Move over, salamanders, we humans can also regrow some of our body tissues. At least, this is what a new study published on October 9, 2019, in the journal Science Advances, reports. Using a mechanism quite similar to that by which amphibians like salamanders, and some zebrafish, grow back lost body parts, human joint cartilage can also regenerate itself.
This ability is more marked in the ankle joints compared to the hip joints. Much more work is needed, but if proved to be so, this study could mean a revolution in the treatment of osteoarthritis – which is the most common joint condition in the world.
Researcher Virgina Byers Kraus says, “An understanding of this ‘salamander-like’ regenerative capacity in humans, and the critically missing components of this regulatory circuit, could provide the foundation for new approaches to repair joint tissues and possibly whole human limbs.”

Image Credit: Ferencik / Shutterstock
Determining protein turnover
The researchers invented a method of assessing protein age. This was by taking advantage of the internal clock in all amino acids, by means of which the molecules shift form from one to the other with absolute regularity. The specific chemical change observed here is deamidation, or the loss of an amide group on the side chains of certain amino acids. This is an irreversible conversion. Once this protein is replaced by newly created proteins, the deamidated protein will be accumulated inside the cell.
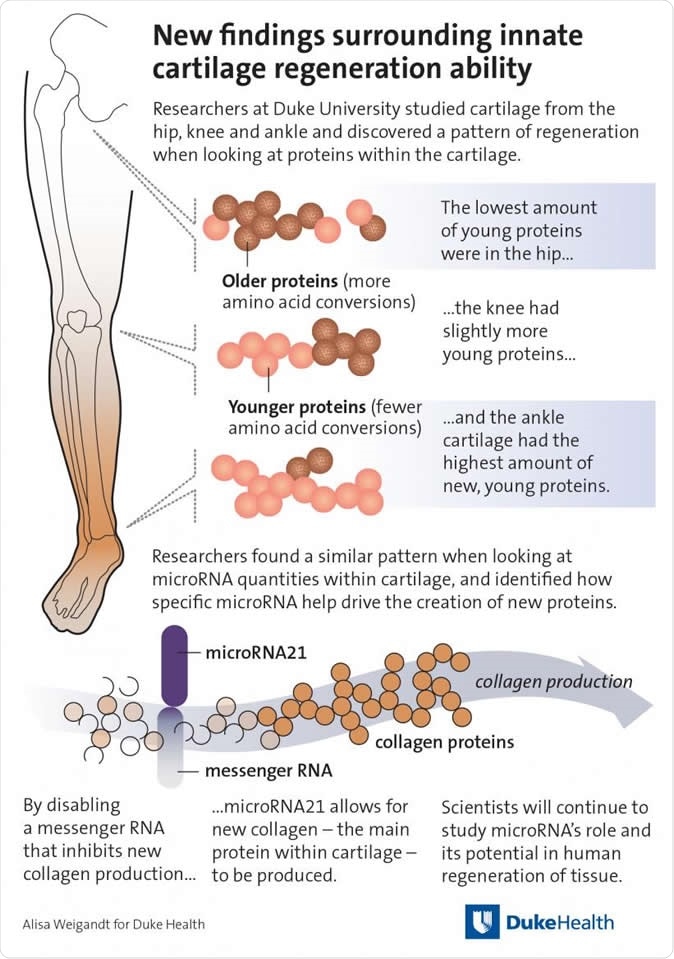
To put this in perspective, we know that proteins that have been just recently synthesized do not show many chemical conversions, while older proteins show multiple conversions, both deamidations and many other non-enzyme-mediated modifications. This enables scientists to find out with great sensitivity, using mass spectrometry, the rate of protein turnover and repair. By finding out the age of important proteins in the human cartilage, such as the various types of collagen, they can fix its age with reasonable certainty, even for proteins that survive for only relatively short periods, because of the fast deamidation of amino acids like asparagine compared to glutamine. This technique is far superior to earlier methods based on the occurrence of racemes or light-polarizing isomers of aspartate, because the latter is extremely slow and requires ultra-purified proteins.
Gradient of protein turnover
Using this method, the scientists found that cartilage age varied with its location in the body. In the ankles, young cartilage is found (with the highest content of non-deamidated protein), but middle-aged at the knee, and old at the hips. This agrees with the speed and promptness with which limb regeneration occurs in salamanders and some other species (African freshwater fish, zebrafish, and lizards) – with the greatest ease at the limb tips, and the slowest speed at the proximal joints.
Another confirmation comes from clinical experience – people with knee and hip injuries take a long time to heal, while ankle injuries are known to heal rapidly. Similarly, hip and knee arthritis following injury are well-known sequelae, but are rare with ankle damage.
miRNA and the repair process
The mediators of this process are microRNAs (miRNAs), which are, again, known to be more active in animals that have a reputation for being able to repair their limbs, fins or tails following injury. These are responsible for suppressing genes that inhibit the synthesis of cartilage proteins. They promote blastema formation, an embryonic structure which is an essential prerequisite for limb regeneration. Human miRNAs carry on this capacity to repair the joints, with the highest activity being in the ankles, then the knees and hips. Not only so, the most superficial and therefore newest layer of cartilage shows greater blastemal miRNA activity compared to the deeper layers. This shows that there is a limited but very real regenerative capacity in human cartilage.
Implications
Investigator Ming-Feng Hsueh says, “We were excited to learn that the regulators of regeneration in the salamander limb appear to also be the controllers of joint tissue repair in the human limb.” The researchers say this capacity, which they have dubbed our “inner salamanders”, could help them develop novel miRNAs to treat, arrest or reverse arthritis. They look forward to boosting the level of activity of miRNA in arthritis joints to achieve complete repair of damaged and broken-down joint cartilage. Moreover, they want to find out the missing component. Kraus comments wistfully, “If we can figure out what regulators we are missing compared with salamanders, we might even be able to add the missing components back and develop a way someday to regenerate part or all of an injured human limb.”
Moreover, this repair pathway could be used to repair multiple tissues as it is a basic cellular mechanism. The linked pathways are implicated in a wide array of disease conditions in humans, including osteoarthritis. Knowing more about the mechanisms of limb regeneration in animals could help uncover parallel processes in human beings. This could in turn help form future treatments for osteoarthritis. Another possibility is the use of injected miRNA in a joint to boost its repair capability, and to bolster it against damage, whether due to accumulated stress or a single major injury. Tissue engineering could also use this concept to increase the rate of production of tissue outside the construct before it is transplanted into the body. The immediate focus is to validate the current study and to find out more missing pieces of the puzzle that prevent complete repair in humans. This could finally lead to the development of a “molecular cocktail” to help humans grow new limbs, after all.
Journal reference:
Analysis of “old” proteins unmasks dynamic gradient of cartilage turnover in human limbs. Ming-Feng Hsueh, Patrik Önnerfjord, Michael P. Bolognesi, Mark E. Easley and Virginia B. Kraus. Science Advances. 09 Oct 2019: Vol. 5, no. 10, eaax3203. DOI: 10.1126/sciadv.aax3203. https://advances.sciencemag.org/content/5/10/eaax3203