacteroides bacteria. We are trying to understand how the immune response to the worms may help repair the gut tissue, in addition to changing the bacterial communities. We are wondering if the Clostridia from helminth colonized people are more potent as probiotics.
Where can readers find more information?
We have written a number of articles on Helminths and gut bacteria that can be found online, by searching for our names.
About Dr Loke and Dr Cadwell
Dr. Ken Cadwell received his Ph.D. from University of California, Berkeley, and performed his postdoctoral research at Washington University School of Medicine where he investigated how gene-microbe interactions can lead to inflammatory bowel disease (IBD). He joined the Skirball Institute and the Department of Microbiology at New York University School of Medicine in 2010 as an assistant professor. His current research is focused on understanding the how immune responses to an infection are regulated. Notable contributions include the discovery that there are beneficial viruses in the intestine, which have similar roles in health and disease as bacteria that are part of the microbiome. For this and other research accomplishments, Dr. Cadwell has received the Dale F. Frey Award for Breakthrough Scientists from the Damon Runyon Foundation, the ICAAC Young Investigator Award from the American Society for Microbiology, and the Investigator in the Pathogenesis of Infectious Disease award from the Burroughs Wellcome Fund.
Dr. P’ng Loke completed his Ph.D. at the University of Edinburgh with Judith Allen and did postdoctoral training with Dr. James Allison at UC Berkeley and at UCSF with Dr. Jim McKerrow, supported by an NRSA fellowship from the NIH. He then received a Career Development Award and started his independent group as an Assistant Research Immunologist at the Division of Experimental Medicine at UCSF. In 2009, he moved to New York University School of Medicine, where he is now an Associate Professor in the Department of Microbiology. His lab studies the immune response to helminth infections and focuses in particular on the effects of helminths on the gut microbiota and macrophage biology.